Abstract
Background and Purpose
The use of oncolytic viruses as a gene therapy vector is an area of active biomedical research, particularly in the context of cancer treatment. However, the actual therapeutic success of this approach to tumor elimination remains limited. As such, the present study was developed with the goal of simultaneously enhancing the antitumor efficacy of oncolytic viruses and the local immune response by combining the Ad-GD55 oncolytic adenovirus and an antibody specific for the TIM-3 immune checkpoint molecule (α-TIM-3).
Approach and Key Results
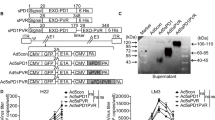
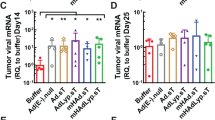
The results of Virus and cell-mediated cytotoxicity assay, qPCR, and Western immunoblotting showed that Ad-GD55–α-Tim-3 oncolytic adenovirus is capable of inducing α-TIM-3 expression within hepatoma cells upon infection, and Ad-GD55–α-TIM-3 exhibited inhibitory efficacy superior to that of Ad-GD55 when used to treat these tumor cells together with the induction of enhanced intracellular immunity. In vivo experiments revealed that Ad-GD55–α-TIM-3 administration was sufficient to inhibit tumor growth and engage in a more robust local immune response within the simulated tumor immune microenvironment.
Conclusion and Implications
These results highlighted the promising therapeutic effects of Ad-GD55–α-TIM-3 oncolytic adenovirus against HCC in vitro and in vivo. As such, this Ad-GD55–α-TIM-3 oncolytic adenovirus may represent a viable approach to the treatment of hepatocellular carcinoma.





Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
References
Bailly C, Thuru X, Goossens L, Goossens JF (2023) Soluble TIM-3 as a biomarker of progression and therapeutic response in cancers and other of human diseases. Biochem Pharmacol 209:115445
Cao W, Li M, Liu J, Zhang S, Noordam L, Verstegen MMA, Wang L, Ma B, Li S, Wang W, Bolkestein M, Doukas M, Chen K, Ma Z, Bruno M, Sprengers D, Kwekkeboom J, van der Laan LJW, Smits R, Peppelenbosch MP, Pan Q (2020) LGR5 marks targetable tumor-initiating cells in mouse liver cancer. Nat Commun 11:1961
Chen K, Ma J, Jia X, Ai W, Ma Z, Pan Q (2019) Advancing the understanding of NAFLD to hepatocellular carcinoma development: From experimental models to humans. Biochim Biophys Acta 1871:117–125
Chen L, Hong J, Hu R, Yu X, Chen X, Zheng S, Qin Y, Zhou X, Wang Y, Zheng L, Fang H, Liu P, Huang B (2021) clinical value of combined detection of serum sTim-3 and pepsinogen for gastric cancer diagnosis. Cancer Manag Res 13:7759–7769
Chen L, Qin Y, Lin B, Yu X, Zheng S, Zhou X, Liu X, Wang Y, Huang B, Jin J, Wang L (2022a) Clinical value of the sTim-3 level in chronic kidney disease. Exp Ther Med 24:606
Chen L, Yu X, Lv C, Dai Y, Wang T, Zheng S, Qin Y, Zhou X, Wang Y, Pei H, Fang H, Huang B (2022b) Increase in serum soluble Tim-3 level is related to the progression of diseases after hepatitis virus infection. Front Med 9:880909
Chesney J, Puzanov I, Collichio F, Singh P, Milhem MM, Glaspy J, Hamid O, Ross M, Friedlander P, Garbe C, Logan TF, Hauschild A, Lebbé C, Chen L, Kim JJ, Gansert J, Andtbacka RHI, Kaufman HL (2018) Randomized, open-label phase II study evaluating the efficacy and safety of talimogene laherparepvec in combination with ipilimumab versus ipilimumab alone in patients with advanced, unresectable melanoma. J Clin Oncol 36:1658–1667
