Abstract
Cytokines play an important role in the immunological pathogenesis of schistosomiasis. Schistosomiasis would be associated with an imbalance in inflammatory cytokines that leads to a decrease of T helper (Th) 1 and an increase of Th2 cytokine secretion. Corresponding data so far have been inconsistent, so we performed a meta-analysis to assess whether cytokine alterations were risk factors for schistosomiasis progression. We searched MEDLINE, EMBASE, and CNKI databases for literatures including abstracts, reviews, and reference lists. Our studies included assessment of cytokine concentrations in vivo plasma or serum and secretion of cytokines in vitro by peripheral blood leukocytes from schistosomiasis patients or infected individuals with schistosome. The prototypic Th1 and Th2 cytokines IFN-γ and interleukin (IL)-4 were assessed as well as IL-13 and tumor necrosis factor-alpha (TNF-α). The results implied that an increase occurs in TNF-α and IL-4 with schistosomiasis progression.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Schistosomiasis continued to be one of the major public health problems affecting millions of people living in tropical and subtropical regions. Over 200 million people were infected with these parasites worldwide. According to a national epidemiological sampling survey, the average prevalence was 2.5% in all surveyed endemic areas (Zhou et al. 2007). To treat the schistosomiasis with repeated praziquantel might probably lead to resistance of schistosome against praziquantel. So far, no genuine effective vaccines were obtained, and it was urgent to get more knowledge about the immune evasion mechanism of schistosome to advance the vaccine development for schistosoma infection or schistosomiasis. Most cytokines were involved in the regulation of inflammatory and immunologic responses in pathologic and physiologic conditions in schistosomiasis (van Die et al. 2010).
Cytokines are a large number of proteins in which most exert overlapping and pleiotropic effects through interaction with specific receptors expressed on different target cells. Cytokine receptors also exist in soluble forms, such as the soluble tumor necrosis factor receptors (sTNFRs). sTNFR-II and soluble intracellular adhesive molecule I are associate with schistosomiasis progression (Ellis et al. 2008). The cytokine network plays an important role in coordinate responses of cells of the innate system. The interferon-gamma (IFN-γ) and tumor necrosis factor-alpha (TNF-α) were considered pro-inflammatory to augment the immune response to infection by promoting leukocyte recruitment to inflammatory site or by activating inflammatory cells. Interleukin (IL)-4 is an anti-inflammatory cytokine that contributes to dampen the inflammatory and immune response.
The CD4+ T helper (Th) cells used to have different identification of Th1 and Th2. The Th1 cells produce IFN-γ and IL-4 was a Th2 prototypic cytokine. IL-13 was produced by Th2 lymphocytes also and promoted Th2 responses. It was effective to induce chemokines, especially CC chemokine receptor-3 (CCR3)-acting chemokines including eotaxin and eotaxin-2. CCR3-acting chemokines themselves may promote the migration of Th2 lymphocytes which produce IL-13.(Rothenberg et al. 2003) Schistosomiasis might be associated with an imbalance in Th1/Th2 cytokine, with a shift toward the Th2 system (Acosta et al. 2002). Some studies about cytokine alteration such as IFN-γ showed a different trend (Aly et al. 2010); due to lack of consistency of results, this meta-analysis aims to verify whether the different cytokine imbalances were substantiated by evidence.
Material and methods
Identification of eligible studies and data extraction
A search of computerized literature databases (Pubmed, EMBASE, and CNKI) was conducted with the following keywords: “schistosomiasis,” “cytokine,” “interleukin,” “interferon,” “IFN-γ,” “Il-4,” “IL-13,” and “TNF-α.” No language restrictions were applied. A study was included in the analysis if (1) it was published up to May 2011, (2) it was not related to a previous publication, and (3) studies that assessed in vivo plasma or serum cytokine concentrations or in vitro secretion of cytokines by peripheral blood leukocytes from schistosomiasis patients stimulated by soluble egg antigen (SEA) or soluble adult worm antigen preparation (SWAP). For the same group, use the same stimulation.
Some research data showed the detectable number of people inside the study group when comparing with the control group. Others gave the detected indicator numerical value or graphs for different groups. Both reaction number of people and the change of the numerical value are also vital for the correlation of the cytokine and the disease. So, we saved the dichotomous data for cytokines IFN-γ, IL-4, and IL-13 using the detectable number of participants and total number for experimental and control group. At the same time, the continuous data for TNF-α focusing on mean, standard deviation, and number of people for different groups were kept. We excluded studies if (1) studies used animal models (e.g., mice, rats, rabbits, or pigs), (2) studies assessed cytokine genes or immune markers, and (3) studies had no control group.
