Abstract
Purpose
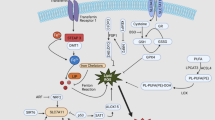
Iron acquisition is key to maintaining cell survival and function. Cancer cells in general are considered to have an insatiable iron need. Iron delivery via the transferrin/transferrin receptor pathway has been the canonical iron uptake mechanism. Recently, however, our laboratory and others have explored the ability of ferritin, particularly the H-subunit, to deliver iron to a variety of cell types. Here, we investigate whether Glioblastoma (GBM) initiating cells (GICs), a small population of stem-like cells, are known for their iron addiction and invasive nature acquire exogenous ferritin, as a source of iron. We further assess the functional impact of ferritin uptake on the invasion capacity of the GICs.
Methods
To establish that H-ferritin can bind to human GBM, tissue-binding assays were performed on samples collected at the time of surgery. To interrogate the functional consequences of H-ferritin uptake, we utilized two patient-derived GIC lines. We further describe H-ferritin’s impact on GIC invasion capacity using a 3D invasion assay.
Results
H-ferritin bound to human GBM tissue at the amount of binding was influenced by sex. GIC lines showed uptake of H-ferritin protein via transferrin receptor. FTH1 uptake correlated with a significant decrease in the invasion capacity of the cells. H-ferritin uptake was associated with a significant decrease in the invasion-related protein Rap1A.
Conclusion
These findings indicate that extracellular H-ferritin participates in iron acquisition to GBMs and patient-derived GICs. The functional significance of the increased iron delivery by H-ferritin is a decreased invasion capacity of GICs potentially via reduction of Rap1A protein levels.






Similar content being viewed by others
Data availability
All data and methodology supporting the findings of this study are available within the paper and its Supplementary Information files.
References
Aisen P, Enns C, Wessling-Resnick M (2001) Chemistry and biology of eukaryotic iron metabolism. Int J Biochem Cell Biol 33(10):940–959. https://doi.org/10.1016/S1357-2725(01)00063-2
Alkhateeb AA, Han B, Connor JR (2013) Ferritin stimulates breast cancer cells through an iron-independent mechanism and is localized within tumor-associated macrophages. Breast Cancer Res Treat 137(3):733–744. https://doi.org/10.1007/S10549-012-2405-X
Bailey CL, Kelly P, Casey PJ (2009) Activation of Rap1 promotes prostate cancer metastasis. Can Res 69(12):4962–4968. https://doi.org/10.1158/0008-5472.CAN-08-4269/654961/P/ACTIVATION-OF-RAP1-PROMOTES-PROSTATE-CANCER
Beunk L, Bakker GJ, van Ens D, Bugter J, Gal F, Svoren M, Friedl P, Wolf K (2022) Actomyosin contractility requirements and reciprocal cell–tissue mechanics for cancer cell invasion through collagen-based channels. Eur Phys J E Soft Matter 45(5):48. https://doi.org/10.1140/EPJE/S10189-022-00182-6
Boylan KLM, Manion RD, Shah H, Skubitz KM, Skubitz APN (2020) Inhibition of ovarian cancer cell spheroid formation by synthetic peptides derived from nectin-4. Int J Mol Sci 21(13):1–16. https://doi.org/10.3390/IJMS21134637
Brown RAM, Richardson KL, Kabir TD, Trinder D, Ganss R, Leedman PJ (2020) Altered iron metabolism and impact in cancer biology, metastasis, and immunology. Front Oncol 10:476. https://doi.org/10.3389/FONC.2020.00476
Chang CC, Wu M, Yuan F (2014) Role of specific endocytic pathways in electrotransfection of cells. Mol Ther Methods Clin Dev 1:14058. https://doi.org/10.1038/mtm.2014.58
