Abstract
Purpose
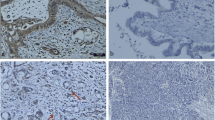
This study aimed to demonstrate the involvement of angiogenesis in cancer-associated acinar-to-ductal metaplasia (CA-ADM) lesion of invasive front pancreatic ductal adenocarcinoma (PDAC) and investigate the possible mechanism.
Methods
Tissue samples from 128 patients with PDAC and 36 LSL-KrasG12D/+; LSL-Trp53R172H/+; Pdx-1-Cre mice were analyzed. Immunohistochemical assay was performed using HE, anti-CK19 and anti-amylase to confirm the presence of CA-ADM lesions, using anti-CD34 and anti-CD31 to measure microvessel density (MVD), and using anti-CD68, anti-CD163, anti-iNOS, or anti-MMP9 to evaluate the immune microenvironment. We performed multiplex immunohistochemical assay to detect the co-expression of MMP9 and CD68 on macrophage. We examined clinical outcomes and other clinicopathological factors to determine the significance of high-level MVD of CA-ADM on survival and liver metastasis. We performed tube formation assay to evaluate the effect of macrophage on angiogenic capacity in vitro.
Results
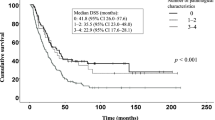
Angiogenesis was significantly abundant in CA-ADM lesions compared with that in PDAC lesions in human and mouse tissues. High-level MVD in CA-ADM lesions was an independent predictor of poor prognosis (P = 0.0047) and the recurrence of liver metastasis (P = 0.0027). More CD68-positive and CD163-positive macrophages were detected in CA-ADM lesions than in PDAC. The percentage of CD68-positive macrophages was positively correlated with MVD in CA-ADM lesions. Multiplex-immunostaining revealed that MMP9 was expressed in CD68-positive macrophages of CA-ADM lesions. In CA-ADM lesions, the percentage of macrophages was positively correlated with MMP9 expression, which positively correlated with microvessel density.
Conclusion
CA-ADM related angiogenesis is a promising predictive marker for poor prognosis of PDAC and may provide an attractive therapeutic target for PDAC.





Similar content being viewed by others
Data availability statement
The data presented in this research are available from the corresponding author upon request.
References
Al-Abd AM, Alamoudi AJ, Abdel-Naim AB, Neamatallah TA, Ashour OM (2017) Anti-angiogenic agents for the treatment of solid tumors: potential pathways, therapy and current strategies—a review. J Adv Res 8:591–605. https://doi.org/10.1016/j.jare.2017.06.006
Ansari D, Tingstedt B, Andersson B, Holmquist F, Sturesson C, Williamsson C, Sasor A, Borg D, Bauden M, Andersson R (2016) Pancreatic cancer: yesterday, today and tomorrow. Future Oncol 12:1929–1946. https://doi.org/10.2217/fon-2016-0010
Ardito CM, Grüner BM, Takeuchi KK, Lubeseder-Martellato C, Teichmann N, Mazur PK, DelGiorno KE, Carpenter ES, Halbrook CJ, Hall JC (2012) EGF receptor is required for KRAS-induced pancreatic tumorigenesis. Cancer Cell 22:304–317. https://doi.org/10.1016/j.ccr.2012.07.024
Augoff K, Hryniewicz-Jankowska A, Tabola R, Stach K (2022) MMP9: a tough target for targeted therapy for cancer. Cancers 14:1847. https://doi.org/10.3390/cancers14071847
Brune K, Abe T, Canto M, O’Malley L, Klein AP, Maitra A, Adsay NV, Fishman EK, Cameron JL, Yeo CJ (2006) Multifocal neoplastic precursor lesions associated with lobular atrophy of the pancreas in patients having a strong family history of pancreatic cancer. Am J Surg Pathol 30:1067
Chari ST, Kelly K, Hollingsworth MA, Thayer SP, Ahlquist DA, Andersen DK, Batra SK, Brentnall TA, Canto M, Cleeter DF (2015) Early detection of sporadic pancreatic cancer: summative review. Pancreas 44:693. https://doi.org/10.1097/MPA.0000000000000368
