Abstract
Purpose
Our study intended to explore the correlation between HER2 alterations and 18F-FDG PET/CT metabolic parameters and their prognostic value in EGFR-negative non-small-cell lung cancer (NSCLC) patients detected by next-generation sequencing (NGS).
Methods
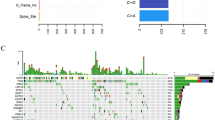
NGS assay was performed in 1737 NSCLC patients, a total of 88 HER2 alterations and 176 negative HER2 with EGFR-negative patients were randomly selected for this study.
Results
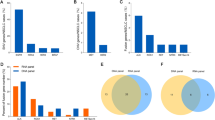
When the HER2 status with EGFR-negative group was analyzed, multivariate analysis showed that smoking status, primary tumor SUVmax (pSUVmax) < 13.03 and stage were the independent deterministic factors of HER2 alterations. Multivariate cox regression analysis revealed that HER2 status, age, smoking status and stage were independent risk factors for overall survival (OS) in EGFR-negative NSCLC patients with different HER2 status. When the HER2 alterations group was separately analyzed, multivariate analysis demonstrated that low pSUVmax < 15.32 and histology were the independent deterministic factors of HER2 mutation. Multivariate cox regression analysis revealed that pSUVmax, smoking status, nodal involvement and treatment methods were independent risk factors for OS in EGFR-negative NSCLC patients with HER2 alterations.
Conclusion
The study revealed that low pSUVmax was associated with HER2 alterations in EGFR-negative NSCLC patients, moreover HER2 mutation and HER2 amplification exhibited distinct 18F-FDG metabolic and clinical characteristics. Furthermore, it explored the prognostic value of HER2 alterations and 18F-FDG PET/CT metabolic parameters of pSUVmax in EGFR-negative NSCLC patients.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author Yuetao Wang, upon reasonable request.
References
Arcila ME, Chaft JE, Nafa K et al (2012) Prevalence, clinicopathologic associations, and molecular spectrum of ERBB2 (HER2) tyrosine kinase mutations in lung adenocarcinomas. Clin Cancer Res 18:4910–4918
Berghmans T, Dusart M, Paesmans M et al (2008) Primary tumor standardized uptake value (SUVmax) measured on fluorodeoxyglucose positron emission tomography (FDG-PET) is of prognostic value for survival in non-small cell lung cancer (NSCLC): a systematic review and meta-analysis (MA) by the European Lung Cancer Working Party for the IASLC Lung Cancer Staging Project. J Thorac Oncol 3:6–12
Borst GR, Belderbos JS, Boellaard R et al (2005) Standardised FDG uptake: a prognostic factor for inoperable non-small cell lung cancer. Eur J Cancer 41:1533–1541
Chen R, Zhou X, Liu J, Huang G (2016) Relationship between 18F-FDG PET/CT findings and HER2 expression in gastric cancer. J Nucl Med 57:1040–1044
Chen D, Xu J, Qiao R et al (2020) Detection of genetic mutations by next-generation sequencing for predicting prognosis of extensive-stage small-cell lung cancer. J Oncol 2020:8811487
Eng J, Hsu M, Chaft JE, Kris MG, Arcila ME, Li BT (2016) Outcomes of chemotherapies and HER2 directed therapies in advanced HER2-mutant lung cancers. Lung Cancer 99:53–56
Ferlay J, Colombet M, Soerjomataram I et al (2019) Estimating the global cancer incidence and mortality in 2018: GLOBOCAN sources and methods. Int J Cancer 144:1941–1953
Higashi K, Ueda Y, Yagishita M et al (2000) FDG PET measurement of the proliferative potential of non-small cell lung cancer. J Nucl Med 41:85–92
Jadvar H, Alavi A, Gambhir SS (2009) 18F-FDG uptake in lung, breast, and colon cancers: molecular biology correlates and disease characterization. J Nucl Med 50:1820–1827
Jo I, Zeon SK, Kim SH et al (2015) Correlation of primary tumor FDG uptake with clinicopathologic prognostic factors in invasive ductal carcinoma of the breast. Nucl Med Mol Imaging 49:19–25
Kalemkerian GP, Narula N, Kennedy EB et al (2018) Molecular testing guideline for the selection of patients with lung cancer for treatment with targeted tyrosine kinase inhibitors: American Society of Clinical Oncology Endorsement of the College of American Pathologists/International Association for the Study of Lung Cancer/Association for Molecular Pathology clinical practice guideline update. J Clin Oncol 36:911–919
Lee K, Jung HA, Sun JM et al (2020) Clinical characteristics and outcomes of non-small cell lung cancer patients with HER2 alterations in Korea. Cancer Res Treat 52:292–300
Li C, Sun Y, Fang R et al (2012) Lung adenocarcinomas with HER2-activating mutations are associated with distinct clinical features and HER2/EGFR copy number gains. J Thorac Oncol 7:85–89
Li BT, Ross DS, Aisner DL et al (2016) HER2 amplification and HER2 mutation are distinct molecular targets in lung cancers. J Thorac Oncol 11:414–419
Li Y, Wu X, Fang J, Zhao Q, Huang Y, Jiang L (2022) Evaluation of fluorine-18-fluorodeoxyglucose PET/computed tomography and human epithelial growth factor receptor 2 expression in treatment-naive patients with lung adenocarcinoma. Nucl Med Commun 43:442–450
Lv Z, Fan J, Xu J et al (2018) Value of (18)F-FDG PET/CT for predicting EGFR mutations and positive ALK expression in patients with non-small cell lung cancer: a retrospective analysis of 849 Chinese patients. Eur J Nucl Med Mol Imaging 45:735–750