Chen Y, Fan Z, Yang Y, Gu C (2019) Iron metabolism and its contribution to cancer (Review). Int J Oncol 54(4):1143–1154. https://doi.org/10.3892/IJO.2019.4720/HTML
Cheng L, Wu Q, Guryanova OA, Huang Z, Huang Q, Rich JN, Bao S (2011) Elevated invasive potential of glioblastoma stem cells. Biochem Biophys Res Commun 406(4):643. https://doi.org/10.1016/J.BBRC.2011.02.123
Chiou B, Connor JR (2018) Emerging and dynamic biomedical uses of ferritin. Pharmaceuticals. https://doi.org/10.3390/PH11040124
Chiou B, Lucassen E, Sather M, Kallianpur A, Connor J (2018) Semaphorin4A and H-ferritin utilize Tim-1 on human oligodendrocytes: a novel neuro-immune axis. Glia 66(7):1317–1330. https://doi.org/10.1002/GLIA.23313
Di Sanzo M, Cozzolino F, Battaglia AM, Aversa I, Monaco V, Sacco A, Biamonte F, Palmieri C, Procopio F, Santamaria G, Ortuso F, Pucci P, Monti M, Faniello MC (2022) Ferritin heavy chain binds peroxiredoxin 6 and inhibits cell proliferation and migration. Int J Mol Sci 23(21):12987. https://doi.org/10.3390/IJMS232112987/S1
Duck KA, Connor JR (2016) Iron uptake and transport across physiological barriers. Biometals 29(4):573. https://doi.org/10.1007/S10534-016-9952-2
Eyler CE, Rich JN (2008) Survival of the fittest: cancer stem cells in therapeutic resistance and angiogenesis. J Clin Oncol 26(17):2839. https://doi.org/10.1200/JCO.2007.15.1829
Fisher J, Devraj K, Ingram J, Slagle-Webb B, Madhankumar AB, Liu X, Klinger M, Simpson IA, Connor JR (2007a) Ferritin: a novel mechanism for delivery of iron to the brain and other organs. Am J Physiol Cell Physiol. https://doi.org/10.1152/AJPCELL.00599.2006
Fisher J, Devraj K, Ingram J, Slagle-Webb B, Madhankumar AB, Liu X, Klinger M, Simpson IA, Connor JR (2007b) Ferritin: a novel mechanism for delivery of iron to the brain and other organs. Am J Phys Cell Physiol 293(2):641–649. https://doi.org/10.1152/AJPCELL.00599.2006/ASSET/IMAGES/LARGE/ZH00080753030006.JPEG
Friedmann-Morvinski D (2014) Glioblastoma heterogeneity and cancer cell plasticity. Crit Rev Oncog 19(5):327–336. https://doi.org/10.1615/CRITREVONCOG.2014011777
Fukuyama T, Ogita H, Kawakatsu T, Fukuhara T, Yamada T, Sato T, Shimizu K, Nakamura T, Matsuda M, Takai Y (2005) Involvement of the c-Src-Crk-C3G-Rap1 signaling in the nectin-induced activation of Cdc42 and formation of adherens junctions. J Biol Chem 280(1):815–825. https://doi.org/10.1074/JBC.M411099200
Gerlee P, Nelander S (2012) The Impact of Phenotypic Switching on Glioblastoma Growth and Invasion. PLoS Comput Biol 8(6):1002556. https://doi.org/10.1371/JOURNAL.PCBI.1002556
Glick RP, Gettleman R, Patel K, Lakshman R, Tsibris JCM (1989) Insulin and insulin-like growth factor I in brain tumors: binding and in vitro effects. Neurosurgery 24(6):791–797. https://doi.org/10.1227/00006123-198906000-00001
Goffart N, Kroonen J, Rogister B (2013) Glioblastoma-initiating cells: relationship with neural stem cells and the micro-environment. Cancers 5(3):1049. https://doi.org/10.3390/CANCERS5031049
Grochans S, Cybulska AM, Simińska D, Korbecki J, Kojder K, Chlubek D, Baranowska-Bosiacka I (2022) Epidemiology of glioblastoma multiforme-literature review. Cancers. https://doi.org/10.3390/CANCERS14102412
Han J, Seaman WE, Di X, Wang W, Willingham M, Torti FM, Torti SV (2011) Iron uptake mediated by binding of H-ferritin to the TIM-2 receptor in mouse cells. PLoS ONE 6(8):e23800. https://doi.org/10.1371/JOURNAL.PONE.0023800
