Abstract
Introduction
Trypanosomiasis caused by Trypanosoma vivax (T. vivax, subgenus Duttonella) is a burden disease in bovines that induces losses of billions of dollars in livestock activity worldwide. To control the disease, the first step is identifying the infected animals at early stages. However, convention tools for animal infection detection by T. vivax present some challenges, facilitating the spread of the pathogenesis.
Objectives
This work aims to develop a new procedure to identify infected bovines by T. vivax using cerumen (earwax) in a volatilomic approach, here named cerumenolomic, which is performed in an easy, quick, accurate, and non-invasive manner.
Methods
Seventy-eight earwax samples from Brazilian Curraleiro Pé-Duro calves were collected in a longitudinal study protocol during health and inoculated stages. The samples were analyzed using Headspace/Gas Chromatography–Mass Spectrometry followed by multivariate analysis approaches.
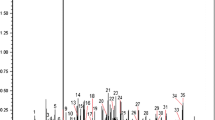
Results
The cerumen analyses lead to the identification of a broad spectrum of volatile organic metabolites (VOMs), of which 20 VOMs can discriminate between healthy and infected calves (AUC = 0.991, sensitivity = 0.967, specificity = 1.000). Furthermore, 13 VOMs can indicate a pattern of discrimination between the acute and chronic phases of the T. vivax infection in the animals (AUC = 0.989, sensitivity = 0.944, specificity = 1.000).
Conclusion
The cerumen volatile metabolites present alterations in their occurrence during the T.vivax infection, which may lead to identifying the infection in the first weeks of inoculation and discriminating between the acute and chronic phases of the illness. These results may be a breakthrough to avoid the T. vivax outbreak and provide a faster clinical approach to the animal.




Similar content being viewed by others
Data availability
The GC–MS raw data files (.qgd extension) are freely available on the Mendeley Data repository: https://doi.org/10.17632/66hzt799fb.1
Code availability
The datasets and R script used in this work are freely available on GitHub: https://github.com/Barbosa-JMG/Metabolomics.git
References
Aittokallio, T., & Schwikowski, B. (2006). Graph-based methods for analysing networks in cell biology. Briefings in Bioinformatics, 7(3), 243–255. https://doi.org/10.1093/bib/bbl022
Barbosa, J. M. G., Pereira, N. Z., David, L. C., de Oliveira, C. G., Soares, M. F. G., Avelino, M. A. G., et al. (2019). Cerumenogram: A new frontier in cancer diagnosis in humans. Scientific Reports, 9(1), 11722. https://doi.org/10.1038/s41598-019-48121-4
Barbosa, J. M., Fernandes Rodrigues, M. K., David, L. C., e Silva, T. C., Fortuna Lima, D. A., Pereira, N. Z., et al. (2020). A volatolomic approach using cerumen as biofluid to diagnose bovine intoxication by Stryphnodendron rotundifolium. Biomedical Chromatography. https://doi.org/10.1002/bmc.4935
Bastos, T. S. A., Faria, A. M., Madrid, D. M. de C., de Bessa, L. C., Linahres, G. F. C., Fidelis Junior, O. L., et al. (2017). First outbreak and subsequent cases of Trypanosoma vivax in the state of Goiás, Brazil. Brazilian Journal of Veterinary Parasitology, 26(3), 366–371. https://doi.org/10.1590/S1984-29612017019
Bastos, T. S. A., Faria, A. M., Cavalcante, A. S. de A., Madrid, D. M. de C., Zapa, D. M. B., Nicaretta, J. E., et al. (2020). Infection capacity of Trypanosoma vivax experimentally inoculated through different routes in bovines with latent Anaplasma marginale. Experimental Parasitology, 211, 107861. https://doi.org/10.1016/j.exppara.2020.107861