Cheng K, Liu C-F, Rao G-W (2021) Anti-angiogenic agents: a review on vascular endothelial growth factor receptor-2 (VEGFR-2) inhibitors. Curr Med Chem 28:2540–2564. https://doi.org/10.2174/0929867327666200514082425
Ellis L, Takahashi Y, Fenoglio C, Cleary K, Bucana C, Evans D (1998) Vessel counts and vascular endothelial growth factor expression in pancreatic adenocarcinoma. Eur J Cancer 34:337–340. https://doi.org/10.1016/s0959-8049(97)10068-5
El-Rouby DH (2010) Association of macrophages with angiogenesis in oral verrucous and squamous cell carcinomas. J Oral Pathol Med 39:559–564. https://doi.org/10.1111/j.1600-0714.2010.00879.x
Feng H, Moriyama T, Ohuchida K, Sheng N, Iwamoto C, Shindo K, Shirahane K, Ikenaga N, Nagai S, Nakata K (2021) N-acetyl cysteine induces quiescent-like pancreatic stellate cells from an active state and attenuates cancer-stroma interactions. J Exp Clin Cancer Res 40:1–19. https://doi.org/10.1186/s13046-021-01939-1
Folkman J (1971) Tumor angiogenesis: therapeutic implications. N Engl J Med 285:1182–1186. https://doi.org/10.1056/NEJM197111182852108
Fujimoto K, Hosotani R, Wada M, Lee J-U, Koshiba T, Miyamoto Y, Tsuji S, Nakajima S, Doi R, Imamura M (1998) Expression of two angiogenic factors, vascular endothelial growth factor and platelet-derived endothelial cell growth factor in human pancreatic cancer, and its relationship to angiogenesis. Eur J Cancer 34:1439–1447. https://doi.org/10.1016/s0959-8049(98)00069-0
Fukuda A, Wang SC, Morris JP IV, Folias AE, Liou A, Kim GE, Akira S, Boucher KM, Firpo MA, Mulvihill SJ (2011) Stat3 and MMP7 contribute to pancreatic ductal adenocarcinoma initiation and progression. Cancer Cell 19:441–455. https://doi.org/10.1016/j.ccr.2011.03.002
Genin M, Clement F, Fattaccioli A, Raes M, Michiels C (2015) M1 and M2 macrophages derived from THP-1 cells differentially modulate the response of cancer cells to etoposide. BMC Cancer 15:1–14. https://doi.org/10.1186/s12885-015-1546-9
Gheorghe G, Bungau S, Ilie M, Behl T, Vesa CM, Brisc C, Bacalbasa N, Turi V, Costache RS, Diaconu CC (2020) Early diagnosis of pancreatic cancer: the key for survival. Diagnostics 10:869. https://doi.org/10.3390/diagnostics10110869
Glass G, Papin JA, Mandell JW (2009) SIMPLE: a sequential immunoperoxidase labeling and erasing method. J Histochem Cytochem 57:899–905. https://doi.org/10.1369/jhc.2009.953612
Grippo PJ, Sandgren EP (2012) Acinar-to-ductal metaplasia accompanies c-myc-induced exocrine pancreatic cancer progression in transgenic rodents. Int J Cancer 131:1243–1248. https://doi.org/10.1002/ijc.27322
Guerra C, Schuhmacher AJ, Cañamero M, Grippo PJ, Verdaguer L, Pérez-Gallego L, Dubus P, Sandgren EP, Barbacid M (2007) Chronic pancreatitis is essential for induction of pancreatic ductal adenocarcinoma by K-Ras oncogenes in adult mice. Cancer Cell 11:291–302. https://doi.org/10.1016/j.ccr.2007.01.012
Hanahan D, Weinberg RA (2011) Hallmarks of cancer: the next generation. Cell 144:646–674. https://doi.org/10.1016/j.cell.2011.02.013
He H, Mack JJ, Güç E, Warren CM, Squadrito ML, Kilarski WW, Baer C, Freshman RD, McDonald AI, Ziyad S (2016) Perivascular macrophages limit permeability. Arterioscler Thromb Vasc Biol 36:2203–2212. https://doi.org/10.1161/ATVBAHA.116.307592
Hingorani SR, Wang L, Multani AS, Combs C, Deramaudt TB, Hruban RH, Rustgi AK, Chang S, Tuveson DA (2005) Trp53R172H and KrasG12D cooperate to promote chromosomal instability and widely metastatic pancreatic ductal adenocarcinoma in mice. Cancer Cell 7:469–483. https://doi.org/10.1016/j.ccr.2005.04.023