Hatoum A, Mohammed R, Zakieh O (2019) The unique invasiveness of glioblastoma and possible drug targets on extracellular matrix. Cancer Manag Res 11:1843. https://doi.org/10.2147/CMAR.S186142
Hatzikirou H, Basanta D, Simon M, Schaller K, Deutsch A (2012) ‘Go or Grow’: the key to the emergence of invasion in tumour progression? Math Med Bio J IMA 29(1):49–65. https://doi.org/10.1093/IMAMMB/DQQ011
Hu W, Zhou C, Jing Q, Li Y, Yang J, Yang C, Wang L, Hu J, Li H, Wang H, Yuan C, Zhou Y, Ren X, Tong X, Du J, Wang Y (2021) FTH promotes the proliferation and renders the HCC cells specifically resist to ferroptosis by maintaining iron homeostasis. Cancer Cell Int 21(1):1–18. https://doi.org/10.1186/S12935-021-02420-X/FIGURES/7
Inoue A, Takahashi H, Harada H, Kohno S, Ohue S, Kobayashi K, Yano H, Tanaka J, Ohnishi T (2010) Cancer stem-like cells of glioblastoma characteristically express MMP-13 and display highly invasive activity. Int J Oncol 37(5):1121–1131. https://doi.org/10.3892/IJO_00000764
Jiang H, Muir RK, Gonciarz RL, Olshen AB, Yeh I, Hann BC, Zhao N, Wang YH, Behr SC, Korkola JE, Evans MJ, Collisson EA, Renslo AR (2022) Ferrous iron–activatable drug conjugate achieves potent MAPK blockade in KRAS-driven tumors. J Exp Med. https://doi.org/10.1084/JEM.20210739/213060
Koneru T, McCord E, Pawar S, Tatiparti K, Sau S, Iyer AK (2021) Transferrin: biology and use in receptor-targeted nanotherapy of gliomas. ACS Omega 6(13):8727–8733. https://doi.org/10.1021/ACSOMEGA.0C05848/ASSET/IMAGES/LARGE/AO0C05848_0004.JPEG
Konijn AM, Glickstein H, Vaisman B, Meyron-Holtz EG, Slotki IN, Cabantchik ZI (1999) The cellular labile iron pool and intracellular ferritin in K562 cells. Blood 94(6):2128–2134. https://doi.org/10.1182/BLOOD.V94.6.2128
Lamb R, Ozsvari B, Lisanti CL, Tanowitz HB, Howell A, Martinez-Outschoorn UE, Sotgia F, Lisanti MP (2015) Antibiotics that target mitochondria effectively eradicate cancer stem cells, across multiple tumor types: Treating cancer like an infectious disease. Oncotarget 6(7):4569. https://doi.org/10.18632/ONCOTARGET.3174
Landry AP, Balas M, Alli S, Spears J, Zador Z (2020) Distinct regional ontogeny and activation of tumor associated macrophages in human glioblastoma. Sci Rep 10(1):1–13. https://doi.org/10.1038/s41598-020-76657-3
Lathia JD, Mack SC, Mulkearns-Hubert EE, Valentim CLL, Rich JN (2015) Cancer stem cells in glioblastoma. Genes Dev 29(12):1203. https://doi.org/10.1101/GAD.261982.115
Lattmann E, Deng T, Hajnal A (2021) To divide or invade: a look behind the scenes of the proliferation-invasion interplay in the Caenorhabditis elegans anchor cell. Front Cell Dev Biol 8:1727. https://doi.org/10.3389/FCELL.2020.616051/BIBTEX
Li L, Fang CJ, Ryan JC, Niemi EC, Lebrón JA, Björkman PJ, Arase H, Torti FM, Torti SV, Nakamura MC, Seaman WE (2010) Binding and uptake of H-ferritin are mediated by human transferrin receptor-1. Proc Natl Acad Sci USA 107(8):3505–3510. https://doi.org/10.1073/PNAS.0913192107/SUPPL_FILE/PNAS.200913192SI.PDF
Lobello N, Biamonte F, Pisanu ME, Faniello MC, Jakopin Ž, Chiarella E, Giovannone ED, Mancini R, Ciliberto G, Cuda G, Costanzo F (2016) Ferritin heavy chain is a negative regulator of ovarian cancer stem cell expansion and epithelial to mesenchymal transition. Oncotarget 7(38):62019. https://doi.org/10.18632/ONCOTARGET.11495
Looi CK, Hii LW, Ngai SC, Leong CO, Mai CW (2020) The role of ras-associated protein 1 (Rap1) in cancer: bad actor or good player? Biomedicines 8(9): 334. https://doi.org/10.3390/BIOMEDICINES8090334