Boudonck, K. J., Mitchell, M. W., Wulff, J., & Ryals, J. A. (2009). Characterization of the biochemical variability of bovine milk using metabolomics. Metabolomics, 5(4), 375–386. https://doi.org/10.1007/s11306-009-0160-8
Boulangé, A., Pillay, D., Chevtzoff, C., Biteau, N., Comé de Graça, V., Rempeters, L., et al. (2017). Development of a rapid antibody test for point-of-care diagnosis of animal African trypanosomosis. Veterinary Parasitology, 233, 32–38. https://doi.org/10.1016/j.vetpar.2016.11.017
Broadhurst, D., Goodacre, R., Jones, A., Rowland, J. J., & Kell, D. B. (1997). Genetic algorithms as a method for variable selection in multiple linear regression and partial least squares regression, with applications to pyrolysis mass spectrometry. Analytica Chimica Acta, 348(1–3), 71–86. https://doi.org/10.1016/S0003-2670(97)00065-2
Burkhart, C. N., Kruge, M. A., Burkhart, C. G., & Black, C. (2001). Cerumen composition by flash pyrolysis-gas chromatography/mass spectrometry. Otology and Neurotology, 22(6), 715–722. https://doi.org/10.1097/00129492-200111000-00002
Cadioli, F. A., Fidelis Junior, O. L., Sampaio, P. H., dos Santos, G. N., André, M. R., de Almeida Castilho, K. J. G., & Machado, R. Z. (2015). Detection of Trypanosoma vivax using PCR and LAMP during aparasitemic periods. Veterinary Parasitology, 214(1–2), 174–177. https://doi.org/10.1016/j.vetpar.2015.09.001
Cevallos-Cevallos, J. M., Danyluk, M. D., & Reyes-De-Corcuera, J. I. (2011). GC-MS based metabolomics for rapid simultaneous detection of Escherichia coli O157:H7, Salmonella typhimurium, Salmonella muenchen, and Salmonella hartford in ground beef and chicken. Journal of Food Science, 76(4), 238–246. https://doi.org/10.1111/j.1750-3841.2011.02132.x
Chong, J., Soufan, O., Li, C., Caraus, I., Li, S., Bourque, G., et al. (2018). MetaboAnalyst 4.0: Towards more transparent and integrative metabolomics analysis. Nucleic Acids Research, 46(W1), W486–W494. https://doi.org/10.1093/nar/gky310
Chong, J., Wishart, D. S., & Xia, J. (2019). Using MetaboAnalyst 4.0 for comprehensive and integrative metabolomics data analysis. Current Protocols in Bioinformatics, 68(1), 1–128. https://doi.org/10.1002/cpbi.86
Clark, M., Murray, J. D., & Maga, E. A. (2014). Assessing unintended effects of a mammary-specific transgene at the whole animal level in host and non-target animals. Transgenic Research, 23(2), 245–256. https://doi.org/10.1007/s11248-013-9768-6
Couch, R. D., Navarro, K., Sikaroodi, M., Gillevet, P., Forsyth, C. B., Mutlu, E., et al. (2013). The approach to sample acquisition and its impact on the derived human fecal microbiome and VOC metabolome. PLoS ONE, 8(11), 1–13. https://doi.org/10.1371/journal.pone.0081163
Dagnachew, S., & Bezie, M. (2015). Review on Trypanosoma vivax. African Journal of Basic & Applied Sciences, 7(1), 41–64. https://doi.org/10.5829/idosi.ajbas.2015.7.1.92116
Dai, W. T., Wang, Q. J., Zou, Y. X., White, R. R., Liu, J. X., & Liu, H. Y. (2017). Short communication: Comparative proteomic analysis of the lactating and nonlactating bovine mammary gland. Journal of Dairy Science, 100(7), 5928–5935. https://doi.org/10.3168/jds.2016-12366
Dávila, A. M. R., & Silva, R. A. M. S. (2000). Animal trypanosomiasis in South America: Current status, partnership, and information technology. Annals of the New York Academy of Sciences, 916, 199–212. https://doi.org/10.1111/j.1749-6632.2000.tb05291.x
De Buck, J., Shaykhutdinov, R., Barkema, H. W., & Vogel, H. J. (2014). Metabolomic profiling in cattle experimentally infected with mycobacterium avium subsp. paratuberculosis. PLoS ONE. https://doi.org/10.1371/journal.pone.0111872
