Abstract
Purpose
Among all primary breast tumors, malignant phyllodes tumor of the breast (MPTB) make up less than 1%. In the treatment of phyllode tumors, surgical procedures such as mastectomy and breast-conserving surgery are the mainstay. MPTB has, however, been controversial when it comes to treating it with RT. We aimed to explore the prognostic impact of RT and other clinicopathologic factors on long-term survival for patients with stage T3 or T4 malignant phyllodes tumors.
Methods
We select patients with stage T3 or T4 MPTB who qualified for the criteria between 2000 and 2018 via the Surveillance, Epidemiology, and End Results (SEER) database. We performed 1:1 propensity score matching (PSM) and Kaplan–Meier analysis to explore the role of RT in long-term survival of patients with stage T3 or T4 MPTB. A univariate and multivariate analysis of breast cancer-specific survival (BCSS) and overall survival (OS) risk factors was carried out using a Cox proportional hazards model. In addition, the nomogram graph of OS and BCSS was constructed.
Results
A total of 583 patients with stage T3 or T4 malignant phyllodes tumors were included in this study, of whom 154 (26.4%) received RT, and 429 (73.6%) were treated without RT. Before adjustment, between groups with and without RT, BCSS (p = 0.1) and OS (p = 0.212) indicated no significant difference respectively. Using of PSM, the two groups still did not differ significantly in BCSS (p = 0.552) and OS (p = 0.172). In multivariate analysis, age (p < 0.001), surgery of primary site (p < 0.001) and distant metastatic status (p < 0.001) were related to prognosis, while RT still did not affect BCSS (p = 0.877) and OS (p = 0.554).
Conclusion
Based on the SEER database analysis, the study suggests that the patients with stage T3 or T4 MPTB treated with RT after surgery didn't have significant differences in BCSS or OS compared to those not treated with RT.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Phyllodes tumor of the breast (PTB) are rare fibroepithelial neoplasms, occupies less than 1% of all primary breast tumors. They are most commonly found in women aged between 45 and 50 year (Mishra et al. 2013) and often unilateral. Based on the number of mesenchymal cells, cell heterogeneity, nuclear division, tumor margins, and the presence or absence of stroma in the tumor, the World Health Organization (WHO) divided them into three levels: benign, borderline, and malignant. Of these, malignant phyllodes tumor of the breast (MPTB) account for about one-fourth of PTBs (Zhang and Kleer 2016) and about 0.5% of all malignant breast tumors. MPTB manifests as an insidious disease that progresses rapidly. Typically, the lesions are unilateral, solitary, nodular, and painless tumor between one centimeter and 40 cm in diameter (Hawkins et al. 1992). A high rate of local recurrence and distant metastasis is characteristic of MPTB (Kapiris et al. 2001). It has been reported that benign, borderline and malignant phyllodes tumors recur at rates of 10–17%, 14–25%, 23–30% (Lu et al. 2019), respectively. For the distant metastasis, the predominant mode of metastasis of MPTB is hematogenous and rarely lymph node metastasis. Lung, bone, and abdominal viscera are the most common sites of distant disease (Parker and Harries 2001). The surgical approach is the mainstay of treatment for PTB. However, radiation therapy (RT) remains controversial in the treatment of phyllodes tumors (Chaney et al. 2000; Macdonald et al. 2006; Belkacemi et al. 2008; Pezner et al. 2008; Barth et al. 2009). In a previous study of MPTB patients, RT led to poorer treatment outcomes (Macdonald et al. 2006). In an article by Zhao et al., it was shown that RT resulted in a prolonged disease-specific survival time and overall survival time for patients with malignant tumors (Zhou et al. 2018). Therefore, despite the increasing use of RT in malignant phyllodes tumors, the role of RT is still unclear.
Therefore, based on the Surveillance, Epidemiology, and End Results (SEER) database, the aim of this study was to evaluate the impact of RT and other clinicopathologic factors on breast cancer-specific survival (BCSS) and overall survival (OS) for patients with stage T3 or T4 MPTB to explore the value of RT in patients with MPTB.