Meyron-Holtz EG, Moshe-Belizowski S, Cohen LA (2011) A possible role for secreted ferritin in tissue iron distribution. J Neural Transmiss (Vienna, Austria: 1996) 118(3):337–347. https://doi.org/10.1007/s00702-011-0582-0
Palsa K, Baringer SL, Shenoy G, Simpson IA, Connor JR (2023) Exosomes are involved in iron transport from human blood-brain barrier endothelial cells and are modified by endothelial cell iron status. J Biol Chem 299(2):102868. https://doi.org/10.1016/j.jbc.2022.102868
Pearson JRD, Cuzzubbo S, McArthur S, Durrant LG, Adhikaree J, Tinsley CJ, Pockley AG, McArdle SEB (2020) Immune escape in glioblastoma multiforme and the adaptation of immunotherapies for treatment. Front Immunol 11:1. https://doi.org/10.3389/FIMMU.2020.582106
Prager BC, Xie Q, Bao S, Rich JN (2019) Cancer stem cells: the architects of the tumor ecosystem. Cell Stem Cell 24(1):41. https://doi.org/10.1016/J.STEM.2018.12.009
Prionisti I, Bühler LH, Walker PR, Jolivet RB (2019) Harnessing microglia and macrophages for the treatment of glioblastoma. Front Pharmacol. https://doi.org/10.3389/FPHAR.2019.00506
Rivera M, Sukhdeo K, Yu J (2013) Ionizing radiation in glioblastoma initiating cells. Front Oncol. https://doi.org/10.3389/FONC.2013.00074/BIBTEX
Sammarco MC, Ditch S, Banerjee A, Grabczyk E (2008) Ferritin L and H subunits are differentially regulated on a post-transcriptional level. J Biol Chem 283(8):4578–4587. https://doi.org/10.1074/JBC.M703456200
Sanchez M, Galy B, Dandekar T, Bengert P, Vainshtein Y, Stolte J, Muckenthaler MU, Hentze MW (2006) Iron regulation and the cell cycle: Identification of an iron-responsive element in the 3′-untranslated region of human cell division cycle 14A mRNA by a refined microarray-based screening strategy. J Biol Chem 281(32):22865–22874. https://doi.org/10.1074/JBC.M603876200
Schonberg DL, Miller TE, Wu Q, Flavahan WA, Das NK, Hale JS, Hubert CG, Mack SC, Jarrar AM, Karl RT, Rosager AM, Nixon AM, Tesar PJ, Hamerlik P, Kristensen BW, Horbinski C, Connor JR, Fox PL, Lathia JD, Rich JN (2015) Preferential iron trafficking characterizes glioblastoma stem-like cells. Cancer Cell 28(4):441–455. https://doi.org/10.1016/j.ccell.2015.09.002
Sesé B, Íñiguez-Muñoz S, Ensenyat-Mendez M, Llinàs-Arias P, Ramis G, Orozco JIJ, de Mattos SF, Villalonga P, Marzese DM (2022) Glioblastoma embryonic-like stem cells exhibit immune-evasive phenotype. Cancers 14(9):2070. https://doi.org/10.3390/CANCERS14092070/S1
Sodek KL, Ringuette MJ, Brown TJ (2009) Compact spheroid formation by ovarian cancer cells is associated with contractile behavior and an invasive phenotype. Int J Cancer 124(9):2060–2070. https://doi.org/10.1002/IJC.24188
Sun T, Wu H, Li Y, Huang Y, Yao L, Chen X, Han X, Zhou Y, Du Z (2017) Targeting transferrin receptor delivery of temozolomide for a potential glioma stem cell-mediated therapy. Oncotarget 8(43):74451. https://doi.org/10.18632/ONCOTARGET.20165
Todorich B, Zhang X, Slagle-Webb B, Seaman WE, Connor JR (2008) Tim-2 is the receptor for H-ferritin on oligodendrocytes. J Neurochem 107(6):1495–1505. https://doi.org/10.1111/J.1471-4159.2008.05678.X
Todorich B, Zhang X, Connor JR (2011) H-ferritin is the major source of iron for oligodendrocytes. Glia 59(6):927–935. https://doi.org/10.1002/GLIA.21164
Treps L, Perret R, Edmond S, Ricard D, Gavard J (2017) Glioblastoma stem-like cells secrete the pro-angiogenic VEGF-A factor in extracellular vesicles. J Extracell Vesic. https://doi.org/10.1080/20013078.2017.1359479