De Lacy Costello, B., Amann, A., Al-Kateb, H., Flynn, C., Filipiak, W., Khalid, T., et al. (2014). A review of the volatiles from the healthy human body. Journal of Breath Research. https://doi.org/10.1088/1752-7155/8/1/014001
De Preter, V., Van Staeyen, G., Esser, D., Rutgeerts, P., & Verbeke, K. (2009). Development of a screening method to determine the pattern of fermentation metabolites in faecal samples using on-line purge-and-trap gas chromatographic-mass spectrometric analysis. Journal of Chromatography A, 1216(9), 1476–1483. https://doi.org/10.1016/j.chroma.2008.12.095
Di Cagno, R., De Angelis, M., De Pasquale, I., Ndagijimana, M., Vernocchi, P., Ricciuti, P., et al. (2011). Duodenal and faecal microbiota of celiac children: Molecular, phenotype and metabolome characterization. BMC Microbiology, 11, 1–21. https://doi.org/10.1186/1471-2180-11-219
Dixon, E., Clubb, C., Pittman, S., Ammann, L., Rasheed, Z., Kazmi, N., et al. (2011). Solid-phase microextraction and the human fecal VOC metabolome. PLoS ONE, 6(4), 1–9. https://doi.org/10.1371/journal.pone.0018471
Fidelis Junior, O. L., Sampaio, P. H., Gonçalves, L. R., André, M. R., Machado, R. Z., Wijffels, G., & Cadioli, F. A. (2019). Comparison of conventional and molecular techniques for Trypanosoma vivax diagnosis in experimentally infected cattle. Revista Brasileira De Parasitologia Veterinaria, 28(2), 203–209. https://doi.org/10.1590/s1984-29612019034
Fiehn, O. (2002). Metabolomics–the link between genotypes and phenotypes. Plant Molecular Biology, 48(1–2), 155–171. https://doi.org/10.1023/A:1013713905833
Filipiak, W., Mochalski, P., Filipiak, A., Ager, C., Cumeras, R., Davis, E. C., et al. (2016). A compendium of volatile organic compounds (VOCs) released by human cell lines. Current Medicinal Chemistry, 23(20), 2112–2131. https://doi.org/10.2174/0929867323666160510122913
Galili, T., Benjamini, Y., Gavin Simpson, Jefferis, G., Gallotta, M., Renaudie, J., et al. (2020). Package ‘dendextend.’ Retrieved from https://cran.r-project.org/web/packages/dendextend/dendextend.pdf.
Gardner, D. R., Welch, K. D., Panter, K. E., Cook, D., dos Santos, F. C., Green, B. T., et al. (2018). An evaluation of hair, oral fluid, earwax, and nasal mucus as non-invasive specimens to determine livestock exposure to teratogenic lupine species. Journal of Agricultural and Food Chemistry, 67(1), 43–49. https://doi.org/10.1021/acs.jafc.8b05673
Garner, C. E., Smith, S., de Lacy Costello, B., White, P., Spencer, R., Probert, C. S. J., & Ratcliffe, N. M. (2007). Volatile organic compounds from feces and their potential for diagnosis of gastrointestinal disease. The FASEB Journal, 21(8), 1675–1688. https://doi.org/10.1096/fj.06-6927com
Giordani, F., Morrison, L. J., Rowan, T. G., De Koning, H. P., & Barrett, M. P. (2016). The animal trypanosomiases and their chemotherapy: A review. Parasitology, 143(14), 1862–1889. https://doi.org/10.1017/S0031182016001268
Goldansaz, S. A., Guo, A. C., Sajed, T., Steele, M. A., Plastow, G. S., & Wishart, D. S. (2017). Livestock metabolomics and the livestock metabolome: A systematic review. PLoS ONE, 12(5), 1–26. https://doi.org/10.1371/journal.pone.0177675
Gómez, E., Caamaño, J. N., Corrales, F. J., Díez, C., Correia-Álvarez, E., Martín, D., et al. (2013). Embryonic sex induces differential expression of proteins in bovine uterine fluid. Journal of Proteome Research, 12(3), 1199–1210. https://doi.org/10.1021/pr300845e
