Abstract
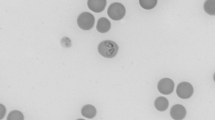
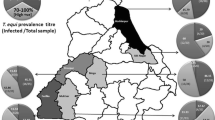
Piroplasmid protozoa Theileria equi and Babesia caballi and zoonotic rickettsial bacterium Anaplasma phagocytophilum are important agents of equine vector-borne diseases (EVBD). This study aimed at investigating the prevalence of infections with or exposure to these pathogens in horses from the north of Portugal. Blood was randomly collected from 162 horses, living in 72 different stables, to prepare Giemsa-stained slide smears. Additionally, plasma samples were tested for antibodies to T. equi and B. caballi by two competitive enzyme-linked immunosorbent assays and to A. phagocytophilum by an indirect fluorescence antibody test. Five horses were positive to T. equi by microscopy (3.1 %), three to B. caballi (1.9 %), and none to A. phagocytophilum with no horse simultaneously positive for the two piroplasms. Clinically suspect animals had a significantly higher positivity to T. equi by microscopy in comparison with the nonsuspect ones (21.4 vs. 1.4 %). Twenty-nine horses were seropositive to T. equi (17.9 %), 18 to B. caballi (11.1 %), and 21 to A. phagocytophilum (13.0 %). Combined serology and microscopy positive results to T. equi and B. caballi were 19.1 and 11.7 %, respectively, with 33.3 % of the horses found positive to at least one agent. Forty horses were positive to single agents and 14 to more than one agent. An outdoor or mixed outdoor/indoor type of housing was found to be a risk factor for the combined positivity to T. equi. Infections with T. equi, B. caballi, and A. phagocytophilum are endemic in the north of Portugal. In addition to the treatment of positive horses, preventive measures should be put in practice to reduce exposure to and infection with agents of EVBD.



Similar content being viewed by others
References
Abutarbush SM, Alqawasmeh DM, Mukbel RM, Al-Majali AM (2011) Equine babesiosis: seroprevalence, risk factors and comparison of different diagnostic methods in Jordan. Transbound Emerg Dis 59:72–78
Acici M, Umur S, Guvenc T, Arslan HH, Kurt M (2008) Seroprevalence of equine babesiosis in the Black Sea region of Turkey. Parasitol Int 57:198–200
Alhassan A, Thekisoe OMM, Yokoyama N, Inoue N, Motloang MY, Mbati PA, Yin H, Katayama Y, Anzai T, Sugimoto C, Igarashi I (2007) Development of loop-mediated isothermal amplification (LAMP) method for diagnosis of equine piroplasmosis. Vet Parasitol 143:155–160
Asgarali Z, Coombs DK, Mohammed F, Campbell MD, Caesar E (2007) A serological study of Babesia caballi and Theileria equi in Thoroughbreds in Trinidad. Vet Parasitol 144:167–171
Bashiruddin JB, Cammà C, Rebêlo E (1998) Molecular detection of Babesia equi and Babesia caballi in horse blood by PCR amplification of part of the 16S rRNA gene. Vet Parasitol 84:75–83
Beugnet F, Mariè J-L (2009) Emerging arthropod-borne diseases of companion animals in Europe. Vet Parasitol 163:298–305
Brüning A (1996) Equine piroplasmosis an update on diagnosis, treatment and prevention. Br Vet J 152:139–151
Butler CM, Nijhof AM, Jongejan F, van der Kolk JH (2008) Anaplasma phagocytophilum infection in horses in the Netherlands. Vet Rec 162:216–217
Caeiro V (1999) General review of tick species present in Portugal. Parassitologia 4(1):11–15
Camacho AT, Guitian FJ, Pallas E, Gestal JJ, Olmeda AS, Habela MA, Telford SR III, Spielman A (2005) Theileria (Babesia) equi and Babesia caballi infections in horses in Galicia, Spain. Trop Anim Health Prod 37:293–302
Dantas-Torres F, Chomel BB, Otranto D (2012) Ticks and tick-borne diseases: a One Health perspective. Trends Parasitol 28:437–446
de Waal DT (1992) Equine piroplasmosis: a review. Br Vet J 148:6–14
Doudier B, Olano J, Parola P, Brouqui P (2010) Factors contributing to emergence of Ehrlichia and Anaplasma spp. as human pathogens. Vet Parasitol 167:149–154
Dumler JS, Barbet AC, Bekker CPJ, Dasch GA, Palmer GH, Ray SC, Rikihisa Y, Rurangirwa FR (2001) Reorganization of genera in the families Rickettsiaceae and Anaplasmataceae in the order Rickettsiales: unification of some species of Ehrlichia with Anaplasma, Cowdria with Ehrlichia and Ehrlichia with Neorickettsia, descriptions of six new species combinations and designation of Ehrlichia equi and ‘HGE agent’ as subjective synonyms of Ehrlichia phagocytophila. Int J Syst Evol Microbiol 51:2145–2165
Dumler JS, Choi KS, Garcia-Garcia JC, Barat NS, Scorpio DG, Garyu JW, Grab DJ, Bakken JS (2005) Human granulocytic anaplasmosis and Anaplasma phagocytophilum. Emerg Infect Dis 11:1828–1834
Franzén P, Aspan A, Egenvall A, Gunnarsson A, Karlstam E, Pringle J (2009) Molecular evidence for persistence of Anaplasma phagocytophilum in the absence of clinical abnormalities in horses after recovery from acute experimental infection. J Vet Intern Med 23:636–642