Cox AG, Hwang KL, Brown KK, Evason K, Beltz S, Tsomides A, O’Connor K, Galli GG, Yimlamai D, Chhangawala S, Yuan M, Lien EC, Wucherpfennig J, Nissim S, Minami A, Cohen DE, Camargo FD, Asara JM, Houvras Y, Stainier DYR, Goessling W (2016) Yap reprograms glutamine metabolism to increase nucleotide biosynthesis and enable liver growth. Nat Cell Biol 18:886–896
Czauderna C, Castven D, Mahn FL, Marquardt JU (2019) Context-Dependent Role of NF-κB Signaling in Primary Liver Cancer-from Tumor Development to Therapeutic Implications. Cancers 11:1053
Das R, Langou S, Le TT, Prasad P, Lin F, Nguyen TD (2021) electrical stimulation for immune modulation in cancer treatments. Front Bioeng Biotechnol 9:795300
Huang H, Liu Y, Liao W, Cao Y, Liu Q, Guo Y, Lu Y, Xie Z (2019) Oncolytic adenovirus programmed by synthetic gene circuit for cancer immunotherapy. Nat Commun 10:4801
Hwang JK, Hong J, Yun CO (2020) Oncolytic Viruses and Immune Checkpoint Inhibitors: Preclinical Developments to Clinical Trials. Int J Mol Sci 21:8627
Ishino R, Kawase Y, Kitawaki T, Sugimoto N, Oku M, Uchida S, Imataki O, Matsuoka A, Taoka T, Kawakami K, van Kuppevelt TH, Todo T, Takaori-Kondo A, Kadowaki N (2021) Oncolytic Virus Therapy with HSV-1 for Hematological Malignancies. Mol Ther J Am Soc Gene Ther 29:762–774
Jia X, Liu Y, Cheng Y, Wang Y, Kang H, Ma Z, Chen K (2022) Inosine monophosphate dehydrogenase type1 sustains tumor growth in hepatocellular carcinoma. J Clin Lab Anal 36:e24416
Ju F, Luo Y, Lin C, Jia X, Xu Z, Tian R, Lin Y, Zhao M, Chang Y, Huang X, Li S, Ren W, Qin Y, Yu M, Jia J, Han J, Luo W, Zhang J, Fu G, Ye X, Huang C, Xia N (2022) Oncolytic virus expressing PD-1 inhibitors activates a collaborative intratumoral immune response to control tumor and synergizes with CTLA-4 or TIM-3 blockade. J Immunother Cancer 10:e004762
Kim H, Kim H, Feng Y, Li Y, Tamiya H, Tocci S, Ronai ZA (2020) PRMT5 control of cGAS/STING and NLRC5 pathways defines melanoma response to antitumor immunity. Sci Transl Med 12:eaaz5683
Lang FF, Conrad C, Gomez-Manzano C, Yung WKA, Sawaya R, Weinberg JS, Prabhu SS, Rao G, Fuller GN, Aldape KD, Gumin J, Vence LM, Wistuba I, Rodriguez-Canales J, Villalobos PA, Dirven CMF, Tejada S, Valle RD, Alonso MM, Ewald B, Peterkin JJ, Tufaro F, Fueyo J (2018) Phase I study of DNX-2401 (Delta-24-RGD) oncolytic adenovirus: replication and immunotherapeutic effects in recurrent malignant glioma. J Clin Oncol 36:1419–1427
LaRocca CJ, Warner SG (2018) Oncolytic viruses and checkpoint inhibitors: combination therapy in clinical trials. Clin Transl Med 7:35
Lebbé C, Meyer N, Mortier L, Marquez-Rodas I, Robert C, Rutkowski P, Menzies AM, Eigentler T, Ascierto PA, Smylie M, Schadendorf D, Ajaz M, Svane IM, Gonzalez R, Rollin L, Lord-Bessen J, Saci A, Grigoryeva E, Pigozzo J (2019) Evaluation of two dosing regimens for nivolumab in combination with ipilimumab in patients with advanced melanoma: results from the phase IIIb/IV checkmate 511 trial. J Clin Oncol 37:867–875
Li H, Wu K, Tao K, Chen L, Zheng Q, Lu X, Liu J, Shi L, Liu C, Wang G, Zou W (2012) Tim-3/galectin-9 signaling pathway mediates T-cell dysfunction and predicts poor prognosis in patients with hepatitis B virus-associated hepatocellular carcinoma. Hepatology (baltimore, MD) 56:1342–1351