Gonçalves Barbosa, J. M., Machado Botelho, A. F., Santana da Silva, R. H., Ferreira de Almeida, S. S., Ferreira, E. R., Caetano David, L., et al. (2020). Identification of cattle poisoning by Bifenthrin via earwax analysis by HS/GC–MS. Biomedical Chromatography. https://doi.org/10.1002/bmc.5017
Gonzatti, M. I., González-Baradat, B., Aso, P. M., & Reyna-Bello, A. (2014). Trypanosoma (Duttonella) vivax and typanosomosis in Latin America: Secadera/Huequera/Cacho Hueco. Springer. https://doi.org/10.1007/978-3-7091-1556-5_11
Gray, L. R., Tompkins, S. C., & Taylor, E. B. (2014). Regulation of pyruvate metabolism and human disease. Cellular and Molecular Life Sciences, 71(14), 2577–2604. https://doi.org/10.1007/s00018-013-1539-2
Han, X., Xiao, H., Chen, J., Xu, W., & Dou, D. (2020). The substance basis of Poria ameliorates hypothyroidism other than hyperthyroidism based on proteomics and metabolomics. FASEB Journal, 34(9), 11970–11982. https://doi.org/10.1096/fj.202000984R
Heimbucher, T., Hog, J., Gupta, P., & Murphy, C. T. (2020). PQM-1 controls hypoxic survival via regulation of lipid metabolism. Nature Communications. https://doi.org/10.1038/s41467-020-18369-w
Hough, R., Archer, D., & Probert, C. (2018). A comparison of sample preparation methods for extracting volatile organic compounds (VOCs) from equine faeces using HS-SPME. Metabolomics, 14(2), 1–10. https://doi.org/10.1007/s11306-017-1315-7
Huang, P. C., Kuo, P. L., Guo, Y. L., Liao, P. C., & Lee, C. C. (2007). Associations between urinary phthalate monoesters and thyroid hormones in pregnant women. Human Reproduction, 22(10), 2715–2722. https://doi.org/10.1093/humrep/dem205
Jackson, A. P., Goyard, S., Xia, D., Foth, B. J., Sanders, M., Wastling, J. M., et al. (2015). Global gene expression profiling through the complete life cycle of Trypanosoma vivax. PLoS Neglected Tropical Diseases, 9(8), 1–29. https://doi.org/10.1371/journal.pntd.0003975
Jensen, R. G. (2002). The composition of bovine milk lipids: January 1995 to December 2000. Journal of Dairy Science. https://doi.org/10.3168/jds.S0022-0302(02)74079-4
Jiménez-Pacheco, A., Salinero-Bachiller, M., Iribar, M. C., López-Luque, A., Miján-Ortiz, J. L., & Peinado, J. M. (2018). Furan and p-xylene as candidate biomarkers for prostate cancer. Urologic Oncology: Seminars and Original Investigations, 36(5), 243.e21-243.e27. https://doi.org/10.1016/j.urolonc.2017.12.026
Kanehisa, M. (2019). Toward understanding the origin and evolution of cellular organisms. Protein Science, 28(11), 1947–1951. https://doi.org/10.1002/pro.3715
Kanehisa, M., & Goto, S. (2000). KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Research, 28(1), 27–30. https://doi.org/10.1093/nar/28.1.27
Kanehisa, M., Sato, Y., Furumichi, M., Morishima, K., & Tanabe, M. (2019). New approach for understanding genome variations in KEGG. Nucleic Acids Research, 47(D1), D590–D595. https://doi.org/10.1093/nar/gky962
Katajamaa, M., & Orešič, M. (2007). Data processing for mass spectrometry-based metabolomics. Journal of Chromatography A, 1158(1–2), 318–328. https://doi.org/10.1016/j.chroma.2007.04.021
Keenan, T. W., James Morré, D., Olson, D. E., Yunghans, W. N., & Patton, S. (1970). Biochemical and morphological comparison of plasma membrane and milk fat globule membrane from bovine mammary gland. Journal of Cell Biology, 44(1), 80–93. https://doi.org/10.1083/jcb.44.1.80
Kell, D. B., & Oliver, S. G. (2016). The metabolome 18 years on: A concept comes of age. Metabolomics, 12(10), 1–8. https://doi.org/10.1007/s11306-016-1108-4
Kuhn, M., Wing, J., Weston, S., Williams, A., Keefer, C., Engelhardt, A., et al. (2022). Package ‘caret’: classification and regression training. Retrieved from https://github.com/topepo/caret/.