Statistical analysis
The detectable numbers of the IFN-γ, IL-4, and IL-13 from the respective studies were drawn out of the data. Odds ratio (OR) was used as the point estimates of risk; it was calculated along with 95% confidence intervals (CI) for each study. Heterogeneity across studies was assessed through I 2 statistic. The OR was obtained by using Mantel–Haenszel method for IFN-γ (I 2 = 76%, P = 0.002), IL-4 (I 2 = 73%, P = 0.001), and IL-13 (I 2 = 69%, P = 0.02) using the random effect model. All analyses with IFN-γ, IL-4, or IL-13 were carried out in respect to the detectable participants’ number of cytokines.
We extracted data (n, mean, and SD) with cytokine TNF-α, and the inverse variance methods can be used to pool all mean differences (MD) and standardized mean differences for continuous data. Two reviewers independently extracted all the data to avoid potential mistakes. If there are discrepancies, discussions are done to reach a consensus. All comparisons were analyzed with Review Manager 5.
Results
A total of 491 studies were identified; of those, 478 studies were rejected for the following reasons: (1) animal model, e.g., mice, rat, >281studies; (2) methodology, e.g., also having other diseases, assessing genes or markers, >150 studies; and (3) the data of interleukin could not be ascertained or incomplete, 47 studies. In total, 13 studies met our research criteria and were available for meta-analysis. The total sample size of 1,080 subjects was categorized as 598 and 482 subjects in the experimental and control group, respectively. On the basis of 13 studies included in the analysis, the ORs of three cytokines including the prototypic Th1 and Th2 cytokines IFN-γ and IL-4 were estimated as well as the following inflammatory cytokine IL-13. Differently, the MD of TNF-α was analyzed also.
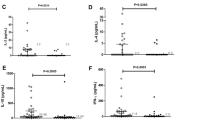
Finally, to evaluate IFN-γ with schistosomiasis, five relevant papers were selected and included in this research. Many more data were rejected because the studies used animal models. Table 1 and Fig. 1a indicate that there was a decreasing trend of IFN-γ in schistosomiasis. However, this was not statistically significant according to the remaining data (pooled OR = 0.58, 95% CI = 0.27–1.25, P>0.10). The results will be more accurate if more data are available. For the same reasons, 82 studies were rejected from the total 86 studies of IL-13. Table 2 and Fig. 1c show that there was no obvious correlation between IL-13 with schistosomiasis (pooled OR = 0.96, 95% CI = 0.28–3.24).
There were remaining seven studies from IL-4 studies data. Table 3 and Fig. 1b suggest that there was a statistical difference on the association between the increasing of IL-4 with schistosomiasis (pooled OR = 2.06, 95% CI = 0.95–4.47, P<0.10). A total of eight studies were selected at first to evaluate TNF-α. Then, three studies were excluded from the analysis because of incomplete data. When comparing the remaining five studies, we found an association of the increasing of TNF-α with schistosomiasis, as showed in Table 4 and Fig. 2d (MD = 5.03, 95% CI = 2.59–7.47).
When we performed the analysis excluding the data on the cytokines with hepatic fibrosis in human schistosoma infection, we found out that the exclusion affects the correlation with cytokine alteration with the schistosomiasis that contained several subjects. The analyses lost a lot of power due to the exclusion, and as a result, the sample size was small. Nevertheless, the result of the meta-analysis also showed the trend of the cytokines’ influence with schistosomiasis.
Discussion
Some experimental studies in mice model indicated that schistosome egg-induced granuloma formation and hepatic fibrosis depend markedly on cytokine regulation (de Morais et al. 2010; Liang et al. 2011; Shen et al. 2010).However, there was no clear consensus about the pattern of cytokine production and the adjustment net. Sometimes, chronic patients developed severe hepatosplenic disease with periportal fibrosis and portal hypertension. In the early stages of infection, the hepatic fibrosis (HS) progression was associated with type 2 cytokines (Mutapi et al. 2010).