Methods
Study population
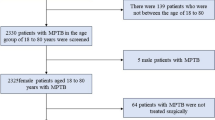
In the SEER database, we conducted a retrospective study of MPTB patients between 2000 and 2018. Patients with MPTB were required to meet the following inclusion criteria: women with stage T3 or T4 malignant phyllodes tumors (ICD-O-39020/3), diagnosed between 2000 and 2018. We excluded patients with incomplete follow-up information, such as those without information of RT. The age at diagnosis, race, marital status, laterality, T stage (T3-4), lymph node status, distant metastatic status, tumor grade, long-term survival, death status, surgery of primary site, local lymphatic biopsy, chemotherapy (CT) and RT were extracted from the SEER database. The corresponding author can provide the data.
Statistical analysis
In our study, we used Chi-square tests to compare the baseline characteristics of patients who underwent RT and those without. In order to control for selection bias, we performed one-to-one propensity score matching (PSM). The primary endpoint measures evaluated were BCSS and OS. The BCSS was regarded as the period of time between tumor diagnosis and death due to MPTB. The OS was regarded as the time between tumor diagnosis and any reason of death. Based on Kaplan–Meier analysis and log rank comparison, we compared OS and BCSS between the two groups. A univariate and multivariate analysis of OS and BCSS risk factors was carried out using a Cox proportional hazards model. In addition, the nomogram graph of OS and BCSS was constructed. The analyses were conducted using R language software. A p value < 0.05 was assumed to be statistically significant.
Results
Characteristics
A total of 583 patients with stage T3 or T4 malignant phyllodes tumors were included in this study, of whom 154 (26.4%) received RT, and 429 (73.6%) were treated without RT. As shown in Table 1, patients who received RT were more likely to receive CT (p = 0.007), biopsy of lymph nodes (p = 0.043), and be in G3 or G4 grades (p = 0.003), compared to those who did not receive RT. As a result of PSM balancing the differences in characteristics, 154 pairs of patients were analyzed (Table 1). The difference between the two groups was not significant.
Survival analysis of radiotherapy
Before adjustment (Fig. 1a, b), neither BCSS (p = 0.1) nor OS (p = 0.212) differed significantly between groups with and without RT. Using of PSM (Fig. 1c, d), the two groups still did not differ significantly in BCSS (p = 0.552) and OS (p = 0.172). Similarly, among the multivariate Cox regression analysis, RT still had no effect on BCSS (p = 0.877) and OS (p = 0.554). Apparently, in this study, the patients with stage T3 or T4 MPTB did not benefit from RT in terms of OS and BCSS.
COX regression analysis
An univariate Cox regression analysis (Table 2) showed the following results for BCSS: higher grade of tumor differentiation (p = 0.009, HR = 3.32, 95% CI = 1.35–8.19), T stage (p < 0.001, HR = 3.38, 95% CI = 2.21–5.18), lymph node status (p = 0.005, HR = 3.23, 95% CI = 1.42–7.35) distant metastasis (p < 0.001, HR = 0.03, 95% CI = 0.02–0.06), local lymphatic biopsy (p = 0.045, HR = 1.46, 95% CI = 1.01–2.10), surgery of primary site (p < 0.001, HR = 0.03, 95% CI = 0.01–0.08 in BCS and p < 0.001, HR = 0.08, 95%CI = 0.03–0.20 in mastectomy), CT (p < 0.001, HR = 4.45, 95% CI = 2.58–7.66) were related to BCSS. In addition, multivariate Cox regression analysis (Table 2) indicated that distant metastasis (p < 0.001, HR = 0.071, 95% CI = 0.034–0.147), surgery of primary site (p < 0.001, HR = 0.059, 95% CI = 0.018–0.200 in BCS and p < 0.001, HR = 0.119, 95%CI = 0.038–0.368 in mastectomy) were independent risk factors for BCSS.