Friedhoff KT, Tenter AM, Müller I (1990) Haemoparasites of equines: impact on international trade of horses. Rev Sci Tech 9:1187–1194
Fritz D (2010) A PCR study of piroplasms in 166 dogs and 111 horses in France (March 2006 to March 2008). Parasitol Res 106:1339–1342
Heim A, Passos LMF, Ribeiro MFB, Costa-Júnior LM, Bastos CV, Cabral DD, Hirzmann J, Pfister K (2007) Detection and molecular characterization of Babesia caballi and Theileria equi isolates from endemic areas of Brazil. Parasitol Res 102:63–68
Jaffer O, Abdishakur F, Hakimuddin F, Riya A, Wernery U, Schuster RK (2010) A comparative study of serological tests and PCR for the diagnosis of equine piroplasmosis. Parasitol Res 106:709–713
Kouam MK, Kantzoura V, Gajadhar AA, Theis JH, Papadopoulos E, Theodoropoulos G (2010) Seroprevalence of equine piroplasms and host-related factors associated with infection in Greece. Vet Parasitol 169:273–278
Leblond A, Pradier S, Pitel PH, Fortier G, Boireau P, Chadoeuf J, Sabatier P (2005) An epidemiological survey of equine anaplasmosis (Anaplasma phagocytophilum) in Southern France. Rev Sci Tech 24:899–908
Lewis SR, Zimmerman K, Dascanio JJ, Pleasant RS, Witonsky SG (2009) Equine granulocytic anaplasmosis: a case report and review. J Equine Vet Sci 29:160–166
Lillini E, Macrì G, Proietti G, Scarpulla M (2006) New findings on anaplasmosis caused by infection with Anaplasma phagocytophilum. Ann N Y Acad Sci 1081:360–370
Madigan JE (1993) Equine ehrlichiosis. Vet Clin Equine 9:423–428
Malta MV (2001) Diagnóstico de babesiose e theileriose equinas no Alentejo. Dissertation, Technical University of Lisbon
Moretti A, Mangili V, Salvatori R, Maresca C, Scoccia E, Torina A, Moretta I, Gabrielli S, Tampieri MP, Pietrobelli M (2010) Prevalence and diagnosis of Babesia and Theileria infections in horses in Italy: a preliminary study. Vet J 184:346–350
Nováková M, Víchová B (2010) Granulocytic anaplasmosis—emerging tick-borne disease of humans and animals. Biologia 65:925–931
OIE (2008) Equine piroplasmosis. In: Manual of diagnostic tests and vaccines for terrestrial animals. Chapter 2. 5. 8. OIE, Paris, pp 884–893
Passamonti F, Veronesi F, Cappelli K, Capomaccio S, Coppola G, Marenzoni ML, Piergili FD, Verini SA, Coletti M (2010) Anaplasma phagocytophilum in horses and ticks: a preliminary survey of Central Italy. Comp Immunol Microbiol Infect Dis 33:73–83
Penzhorn BL (2011) Why is Southern African canine babesiosis so virulent? An evolutionary perspective. Parasit Vectors 4:51
Petrie A, Watson P (1999) Statistics for veterinary and animal science. Blackwell, Oxford
Pitel PH, Pronost S, Scrive T, Léon A, Richard E, Fortier G (2010) Molecular detection of Theileria equi and Babesia caballi in the bone marrow of asymptomatic horses. Vet Parasitol 170:182–184
Rashid HB, Chaudhry M, Rashid H, Pervez K, Khan MA, Mahmood AK (2008) Comparative efficacy of diminazene diaceturate and diminazene aceturate for the treatment of babesiosis in horses. Trop Anim Health Prod 40:463–467
Santos AS, Bacellar F, Dumler JS (2008) A 4-year study of Anaplasma phagocytophilum in Portugal. Clin Microbiol Infect 15(2):46–47
Sellon DC (2007) Miscellaneous parasitic diseases. In: Sellon DC, Long MT (eds) Equine infectious diseases. Saunders, St. Louis, pp 473–480
Steinman A, Zimmerman T, Klement E, Lensky IM, Berlin D, Gottlieb Y, Baneth G (2012) Demographic and environmental risk factors for infection by Theileria equi in 590 horses in Israel. Vet Parasitol 187:558–562
Sousa S, Almeida A, Simão L, Anastácio S, Madeira de Carvalho LM (2010) Theleria equi infection in Puro Sangue Lusitano horses from Vila Viçosa. In: Proceedings of the II Iberian Congress of Veterinary Epidemiology, Barcelona (P.19)
von Loewenich FD, Stumpf G, Baumgarten BU, Röllinghoff M, Dumler JS, Bogdan C (2003) A case of equine granulocytic ehrlichiosis provides molecular evidence for the presence of pathogenic Anaplasma phagocytophilum (HGE agent) in Germany. Eur J Clin Microbiol Infect Dis 22:303–305
Woldehiwet Z (2010) The natural history of Anaplasma phagocytophilum. Vet Parasitol 167:108–122
Acknowledgments
The authors would like to express their gratitude to Prof. P.A. Rodrigues (UTAD) for her assistance with map design and to Dr. J. Gomes (National Laboratory for Veterinary Research, INIAV, Ministry of Agriculture, Portugal) for his advice on the serological testing for A. phagocytophilum.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ana J. Ribeiro and Luís Cardoso contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ribeiro, A.J., Cardoso, L., Maia, J.M. et al. Prevalence of Theileria equi, Babesia caballi, and Anaplasma phagocytophilum in horses from the north of Portugal. Parasitol Res 112, 2611–2617 (2013). https://doi.org/10.1007/s00436-013-3429-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-013-3429-9