Some studies indicated that HS disease was characterized by a predominant T helper 1 profile (Hau et al. 2000). De Morais performed one research which showed that after SWAP stimulation, chronic patients, particularly hepatointestinals, produced higher levels of IL-4 in comparison with acute patients, suggesting the presence of a type 2 cytokine profile in these patients. Following SEA and SWAP stimulation, HS patients showed increased levels of IFN-gamma when compared with acute patients, indicating that HS disease in humans was also associated with a type 1 cytokine response (de Morais et al. 2008).
The current meta-analysis was conducted to verify whether the cytokine imbalance and Th2 predomination of human schistosomiasis were substantiated by evidence. The result suggested an increase in peripheral levels of TNF-α and IL-4. No significant difference was obtained for IL-13 and IFN-γ statistically. These results contributed to the hypothesis of a shift from Th1 to Th2 cytokines on the basis of studies showing increase in TNF-α and IL-4 and the trend of decreasing of IFN-γ shown by the OR value. New classification of T cell such as CD4+ CD25+ Treg cells which excreted IL-10, TGF-β, and Th17 cell excreting IL-17 was involved in the research of adjustment net of cytokines in schistosomiasis nearly in an animal model (el Ridi et al. 2009). The data about these adjusted factors such as IL-17 in schistosomiasis were still insufficient.
The increase in peripheral levels of TNF-α and IL-4 in schistosomiasis patients as shown in this meta-analysis provided evidence of immune alteration and Th2 polarization in schistosomiasis as pointed out by previous studies (Hau et al. 2007; Silveira et al. 2004; van Die et al. 2010). Some studies assessed combination occurrence of decreased IFN-γ (Gattass et al. 2002). Although OR value showed the trend, there was no significant difference statistically. Similar to IFN-γ, no obvious association was observed for IL-13 with schistosomiasis. Further studies with large sample size are needed.
Alterations in the cytokine network could be related to the physiopathology of schistosomiasis. Sandrine Henri pointed out that TNF-α was positively associated with periportal fibrosis, and high TNF-α levels were associated with a risk of FII–III on average four times higher in the high TNF-α producers than in low TNF-α producers (Dessein et al. 2002). Despite the different pathological phrases, heterogeneity remained a concern. Some uncontrolled factors including age, gender, type of schistosomiasis, duration of this disease, and dietary habits are all potentially influencing the immune system. Scott’s study showed that children and adults tended to respond differently to a schistosome antigen (Scott et al. 2004).
Meta-analysis was a very powerful tool when correctly used and can provide a consensus answer from different sources when studying the same research problem. Also, the meta-analysis comprised limitations. However, some limitations may be applied. First, some data were not available or some data could not be retrieved. Another potential limitation was heterogeneity; for example, there was no consideration about the methods of cytokine assessment, the storing time for samples, and different stages of disease which could influence cytokine levels and the final results.
To our knowledge, this was the first meta-analysis that was conducted on cytokine alterations in schistosomiasis. Although a number of studies have been performed on cytokine research, the number included was small. In the case of TNF-α and IL-4, the results indicated that TNF-α and IL-4 were increasing in schistosomiasis. Although Elsammak showed that schsitosomal infection triggered a Th2 type immune response as indicated by the high plasma IL-4 (Elsammak et al. 2008), yet, no clear cytokine net mechanism has been suggested. TNF-α and IL-10 had also been found to modulate the in vitro granuloma reaction in humans or in experimental schistosomiasis (Silveira-Lemos et al. 2008). Perhaps, the impaired IL-10 immunoregulatory function and IL-10-producing Tregs also played an important role in cytokine networks in schistosomiasis.