An univariate Cox regression analysis (Table 3) displayed the following results for OS that age (p < 0.001, HR = 1.82, 95% CI = 1.33–2.48), T stage (p < 0.001, HR = 2.97, 95% CI = 2.06–4.29), distant metastasis (p < 0.001, HR = 0.03, 95% CI = 0.02–0.06), lymph node status (p = 0.003, HR = 2.93, 95% CI = 1.44–5.98), local lymphatic biopsy (p = 0.002, HR = 1.59, 95% CI = 1.18–2.14), surgery of primary site (p < 0.001, HR = 0.04, 95% CI = 0.01–0.09 in breast-conserving surgery (BCS) and p < 0.001, HR = 0.10, 95% CI = 0.04–0.24 in mastectomy) and CT (p < 0.001, HR = 4.44, 95% CI = 2.81–7.02) were related to OS. Additionally, the multivariate Cox regression analysis (Table 3) revealed that age (p < 0.001, HR = 1.799, 95% CI = 1.292–2.504), distant metastasis (p < 0.001, HR = 0.079, 95% CI = 0.041–0.051), surgery of primary site (p < 0.001, HR = 0.051, 95% CI = 0.017–0.154 in BCS and p < 0.001, HR = 0.106, 95% CI = 0.037–0.302 in mastectomy) were independent risk factors for OS.
Construction of the prognostic model for OS and BCSS
With the aim of predicting the 1-, 3-, and 5-years OS and BCSS for patients with stage T3 or T4 MPTB, we combined age, T stage, distant metastases, surgical removal of the primary site, radiotherapy, and chemotherapy. Prognostication factors for BCSS (Fig. 2) and OS (Fig. 3) can be scored on a score scale in the model. Patients with stage T3 or T4 MPTB can be predicted to have an overall survival of 1-, 3-, and 5-years based on the total of these scores. In one case, a middle-aged woman with MPTB and a stage T4 tumor had a BCSS and OS score of 70 and 60, respectively. Consequently, OS rates were assessed to be 85%, 68%, and 57% for patient’s 1-, 3-, and 5-years individually. Similarly, BCSS rates were assessed to be 82%, 65%, and 54% for patient’s 1-, 3-, and 5-years individually.
Discussion
Among all primary breast tumors, MPTB make up less than 1%. In clinical practice, phyllodes tumors are characterized by a single, mobile, round tumor with painless, progressive enlargement, and are often the main reason for patients visit. According to statistics, the number of patients with MPTB more than doubled from 830 to 1980 in the 11 years from 2002 to 2013 (Macdonald et al. 2006). It seems that patients with MPTB are growing in number. The mainstays of curative treatment of phyllodes tumors are surgical procedures, such as breast-conserving surgery (BCS) and mastectomy. In spite of this, MPTB are associated with a high risk of local recurrence. In a study of 5,530 patients with phyllodes tumors, the overall recurrence rate was 19.4%, while malignant phyllodes tumors recurred at a rate of 26% (Spitaleri et al. 2013). As well, MPTB is still related to high mortality and metastasis rates (Kapiris et al. 2001; Asoglu et al. 2004; Fou et al. 2006; Onkendi et al. 2014). Thus, the local control and prevention of metastasis after surgery for MPTB is particularly important.