Li Q, Chen Q, Klauser PC, Li M, Zheng F, Wang N, Li X, Zhang Q, Fu X, Wang Q, Xu Y, Wang L (2020) Developing covalent protein drugs via proximity-enabled reactive therapeutics. Cell 182:85-97.e16
Li Z, Li N, Li F, Zhou Z, Sang J, Chen Y, Han Q, Lv Y, Liu Z (2016) Immune checkpoint proteins PD-1 and TIM-3 are both highly expressed in liver tissues and correlate with their gene polymorphisms in patients with HBV-related hepatocellular carcinoma. Medicine 95:e5749
Lin CZ, Xiang GL, Zhu XH, Xiu LL, Sun JX, Zhang XY (2018) Advances in the mechanisms of action of cancer-targeting oncolytic viruses. Oncol Lett 15:4053–4060
Minott JA, van Vloten JP, Chan L, Mehrani Y, Bridle BW, Karimi K (2022) The role of neutrophils in oncolytic orf virus-mediated cancer immunotherapy. Cells 11:2858
Monney L, Sabatos CA, Gaglia JL, Ryu A, Waldner H, Chernova T, Manning S, Greenfield EA, Coyle AJ, Sobel RA, Freeman GJ, Kuchroo VK (2002) Th1-specific cell surface protein Tim-3 regulates macrophage activation and severity of an autoimmune disease. Nature 415:536–541
Qin S, Dong B, Yi M, Chu Q, Wu K (2020) Prognostic values of TIM-3 expression in patients with solid tumors: a meta-analysis and database evaluation. Front Oncol 10:1288
Raja J, Ludwig JM, Gettinger SN, Schalper KA, Kim HS (2018) Oncolytic virus immunotherapy: future prospects for oncology. J Immunother Cancer 6:140
Ribas A, Dummer R, Puzanov I, VanderWalde A, Andtbacka RHI, Michielin O, Olszanski AJ, Malvehy J, Cebon J, Fernandez E, Kirkwood JM, Gajewski TF, Chen L, Gorski KS, Anderson AA, Diede SJ, Lassman ME, Gansert J, Hodi FS, Long GV (2017) Oncolytic virotherapy promotes intratumoral T cell infiltration and improves anti-PD-1 immunotherapy. Cell 170:1109-1119.e1110
Sabatos-Peyton CA, Nevin J, Brock A, Venable JD, Tan DJ, Kassam N, Xu F, Taraszka J, Wesemann L, Pertel T, Acharya N, Klapholz M, Etminan Y, Jiang X, Huang YH, Blumberg RS, Kuchroo VK, Anderson AC (2018) Blockade of Tim-3 binding to phosphatidylserine and CEACAM1 is a shared feature of anti-Tim-3 antibodies that have functional efficacy. Oncoimmunology 7:e1385690
Samson A, West EJ, Carmichael J, Scott KJ, Turnbull S, Kuszlewicz B, Dave RV, Peckham-Cooper A, Tidswell E, Kingston J, Johnpulle M, da Silva B, Jennings VA, Bendjama K, Stojkowitz N, Lusky M, Prasad KR, Toogood GJ, Auer R, Bell J, Twelves CJ, Harrington KJ, Vile RG, Pandha H, Errington-Mais F, Ralph C, Newton DJ, Anthoney A, Melcher AA, Collinson F (2022) Neoadjuvant intravenous oncolytic vaccinia virus therapy promotes anticancer immunity in patients. Cancer Immunol Res 10:745–756
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F (2021) Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA a Cancer J Clin 71:209–249
Tang W, Chen Z, Zhang W, Cheng Y, Zhang B, Wu F, Wang Q, Wang S, Rong D, Reiter FP, De Toni EN, Wang X (2020) The mechanisms of sorafenib resistance in hepatocellular carcinoma: theoretical basis and therapeutic aspects. Signal Transduct Target Ther 5:87
Tang Y, Li K, Hu B, Cai Z, Li J, Tao H, Cao J (2022) Fatty acid binding protein 5 promotes the proliferation, migration, and invasion of hepatocellular carcinoma cells by degradation of Krüppel-like factor 9 mediated by miR-889-5p via cAMP-response element binding protein. Cancer Biol Ther 23:424–438
Thongsri P, Pewkliang Y, Borwornpinyo S, Wongkajornsilp A, Hongeng S, Sa-Ngiamsuntorn K (2021) Curcumin inhibited hepatitis B viral entry through NTCP binding. Sci Rep 11:19125