References
Acosta LP, Waine G, Aligui GDL, Tiu WU, Olveda RM, McManus DP (2002) Immune correlate study on human Schistosoma japonicum in a well-defined population in Leyte, Philippines: II. Cellular immune responses to S. japonicum recombinant and native antigens. Acta Trop 84:137–149
Aly IRB, Hendawy MA, Ali E, Hassan E, Nosseir MMF (2010) Immunological and parasitological parameters after treatment with dexamethasone in murine Schistosoma mansoni. Memorias Do Instituto Oswaldo Cruz 105:729–735
Caiweimin, Sunyongliang, Zhang L, Liu R, Yuyaxin, Yanghanqing, Zhulizhuan (1996) Assay and clinical signification of 12 parameters of serum in patients with hepatic fibrosis of schistosomiasis. Chinese Journal of zoonoses 12:13–15
de Morais CNL, de Souza JR, Melo WG, Aroucha ML, Miranda P, Domingues ALC, Abath FGC, Montenegro SML (2008) Cytokine profile associated with chronic and acute human Schistosomiasis mansoni. Memorias Do Instituto Oswaldo Cruz 103:561–568
de Morais CNL, Carvalho BD, de Melo WG, de Melo FL, Lopes EPD, Domingues ALC, Juca N, Martins JRM, Diniz GTN, Montenegro SML (2010) Correlation of biological serum markers with the degree of hepatic fibrosis and necroinflammatory activity in hepatitis C and schistosomiasis patients. Memorias Do Instituto Oswaldo Cruz 105:460–466
Dessein AJ, Henri S, Chevillard C, Mergani A, Paris P, Gaudart J, Camilla C, Dessein H, Montero F, Elwali NEMA, Saeed OK, Magzoub M (2002) Cytokine regulation of periportal fibrosis in humans infected with Schistosoma mansoni: IFN-gamma is associated with protection against fibrosis and TNF-alpha with aggravation of disease. J Immunol 169:929–936
Elsammak MY, Al-Sharkaweey RM, Ragab MS, Amin GA, Kandil MH (2008) IL-4 and reactive oxygen species are elevated in Egyptian patients affected with schistosomal liver disease. Parasite Immunol 30:603–609
El Ridi R, Tallima H, Salah M, Guirguis FR (2009) Transforming growth factor-beta and Th17 responses in resistance to primary murine schistosomiasis mansoni. Cytokine 48:239–245
Ellis MK, Li YS, Hou XY, Chen HG, McManus DP (2008) sTNFR-II and sICAM-1 are associated with acute disease and hepatic inflammation in schistosomiasis japonica. Int J Parasitol 38:717–723
Ferrari TCA, Moreira PRR, Sampaio MJ, da Cunha AS, de Oliveira JT, Gazzinelli G, Correa-Oliveira R (2006) Intrathecal cytokines in spinal cord schistosomiasis. J Neuroimmunol 177:136–141
Gattass CR, Pyrrho AD, Ramos JA, Neto RM, da Silva CS, Lenzi HL, Takiya CM (2002) Dexamethasone, a drug for attenuation of Schistosoma mansoni infection morbidity. Antimicrob Agents Chemother 46:3490–3498
Hau J, Farah IO, Johansson M, Lovgren-Bengtson K (2000) Schistosoma mansoni in mice: the pattern of primary cercarial exposure determines whether a secondary infection post-chemotherapy elicits a T helper 1- or a T helper 2-associated immune response. Scand J Immunol 51:237–243
Hau J, Farah IO, Langoi D, Nyaundi J (2007) Schistosome-induced pathology is exacerbated and Th2 polarization is enhanced during pregnancy. In Vivo 21:599–602
Li W, Chen Y, Liu C, Yu D (2002) Detection of THF-α, IL-βand NO in sera of patients with schistosomiasis japonicum. Chongqing medicine 31:1157–1158
Liang YJ, Luo J, Yuan Q, Zheng D, Liu YP, Shi L, Zhou Y, Chen AL, Ren YY, Sun KY, Sun Y, Wang Y, Zhang ZS (2011) New insight into the antifibrotic effects of praziquantel on mice in infection with Schistosoma japonicum. PLoS One 6(5):e20247
Milner T, Reilly L, Nausch N, Midzi N, Mduluza T, Maizels R, Mutapi F (2010) Circulating cytokine levels and antibody responses to human Schistosoma haematobium: IL-5 and IL-10 levels depend upon age and infection status. Parasite Immunol 32:710–721
Mutapi F, Milner T, Reilly L, Nausch N, Midzi N, Mduluza T, Maizels R (2010) Circulating cytokine levels and antibody responses to human Schistosoma haematobium: IL-5 and IL-10 levels depend upon age and infection status. Parasite Immunol 32:710–721