There has been an increase in the use of RT in recent years. Commission on Cancer of the American College of Surgeons’ National Cancer Databases show that 19.5% of patients receiving RT for phyllodes tumors in 2008–2009 received RT, a substantial increase over the 9.5% rate in 1998–1999 (Gnerlich et al. 2014). Phyllodes tumors, however, remain controversial when it comes to RT. Previous studies have demonstrated the ability of RT to reduce local recurrence after surgery for MPTB (Belkacemi et al. 2008; Barth et al. 2009). Nevertheless, the decrease in local recurrence is not associated with a significant improvement in long-term survival after RT (Neron et al. 2020). In this study, both before and after adjustment, according to our survival analyses, RT did not affect long-term survival for patients with stage T3 or T4 malignant phyllodes tumors (Fig. 1). Before adjustment, no significant difference in OS (p = 0.212) and BCSS (p = 0.1) was found between groups with and without RT. Using of PSM, there was still no significant difference between the two groups in OS (p = 0.172) or BCSS (p = 0.552). It is worth noting that the survival curve (Fig. 1) shows that the rate of long-term survival in the non-RT group is higher than that in the RT group, although there is no difference between the two groups. Univariate and multivariate Cox regression analyses in this study also showed (Table 2 and Table 3) that RT did not affect BCSS (p = 0.1, HR = 1.38, 95% CI = 0.94–2.03 in the univariate Cox regression analyses and p = 0.877, HR = 1.03, 95% CI = 0.68–1.56 in the multivariate Cox regression analyses) and OS (p = 0.373, HR = 1.16, 95% CI = 0.84–1.61 in the univariate Cox regression analyses and p = 0.554, HR = 0.899, 95% CI = 0.634–1.276 in the multivariate Cox regression analyses). In the previous literature, similar results have been confirmed. According to Pandey et al., the addition of RT did not improve 5-year disease-free survival (Pandey et al. 2001). It was concluded by Confavreux et al. that the effects of RT on long-term survival in MPTB patients were insignificant, and even unneeded RT may even have been harmful in some cases (Confavreux et al. 2006). Of course, there is evidence in the literature that MPTB patients with these types such as those who are younger (< 45 years), those with larger tumors, and those with more extensive resections are more likely to benefit from RT (Chao et al. 2019). At least, the outcome of this study of patients with stage T3 or T4 MPTB revealed no significant effect of RT on BCSS and OS, which is consistent with previous findings.
In our study, an analysis of univariate and multivariate data showed that patients with stage T3 or T4 MPTB had poor survival rates when their age ≥ 50 years. Age ≥ 50 years in MPTB may be associated with the risk of metastasis (Neron et al. 2020). Regarding the prognostic impact of surgical approach, the survival rates from BCS and mastectomy were similar in one study (Mitus et al. 2014). Nevertheless, in a study of patients with stage T1-2 MPTB, BCS was associated with better OS and BCS (Chen and Ya 2023). This may be due to the fact that BCS can lead to better cosmetic results, higher quality of life, and other benefits. In our subgroup of patients with stage T3 or T4 MPTB, the surgery of primary site significantly affected OS and BCSS from univariate and multivariate Cox regression analyses. As the nomogram model (Figs. 2 and 3) shows, mastectomy is more favorable for patients’ long-term survival. Since patients with stage T3 or T4 MPTB may have more unfavorable features, mastectomy is the preferred surgical approach. Moreover, few studies have shown that long-term survival for MPTB patients can be enhanced by CT. CT was found to have little effect on survival of patients in a prospective study (Ramakant et al. 2015). It is important to note, however, that the sample size of this literature is small. Furthermore, an earlier study by Broglio K et al. concluded CT had no effect on survival (Morales-Vasquez et al. 2007). In our study, multivariate Cox regression analyses showed no significant effect of CT (p = 0.311, HR = 1.419, 95% CI = 0.721–2.794 in the BCSS and p = 0.424, HR = 1.921, 95% CI = 1.090–3.387 in OS) on long-term survival in patients with stage T3 or T4 MPTB, which is in accordance with previous studies. It is recommended that CT can be considered in extreme cases, such as when the tumor is large or invades structures such as the chest wall (Strode et al. 2017). In conclusion, chemotherapy is not recommended as first-line treatment for phyllodes tumors so far.
In this study, there are several limitations. Firstly, in the SEER database, borderline phyllodes tumors may be incorrectly coded as malignant diseases, affecting the results of the analysis. Secondly, approximately half of the patients had no tumor grade reported. Thirdly, it is also necessary for the SEER database to be continuously improved, as it does not provide data on local recurrence and histopathological, including resection margin status. Therefore, we could not explore the impact of RT in recurrence of MPTB.