Leardi, R., & Lupiáñez González, A. (1998). Genetic algorithms applied to feature selection in PLS regression: How and when to use them. Chemometrics and Intelligent Laboratory Systems, 41(2), 195–207. https://doi.org/10.1016/S0169-7439(98)00051-3
Lee, J. H. J., Jayaprakasha, G. K., Rush, C. M., Crosby, K. M., & Patil, B. S. (2018). Production system influences volatile biomarkers in tomato. Metabolomics, 14(7), 1–13. https://doi.org/10.1007/s11306-018-1385-1
Liland, K. H., Mehmood, T., & Sæbø, S. (2020). Package ‘ plsVarSel ’—variable selection in partial least squares. Retrieved from https://cran.r-project.org/web/packages/plsVarSel/plsVarSel.pdf.
Maher, A. D., Hayes, B., Cocks, B., Marett, L., Wales, W. J., & Rochfort, S. J. (2013). Latent biochemical relationships in the blood-milk metabolic axis of dairy cows revealed by statistical integration of 1H NMR spectroscopic data. Journal of Proteome Research, 12(3), 1428–1435. https://doi.org/10.1021/pr301056q
Meier, S. I., Koelzer, S. C., Schubert-Zsilavecz, M., & Toennes, S. W. (2017). Analysis of drugs of abuse in Cerumen—correlation of postmortem analysis results with those for blood, urine and hair. Drug Testing and Analysis, 9(10), 1572–1585. https://doi.org/10.1002/dta.2177
Menezes, E. B., Velho, A. L. C., Santos, F., Dinh, T., Kaya, A., Topper, E., et al. (2019). Uncovering sperm metabolome to discover biomarkers for bull fertility. BMC Genomics, 20(1), 1–16. https://doi.org/10.1186/s12864-019-6074-6
Meyer, D., Dimitriadou, E., Hornik, K., Weingessel, A., Leisch, F., Chang, C.-C., & Lin, C.-C. (2019). Package ‘e1071’: Misc Functions of the Department of Statistics, Probability Theory Group (Formerly: E1071), TU Wien. R package version 1.7–3. Retrieved from https://cran.r-project.org/package=e1071.
Miakar, A., & Spiteller, G. (1994). Reinvestigation of lipid peroxidation of linolenic acid. Biochimica Et Biophysica Acta (BBA)/Lipids and Lipid Metabolism, 1214(2), 209–220. https://doi.org/10.1016/0005-2760(94)90046-9
Osorio, M. T., Downey, G., Moloney, A. P., Röhrle, F. T., Luciano, G., Schmidt, O., & Monahan, F. J. (2013). Beef authentication using dietary markers : Chemometric selection and modelling of significant beef biomarkers using concatenated data from multiple analytical methods. Food Chemistry, 141(3), 2795–2801. https://doi.org/10.1016/j.foodchem.2013.05.118
Paradis, E., Blomberg, S., Bolker, B., Brown, J., Claramunt, S., Claude, J., & Cuong, H. S. (2020). Package ‘ape’: Analyses of phylogenetics and evolution. Retrievedfrom http://ape-package.ird.fr/.
Parveen, I., Moorby, J. M., Fraser, M. D., Allison, G. G., & Kopka, J. (2007). Application of gas chromatography mass—spectrometry metabolite profiling techniques to the analysis of heathland plant diets of sheep. Journal of Agricultural and Food Chemistry, 55(4), 1129–1138. https://doi.org/10.1021/jf062995w
R Core Team. (2020). R: A language and environment for statistical computing. Vienna, Austria: R Foundation for Statistical Computing. Retrieved from https://www.r-project.org/.
RStudio Team. (2015). RStudio: Integrated Development Environment for R. Boston, MA: RStudio, Inc. Retrieved from http://www.rstudio.com/.