Tseng HC, Xiong W, Badeti S, Yang Y, Ma M, Liu T, Ramos CA, Dotti G, Fritzky L, Jiang JG, Yi Q, Guarrera J, Zong WX, Liu C, Liu D (2020) Efficacy of anti-CD147 chimeric antigen receptors targeting hepatocellular carcinoma. Nat Commun 11:4810
Villanueva A (2019) Hepatocellular carcinoma. N Engl J Med 380:1450–1462
Wang G, Kang X, Chen KS, Jehng T, Jones L, Chen J, Huang XF, Chen SY (2020) An engineered oncolytic virus expressing PD-L1 inhibitors activates tumor neoantigen-specific T cell responses. Nat Commun 11:1395
Xiao Y, Qing J, Li B, Chen L, Nong S, Yang W, Tang X, Chen Z (2020) TIM-3 participates in the invasion and metastasis of nasopharyngeal carcinoma via SMAD7/SMAD2/SNAIL1 axis-mediated epithelial-mesenchymal transition. Onco Targets Ther 13:1993–2006
Yang ZY, Liu L, Mao C, Wu XY, Huang YF, Hu XF, Tang JL (2014) Chemotherapy with cetuximab versus chemotherapy alone for chemotherapy-naive advanced non-small cell lung cancer. Cochrane Database Syst Rev, p Cd009948
Yi M, Jiao D, Xu H, Liu Q, Zhao W, Han X, Wu K (2018) Biomarkers for predicting efficacy of PD-1/PD-L1 inhibitors. Mol Cancer 17:129
Ying C, Xiao BD, Qin Y, Wang BR, Liu XY, Wang RW, Fang L, Yan H, Zhou XM, Wang YG (2018) GOLPH2-regulated oncolytic adenovirus, GD55, exerts strong killing effect on human prostate cancer stem-like cells in vitro and in vivo. Acta Pharmacol Sin 39:405–414
Yoshida H, Sato-Dahlman M, Hajeri P, Jacobsen K, Koodie L, Yanagiba C, Shanley R, Yamamoto M (2021) Mutant myogenin promoter-controlled oncolytic adenovirus selectively kills PAX3-FOXO1-positive rhabdomyosarcoma cells. Translational Oncology 14:100997
Zhang ZL, Zou WG, Luo CX, Li BH, Wang JH, Sun LY, Qian QJ, Liu XY (2003) An armed oncolytic adenovirus system, ZD55-gene, demonstrating potent antitumoral efficacy. Cell Res 13:481–489
Zhu C, Sakuishi K, Xiao S, Sun Z, Zaghouani S, Gu G, Wang C, Tan DJ, Wu C, Rangachari M, Pertel T, Jin HT, Ahmed R, Anderson AC, Kuchroo VK (2015) An IL-27/NFIL3 signalling axis drives Tim-3 and IL-10 expression and T-cell dysfunction. Nat Commun 6:6072
Funding
The research leading to these results received funding from the Applied Research and Cultivation Program of Jiangxi Province, No. 20212BAG70043; the Public Welfare Technology Project of Zhejiang Province, No. LGF21H160033; Zhejiang Medical Technology Plan Project, No. 2021KY047; Hangzhou Science and Technology Bureau, No. 20201203B44; Hangzhou Medical Health Science and Technology Project, No. B20220173; the National Natural Science Foundation of China, No. 81803069 and the Grant for 521 talent project of ZSTU.
Author information
Authors and Affiliations
Contributions
The writing of the article was mainly completed by LQ and WYG., the experimental part was mainly completed by LQ., and the technical support was provided by ZLL, ZHL, WF, WH, WYQ and WXY. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest or non-financial interest in the subject matter or materials discussed in this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Qiang, L., Huili, Z., Leilei, Z. et al. Intratumoral delivery of a Tim-3 antibody-encoding oncolytic adenovirus engages an effective antitumor immune response in liver cancer. J Cancer Res Clin Oncol 149, 18201–18213 (2023). https://doi.org/10.1007/s00432-023-05501-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-05501-8