Conclusions
Based on the SEER database analysis, the study suggests that the patients with stage T3 or T4 MPTB treated with RT after surgery didn't have significant differences in BCSS or OS compared to those not treated with RT. However, the value of RT in MPTB still needs to be validated based on adequate data from large prospective studies.
Data availability
The data supporting the findings of this study can be found at http://seer.cancer.gov, a database maintained by the Surveillance, Epidemiology, and End Results (SEER) program.
References
Asoglu O, Ugurlu MM, Blanchard K, Grant CS, Reynolds C, Cha SS, Donohue JH (2004) Risk factors for recurrence and death after primary surgical treatment of malignant phyllodes tumors. Ann Surg Oncol 11:1011–1017
Barth RJ Jr, Wells WA, Mitchell SE, Cole BF (2009) A prospective, multi-institutional study of adjuvant radiotherapy after resection of malignant phyllodes tumors. Ann Surg Oncol 16:2288–2294
Belkacemi Y, Bousquet G, Marsiglia H, Ray-Coquard I, Magne N, Malard Y, Lacroix M, Gutierrez C, Senkus E, Christie D, Drumea K, Lagneau E, Kadish SP, Scandolaro L, Azria D, Ozsahin M (2008) Phyllodes tumor of the breast. Int J Radiat Oncol Biol Phys 70:492–500
Chaney AW, Pollack A, McNeese MD, Zagars GK, Pisters PW, Pollock RE, Hunt KK (2000) Primary treatment of cystosarcoma phyllodes of the breast. Cancer 89:1502–1511
Chao X, Chen K, Zeng J, Bi Z, Guo M, Chen Y, Yao Y, Wu W, Liang S, Nie Y (2019) Adjuvant radiotherapy and chemotherapy for patients with breast phyllodes tumors: a systematic review and meta-analysis. BMC Cancer 19:372
Chen CY, Ya C (2023) Better survival was found in patients treated with breast-conserving surgery compared with mastectomy in malignant phyllodes tumor of the breast. Updates Surg. https://doi.org/10.1007/s13304-023-01547-y
Confavreux C, Lurkin A, Mitton N, Blondet R, Saba C, Ranchere D, Sunyach MP, Thiesse P, Biron P, Blay JY, Ray-Coquard I (2006) Sarcomas and malignant phyllodes tumours of the breast–a retrospective study. Eur J Cancer 42:2715–2721
Fou A, Schnabel FR, Hamele-Bena D, Wei XJ, Cheng B, El Tamer M, Klein L, Joseph KA (2006) Long-term outcomes of malignant phyllodes tumors patients: an institutional experience. Am J Surg 192:492–495
Gnerlich JL, Williams RT, Yao K, Jaskowiak N, Kulkarni SA (2014) Utilization of radiotherapy for malignant phyllodes tumors: analysis of the National Cancer Data Base, 1998–2009. Ann Surg Oncol 21:1222–1230
Hawkins RE, Schofield JB, Fisher C, Wiltshaw E, McKinna JA (1992) The clinical and histologic criteria that predict metastases from cystosarcoma phyllodes. Cancer 69:141–147
Kapiris I, Nasiri N, A’Hern R, Healy V, Gui GP (2001) Outcome and predictive factors of local recurrence and distant metastases following primary surgical treatment of high-grade malignant phyllodes tumours of the breast. Eur J Surg Oncol 27:723–730
Lu Y, Chen Y, Zhu L, Cartwright P, Song E, Jacobs L, Chen K (2019) Local recurrence of benign, borderline, and malignant phyllodes tumors of the breast: a systematic review and meta-analysis. Ann Surg Oncol 26:1263–1275
Macdonald OK, Lee CM, Tward JD, Chappel CD, Gaffney DK (2006) Malignant phyllodes tumor of the female breast: association of primary therapy with cause-specific survival from the Surveillance, Epidemiology, and End Results (SEER) program. Cancer 107:2127–2133
Mishra SP, Tiwary SK, Mishra M, Khanna AK (2013) Phyllodes tumor of breast: a review article. ISRN Surg 2013:361469