Oliveira LFA, Moreno EC, Gazzinelli G, Martins-Filho OA, Silveira AMS, Gazzinelli A, Malaquias LCC, Loverde P, Leite PM, Correa-Oliveira R (2006) Cytokine production associated with periportal fibrosis during chronic schistosomiasis mansoni in humans. Infect Immun 74:1215–1221
Rothenberg ME, Zimmermann N, Hershey GK, Foster PS (2003) Chemokines in asthma: Cooperative interaction between chemokines and IL-13. J Allergy Clin Immun 111:227–242
Scott JT, Turner CMR, Mutapi F, Woolhouse MEJ, Ndhlovu PD, Hagan P (2001) Cytokine responses to mitogen and Schistosoma haematobium antigens are different in children with distinct infection histories. Parasite Immunol 23:519–526
Scott JT, Vereecken K, Diakhate MM, Vanagt T, Sambou V, Diop M, Ly A, Vercruysse J, Gryseels B, Kestens L (2004) Are the cellular immune responses of children and adults with Schistosoma mansoni infection intrinsically different? Cytokines produced ex vivo in response to antigens and mitogens. Parasite Immunol 26:29–36
Shen L, Wu H, Zhaosong Z, Weir R, Lijun S, Zhangwu X, Linsheng HU, Chen S, Su C, Zhang T, Taylor MG, Wu G (2000) Studies on human cytokine responses before and after praziquantel chemotherapy in an endemic area of schistosomiasis japonica. China Journal of Parasitology and Parasit Diseases 18:14–17
Shen JL, Li XY, Zhong ZR, Peng J, Wen HQ, Li J, Luo QL, Wei W (2010) Paeoniflorin ameliorates schistosomiasis liver fibrosis through regulating IL-13 and its signalling molecules in mice. Parasitology 137:1213–1225
Silveira AMS, Gazzinelli G, Alves-Oliveira LF, Bethony J, Gazzinelli A, Carvalho-Queiroz C, Alvarez MCB, Lima-Silva FC, Prata A, Loverde PT, Correa-Oliveira R (2004) Human Schistosomiasis mansoni: intensity of infection differentially affects the production of interleukin-10, interferon-gamma and interleukin-13 by soluble egg antigen or adult worm antigen stimulated cultures. Trans R Soc Trop Med Hyg 98:514–519
Silveira-Lemos D, Teixeira-Carvalho A, Martins-Filho OA, Oliveira LFA, Costa-Silva MF, Matoso LF, de Souza LJ, Gazzinelli A, Correa-Oliveira R (2008) Eosinophil activation status, cytokines and liver fibrosis in Schistosoma mansoni infected patients. Acta Trop 108:150–159
Tong Q, Luo R (2003) Tumour necrosis factor and disease of acute schistosomiasis. J Trop Med 3:79–80
van Die I, van Stijn CMW, Meyer S, van den Broek M, Bruijns SCM, van Kooyk Y, Geyer R (2010) Schistosoma mansoni worm glycolipids induce an inflammatory phenotype in human dendritic cells by cooperation of TLR4 and DC-SIGN. Mol Immunol 47:1544–1552
Violaine A, Jun L, Yuanyuan W, Xiao F, Shi M, Xinsong L, Xunya H, Helia D, Zhou J, Yu X-L, Hongbin H, Donald PM, Yuesheng L, Dessein A (2008) Regulatory role of interleukin-10 and interferon-γ in severe hepatic central and peripheral fibrosis in humans infected with Schistosoma japonicum. J Infect Dis 198:418–426
Wu H, Shen L, Zhang Zhaoso NG et al (1998) Human cytokine responses stimulated by schistosome antigens in Schistosomiasis japonica endemic area. Acta Un Iversit Atis Medicinalis Nanjing 18:472–474
Zhou N, He H, Liou X, Zhou J, Wang J (1996) The study of the clinic meaning of TNF-α of schistosomiasis patients. China Journal of Schistosomia Prevention 8:114
Zhou XN, Guo JG, Wu XH, Jiang QW, Zheng J, Dang H, Wang XH, Xu J, Zhu HQ, Wu GL, Li YS, Xu XJ, Chen HG, Wang TP, Zhu YC, Qiu DC, Dong XQ, Zhao GM, Zhang SJ, Zhao NQ, Xia G, Wang LY, Zhang SQ, Lin DD, Chen MG, Hao Y (2007) Epidemiology of schistosomiasis in the People’s Republic of China, 2004. Emerg Infect Dis 13:1470–1476
Acknowledgments
This work was supported by grants from the National Science Foundation of China (NO: 30972574, 30771888, 81000743 and 30800966) and the National ‘973’ project of China (grant no. 2007CB513102).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yu, L., Sun, X., Yang, F. et al. Inflammatory cytokines IFN-γ, IL-4, IL-13 and TNF-α alterations in schistosomiasis: a meta-analysis. Parasitol Res 110, 1547–1552 (2012). https://doi.org/10.1007/s00436-011-2661-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-011-2661-4