Mazières J, Peters S, Lepage B et al (2013) Lung cancer that harbors an HER2 mutation: epidemiologic characteristics and therapeutic perspectives. J Clin Oncol 31:1997–2003
Meng X, Sun X, Mu D et al (2012) Noninvasive evaluation of microscopic tumor extensions using standardized uptake value and metabolic tumor volume in non-small-cell lung cancer. Int J Radiat Oncol Biol Phys 82:960–966
Na II, Byun BH, Kim KM et al (2010) 18F-FDG uptake and EGFR mutations in patients with non-small cell lung cancer: a single-institution retrospective analysis. Lung Cancer 67:76–80
Patil T, Mushtaq R, Marsh S et al (2020) Clinicopathologic characteristics, treatment outcomes, and acquired resistance patterns of atypical EGFR mutations and HER2 alterations in stage IV non-small-cell lung cancer. Clin Lung Cancer 21:e191–e204
Peters S, Zimmermann S (2014) Targeted therapy in NSCLC driven by HER2 insertions. Transl Lung Cancer Res 3:84–88
Putora PM, Szentesi K, Glatzer M et al (2016) SUVmax and tumour location in PET-CT predict oncogene status in lung cancer. Oncol Res Treat 39:681–686
Riudavets M, Sullivan I, Abdayem P, Planchard D (2021) Targeting HER2 in non-small-cell lung cancer (NSCLC): a glimpse of hope? An updated review on therapeutic strategies in NSCLC harbouring HER2 alterations. ESMO Open 6:100260
Shen H, Du G, Liu Z et al (2015) Assessment and prognostic analysis of EGFR mutations or/and HER2 overexpression in Uygur’s non-small cell lung cancer. Int J Clin Exp Med 8:22300–22309
Shigematsu H, Takahashi T, Nomura M et al (2005) Somatic mutations of the HER2 kinase domain in lung adenocarcinomas. Cancer Res 65:1642–1646
Song Z, Yu X, Shi Z, Zhao J, Zhang Y (2016) HER2 mutations in Chinese patients with non-small cell lung cancer. Oncotarget 7:78152–78158
Stephens P, Hunter C, Bignell G et al (2004) Lung cancer: intragenic ERBB2 kinase mutations in tumours. Nature 431:525–526
Suzuki M, Shiraishi K, Yoshida A et al (2015) HER2 gene mutations in non-small cell lung carcinomas: concurrence with Her2 gene amplification and Her2 protein expression and phosphorylation. Lung Cancer 87:14–22
Tomizawa K, Suda K, Onozato R et al (2011) Prognostic and predictive implications of HER2/ERBB2/neu gene mutations in lung cancers. Lung Cancer 74:139–144
Wei XW, Gao X, Zhang XC et al (2020) Mutational landscape and characteristics of ERBB2 in non-small cell lung cancer. Thorac Cancer 11:1512–1521
Wu R, Yuan B, Li C, Wang Z, Song Y, Liu H (2021) A narrative review of advances in treatment and survival prognosis of HER2-positive malignant lung cancers. J Thorac Dis 13:3708–3720
Xu F, Yang G, Xu H, Yang L, Qiu W, Wang Y (2020) Treatment outcome and clinical characteristics of HER2 mutated advanced non-small cell lung cancer patients in China. Thorac Cancer 11:679–685
Yoshizawa A, Sumiyoshi S, Sonobe M et al (2014) HER2 status in lung adenocarcinoma: a comparison of immunohistochemistry, fluorescence in situ hybridization (FISH), dual-ISH, and gene mutations. Lung Cancer 85:373–378
Yu HA, Planchard D, Lovly CM (2018) Sequencing therapy for genetically defined subgroups of non-small cell lung cancer. Am Soc Clin Oncol Educ Book 38:726–739
Funding
This work was supported by the National Natural Science Foundation of China (81871381, 82202219 and 82272044), Key Laboratory of Changzhou High-tech Research Project (CM20193010), Shanghai Sailing Program (20YF1444500, 21YF1442700), the basic research of Shanghai Chest Hospital (2020YNJCM07, 2020YNJCQ11, 2021YNJCM09 and 2020YNJCQ06), the Nurture projects for the special project of integrated traditional Chinese and Western medicine in general hospital of Shanghai Health Committee (ZHYY-ZXYJHZX-202023), the Youth Medical Talents-Medical Imaging Practitioners Program (SHWRS(2020)_087), the Natural Science Foundation of Shanghai (21ZR1458900).
Author information
Authors and Affiliations
Contributions
Each author participated sufficiently in the work and approved the final version. YW, MR and WX had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. CC, and JS contributed to the study design and data analysis and interpretation. LL, LW and BL contributed to data analysis and interpretation. HY and HZ contributed to writing and revising of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
No potential conflicts of interest were disclosed.
Ethics approval
All procedures performed in studies involving human participants were approved by the Institutional Review Board of Shanghai Jiao Tong University School of Medicine-affiliated Shanghai Chest Hospital (No.IS22048). This study was performed in accordance with the ethical standards as laid down in the 1964 Declaration of Helsinki and its later amendments.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ruan, M., Chang, C., Sun, J. et al. Exploring the correlation between HER2 alterations and 18F-FDG PET/CT metabolic parameters and their prognostic value in EGFR-negative non-small-cell lung cancer patients. J Cancer Res Clin Oncol 149, 14493–14507 (2023). https://doi.org/10.1007/s00432-023-05218-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-05218-8