Mitus J, Reinfuss M, Mitus JW, Jakubowicz J, Blecharz P, Wysocki WM, Skotnicki P (2014) Malignant phyllodes tumor of the breast: treatment and prognosis. Breast J 20:639–644
Morales-Vasquez F, Gonzalez-Angulo AM, Broglio K, Lopez-Basave HN, Gallardo D, Hortobagyi GN, De La Garza JG (2007) Adjuvant chemotherapy with doxorubicin and dacarbazine has no effect in recurrence-free survival of malignant phyllodes tumors of the breast. Breast J 13:551–556
Neron M, Sajous C, Thezenas S, Piperno-Neumann S, Reyal F, Lae M, Chakiba C, Michot A, Penel N, Honore C, Owen C, Bertucci F, Salas S, Saada-Bouzid E, Valentin T, Bompas E, Brahmi M, Ray-Coquard I, Blay JY, Firmin N, Group French Sarcoma (2020) Surgical margins and adjuvant therapies in malignant phyllodes tumors of the breast: a multicenter retrospective study. Ann Surg Oncol 27:1818–1827
Onkendi EO, Jimenez RE, Spears GM, Harmsen WS, Ballman KV, Hieken TJ (2014) Surgical treatment of borderline and malignant phyllodes tumors: the effect of the extent of resection and tumor characteristics on patient outcome. Ann Surg Oncol 21:3304–3309
Pandey M, Mathew A, Kattoor J, Abraham EK, Mathew BS, Rajan B, Nair KM (2001) Malignant phyllodes tumor. Breast J 7:411–416
Parker SJ, Harries SA (2001) Phyllodes tumours. Postgrad Med J 77:428–435
Pezner RD, Schultheiss TE, Paz IB (2008) Malignant phyllodes tumor of the breast: local control rates with surgery alone. Int J Radiat Oncol Biol Phys 71:710–713
Ramakant P, Selvamani P, Therese MM, Paul MJ (2015) Metastatic malignant phyllodes tumor of the breast: an aggressive disease-analysis of 7 cases. Indian J Surg Oncol 6:363–369
Spitaleri G, Toesca A, Botteri E, Bottiglieri L, Rotmensz N, Boselli S, Sangalli C, Catania C, Toffalorio F, Noberasco C, Delmonte A, Luini A, Veronesi P, Colleoni M, Viale G, Zurrida S, Goldhirsch A, Veronesi U, De Pas T (2013) Breast phyllodes tumor: a review of literature and a single center retrospective series analysis. Crit Rev Oncol Hematol 88:427–436
Strode M, Khoury T, Mangieri C, Takabe K (2017) Update on the diagnosis and management of malignant phyllodes tumors of the breast. Breast 33:91–96
Zhang Y, Kleer CG (2016) Phyllodes tumor of the breast: histopathologic features, differential diagnosis, and molecular/genetic updates. Arch Pathol Lab Med 140:665–671
Zhou ZR, Wang CC, Sun XJ, Yang ZZ, Chen XX, Shao ZM, Yu XL, Guo XM (2018) Prognostic factors in breast phyllodes tumors: a nomogram based on a retrospective cohort study of 404 patients. Cancer Med 7:1030–1042
Acknowledgements
The study was approved by the First Affiliated Hospital of Nanchang University. The authors would like to express their appreciation to N. Bataille (Department of Oral and Maxillofacial Surgery) for the SEER database for sharing the worthy data.
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
GZ and JZ designed the study. data curation was done by CW. Analysis of data is done by PY. The original manuscript was written by GZ, PY and CW. Supervision was done by JZ. All authors contributed to data analyses and the drafting and revising of the paper. All authors agreed to be accountable for all aspects of the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zhang, G., Yang, P., Zeng, J. et al. Effect of radiation therapy on patients with stage T3 or T4 malignant phyllodes tumors: a retrospective observational study based on SEER. J Cancer Res Clin Oncol 150, 2 (2024). https://doi.org/10.1007/s00432-023-05517-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00432-023-05517-0