Truman-Rosentsvit M, Berenbaum D, Spektor L, Cohen LA, Belizowsky-Moshe S, Lifshitz L, Ma J, Li W, Kesselman E, Abutbul-Ionita I, Danino D, Gutierrez L, Li H, Li K, Lou H, Regoni M, Poli M, Glaser F, Rouault TA, Meyron-Holtz EG (2018) Ferritin is secreted via 2 distinct nonclassical vesicular pathways. Blood 131(3):342–352. https://doi.org/10.1182/BLOOD-2017-02-768580
Volovetz J, Berezovsky AD, Alban T, Chen Y, Lauko A, Aranjuez GF, Burtscher A, Shibuya K, Silver DJ, Peterson J, Manor D, McDonald JA, Lathia JD (2020) Identifying conserved molecular targets required for cell migration of glioblastoma cancer stem cells. Cell Death Dis. https://doi.org/10.1038/S41419-020-2342-2
Wilkinson N, Pantopoulos K (2014) The IRP/IRE system in vivo: insights from mouse models. Front Pharmacol. https://doi.org/10.3389/FPHAR.2014.00176
You GR, Chang JT, Li YL, Chen YJ, Huang YC, Fan KH, Chen YC, Kang CJ, Cheng AJ (2021) Molecular interplays between cell invasion and radioresistance that lead to poor prognosis in head-neck cancer. Front Oncol 11:2724. https://doi.org/10.3389/FONC.2021.681717/BIBTEX
Zhang YL, Wang RC, Cheng K, Ring BZ, Su L (2017) Roles of Rap1 signaling in tumor cell migration and invasion. Cancer Biol Med 14(1):90. https://doi.org/10.20892/J.ISSN.2095-3941.2016.0086
Acknowledgements
We would like to sincerely thank Gonzalo Hernandez Viedma and Dr. Mayka Sánchez Fernández of UIC Barcelona International University of Catalonia for their help in analyzing the mRNA sequences for IRE. We also thank Nate Sheaffer, Joseph Bednarczyk, and Dr. Thomas Abraham from Penn State College of Medicine’s Flow Cytometry and Microscopy cores for their assistance and guidance with flow cytometry and microscopy analysis. The Advanced Light Microscopy core (RRID: SCR_022526) services and instruments used in this project were funded, in part, by the Pennsylvania State University College of Medicine via the Office of the Vice Dean of Research and Graduate Students. We also thank Penn State Laboratory for Isotopes and Metals in the Environment (LIME) for performing ICP-AES on our protein samples. The GBM tissue was obtained from the Penn State Neurosurgery BioRepository.
Funding
This work was funded by National Institutes of Health under Grant P01CA245705. The contents of this paper are solely the responsibility of the authors and do not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
Author contributions are as follows. BPS: conceptualization, data curation, formal analysis, investigation, methodology, writing—original draft, and writing—review and editing. BWS: methodology, and human subjects’ data collection and analysis. GS: data curation, formal analysis, and software. VS: data curation, formal analysis, and software. BEZ: human patient tissue resection and collection and patient data collection. JRC: conceptualization, funding acquisition, supervision, and writing—review and editing. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
Human GBM tissues used in the study were collected following an institutional review board (IRB)-compliant protocol. (Penn State Hershey Neuroscience Institute Biorepository IRB #2914, approved). Informed consents were obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
(Pandya) Shesh, B., Slagle-Webb, B., Shenoy, G. et al. Uptake of H-ferritin by Glioblastoma stem cells and its impact on their invasion capacity. J Cancer Res Clin Oncol 149, 9691–9703 (2023). https://doi.org/10.1007/s00432-023-04864-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-04864-2