Saleem, F., Bouatra, S., Guo, A. C., Psychogios, N., Mandal, R., Dunn, S. M., et al. (2013). The bovine ruminal fluid metabolome. Metabolomics, 9(2), 360–378. https://doi.org/10.1007/s11306-012-0458-9
Salek, R. M., Steinbeck, C., Viant, M. R., Goodacre, R., & Dunn, W. B. (2013). The role of reporting standards for metabolite annotation and identification in metabolomic studies. GigaScience, 2(1), 1. https://doi.org/10.1186/2047-217X-2-13
Shaw, A. P. M., Cecchi, G., Wint, G. R. W., Mattioli, R. C., & Robinson, T. P. (2014). Mapping the economic benefits to livestock keepers from intervening against bovine trypanosomosis in Eastern Africa. Preventive Veterinary Medicine, 113(2), 197–210. https://doi.org/10.1016/j.prevetmed.2013.10.024
Shokry, E., de Oliveira, A. E., Avelino, M. A. G., de Deus, M. M., & Filho, N. R. A. (2017a). Earwax: A neglected body secretion or a step ahead in clinical diagnosis? A pilot study. Journal of Proteomics, 159, 92–101. https://doi.org/10.1016/j.jprot.2017.03.005
Shokry, E., de Oliveira, A. E., Avelino, M. A. G., de Deus, M. M., Pereira, N. Z., & Filho, N. R. A. (2017b). Earwax: An innovative tool for assessment of tobacco use or exposure. A pilot study in young adults. Forensic Toxicology, 35(2), 389–398. https://doi.org/10.1007/s11419-017-0370-0
Shokry, E., dos Santos, F. C., da Cunha, P. H. J., Fioravanti, M. C. S., Noronha Filho, A. D. F., Pereira, N. Z., & Antoniosi Filho, N. R. (2017c). Earwax: A clue to discover fluoroacetate intoxication in cattle. Toxicon, 137, 54–57. https://doi.org/10.1016/j.toxicon.2017.07.014
Shokry, E., Marques, J. G., Ragazzo, P. C., Pereira, N. Z., & Filho, N. R. A. (2017d). Earwax as an alternative specimen for forensic analysis. Forensic Toxicology, 35(2), 348–358. https://doi.org/10.1007/s11419-017-0363-z
Shokry, E., Pereira, J., Marques Júnior, J. G., Da Cunha, P. H. J., Noronha Filho, A. D. F., Da Silva, J. A., et al. (2017e). Earwax metabolomics: An innovative pilot metabolic profiling study for assessing metabolic changes in ewes during periparturition period. PLoS ONE, 12(8), 1–22. https://doi.org/10.1371/journal.pone.0183538
Silva Pereira, S., de Almeida Castilho Neto, K. J. G., Duffy, C. W., Richards, P., Noyes, H., Ogugo, M., et al. (2020). Variant antigen diversity in Trypanosoma vivax is not driven by recombination. Nature Communications, 11(1), 1–13. https://doi.org/10.1038/s41467-020-14575-8
Smolinska, A., Klaassen, E. M. M., Dallinga, J. W., Van De Kant, K. D. G., Jobsis, Q., Moonen, E. J. C., et al. (2014). Profiling of volatile organic compounds in exhaled breath as a strategy to find early predictive signatures of asthma in children. PLoS ONE. https://doi.org/10.1371/journal.pone.0095668
Soini, H. A., Klouckova, I., Wiesler, D., Oberzaucher, E., Grammer, K., Dixon, S. J., et al. (2010). Analysis of volatile organic compounds in human saliva by a static sorptive extraction method and gas chromatography-mass spectrometry. Journal of Chemical Ecology, 36(9), 1035–1042. https://doi.org/10.1007/s10886-010-9846-7
Stonecipher, C. A., Lee, S. T., Green, B. T., Cook, D., Welch, K. D., Pfister, J. A., & Gardner, D. R. (2019). Evaluation of non-invasive specimens to diagnose livestock exposure to toxic larkspur (Delphinium spp.). Toxicon, 161, 33–39. https://doi.org/10.1016/j.toxicon.2019.02.013
Sun, Y., Xu, C., Li, C., Xia, C., Xu, C., Wu, L., & Zhang, H. (2014). Characterization of the serum metabolic profile of dairy cows with milk fever using 1H-NMR spectroscopy. Veterinary Quarterly, 34(3), 159–163. https://doi.org/10.1080/01652176.2014.924642
Worton, D. R., Decker, M., Isaacman-VanWertz, G., Chan, A. W. H., Wilson, K. R., & Goldstein, A. H. (2017). Improved molecular level identification of organic compounds using comprehensive two-dimensional chromatography, dual ionization energies and high resolution mass spectrometry. The Analyst, 142(13), 2395–2403. https://doi.org/10.1039/c7an00625j
Xia, J., Wishart, D. S., & Valencia, A. (2011). MetPA: A web-based metabolomics tool for pathway analysis and visualization. Bioinformatics, 27(13), 2342–2344. https://doi.org/10.1093/bioinformatics/btq418
Xia, J., Broadhurst, D. I., Wilson, M., & Wishart, D. S. (2013). Translational biomarker discovery in clinical metabolomics: An introductory tutorial. Metabolomics, 9(2), 280–299. https://doi.org/10.1007/s11306-012-0482-9
Xia, L., Zhao, Z., Yang, R., Jiang, P., Liu, Y., Yu, H., et al. (2020). miR-2382-5p regulates lipid metabolism by targeting NDRG2 in mammary epithelial cells of dairy cattle. DNA and Cell Biology, 00(00), 1–9. https://doi.org/10.1089/dna.2020.5658
Xu, Y., Zhao, T., Ren, H., Xie, Y., An, J., Shang, J., et al. (2020). Urinary metabolic profiling via LC-MS/MS reveals impact of bovine lactoferrin on bone formation in growing sd rats. Nutrients. https://doi.org/10.3390/nu12041116
Zhang, H., Wu, L., Xu, C., Xia, C., Sun, L., & Shu, S. (2013). Plasma metabolomic profiling of dairy cows affected with ketosis using gas chromatography/mass spectrometry. BMC Veterinary Research. https://doi.org/10.1186/1746-6148-9-186
Funding
This work was supported by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior— Brazil (CAPES)—Finance Code 001—for fellowship to J.M.G.B. (Process Number: 88882.386430/2019-01); the research productivity Grant to N.R.A.F. and M.C.S.F. by the Conselho Nacional de Desenvolvimento Científico e Tecnológico, Brazil (CNPQ Number 312280/2016-5 and 407774/2013-0); and the management of financial resources by the Fundação de Apoio à Pesquisa (FUNAPE), Universidade Federal de Goiás.
Author information
Authors and Affiliations
Contributions
N.R.A.F. is responsible for the original idea and the analysis conditions for the use of cerumen for clinical diagnoses in humans and animals. J.M.G.B., D.R.M., W.D.Z.L., M.C.S.F., P.H.J.C., and N.R.A.F. developed the work conceptualization, visualization, and study design. D.R.M., W.D.Z.L., M.C.S.F., and P.H.J.C. performed the biochemical exams and clinical evaluation of the animals. J.M.G.B., L.C.D., T.C.S., and D.A.F.L. realized the earwax sample collection and HS/GC–MS analysis. J.M.G.B., A.E.O., and N.R.A.F. conducted the software analysis and interpretation. J.M.G.B. wrote the original draft. J.M.G.B., L.C.D., A.E.O., P.H.J.C., W.D.Z.L., M.C.S.F., and N.R.A.F. performed the formal investigation, data curation, and writing review and editing. N.R.A.F. provided the funding acquisition and project administration. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All the authors declare no conflict of interest.
Ethical approval
This study was approved by the ethical standards of the local ethical committee at the Universidade Federal de Goiás, Brazil (protocol numbers: 090/20 and 027/16). All applicable international, national, and institutional guidelines for the care and use of animals were strictly followed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Barbosa, J.M.G., de Mendonça, D.R., David, L.C. et al. A cerumenolomic approach to bovine trypanosomosis diagnosis. Metabolomics 18, 42 (2022). https://doi.org/10.1007/s11306-022-01901-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-022-01901-y