Huang H (2018) Matrix metalloproteinase-9 (MMP-9) as a cancer biomarker and MMP-9 biosensors: recent advances. Sensors 18:3249. https://doi.org/10.3390/s18103249
Huang X, Bhugul PA, Fan G, Ye T, Huang S, Dai S, Chen B, Zhou M (2018) Luteolin inhibits pancreatitis-induced acinar-ductal metaplasia, proliferation and epithelial-mesenchymal transition of acinar cells. Mol Med Rep 17:3681–3689. https://doi.org/10.3892/mmr.2017.8327
Illemann M, Bird N, Majeed A, Sehested M, Laerum OD, Lund LR, Danø K, Nielsen BS (2006) MMP-9 is differentially expressed in primary human colorectal adenocarcinomas and their metastases. Mol Cancer Res 4:293–302. https://doi.org/10.1158/1541-7786.MCR-06-0003
Jiemy WF, van Sleen Y, van der Geest KS, Ten Berge HA, Abdulahad WH, Sandovici M, Boots AM, Heeringa P, Brouwer E (2020) Distinct macrophage phenotypes skewed by local granulocyte macrophage colony-stimulating factor (GM-CSF) and macrophage colony-stimulating factor (M-CSF) are associated with tissue destruction and intimal hyperplasia in giant cell arteritis. Clin Transl Immunol 9:e1164. https://doi.org/10.1002/cti2.1164
Kibe S, Ohuchida K, Ando Y, Takesue S, Nakayama H, Abe T, Endo S, Koikawa K, Okumura T, Iwamoto C (2019) Cancer-associated acinar-to-ductal metaplasia within the invasive front of pancreatic cancer contributes to local invasion. Cancer Lett 444:70–81. https://doi.org/10.1016/j.canlet.2018.12.005
Koikawa K, Ohuchida K, Takesue S, Ando Y, Kibe S, Nakayama H, Endo S, Abe T, Okumura T, Horioka K (2018) Pancreatic stellate cells reorganize matrix components and lead pancreatic cancer invasion via the function of Endo180. Cancer Lett 412:143–154. https://doi.org/10.1016/j.canlet.2017.10.010
Kopp JL, von Figura G, Mayes E, Liu F-F, Dubois CL, Morris JP IV, Pan FC, Akiyama H, Wright CV, Jensen K (2012) Identification of Sox9-dependent acinar-to-ductal reprogramming as the principal mechanism for initiation of pancreatic ductal adenocarcinoma. Cancer Cell 22:737–750. https://doi.org/10.1016/j.ccr.2012.10.025
Li L, Fan P, Chou H, Li J, Wang K, Li H (2019a) Herbacetin suppressed MMP9 mediated angiogenesis of malignant melanoma through blocking EGFR-ERK/AKT signaling pathway. Biochimie 162:198–207. https://doi.org/10.1016/j.biochi.2019.05.003
Li S, Xu H-X, Wu C-T, Wang W-Q, Jin W, Gao H-L, Li H, Zhang S-R, Xu J-Z, Qi Z-H (2019b) Angiogenesis in pancreatic cancer: current research status and clinical implications. Angiogenesis 22:15–36. https://doi.org/10.1007/s10456-018-9645-2
Liou G-Y, Döppler H, Necela B, Krishna M, Crawford HC, Raimondo M, Storz P (2013) Macrophage-secreted cytokines drive pancreatic acinar-to-ductal metaplasia through NF-κB and MMPs. J Cell Biol 202:563–577. https://doi.org/10.1083/jcb.201301001
Mantovani A, Marchesi F, Malesci A, Laghi L, Allavena P (2017) Tumour-associated macrophages as treatment targets in oncology. Nat Rev Clin Oncol 14:399–416. https://doi.org/10.1038/nrclinonc.2016.217
Mitrofanova I, Zavyalova M, Riabov V, Cherdyntseva N, Kzhyshkowska J (2018) The effect of neoadjuvant chemotherapy on the correlation of tumor-associated macrophages with CD31 and LYVE-1. Immunobiology 223:449–459. https://doi.org/10.1016/j.imbio.2017.10.050
Mondal S, Adhikari N, Banerjee S, Amin SKA, Jha Tarun (2020) Matrix metalloproteinase-9 (MMP-9) and its inhibitors in cancer: A minireview. Eur J Med Chem 194112260-S0223523420302270 112260. https://doi.org/10.1016/j.ejmech.2020.112260
Nucera S, Biziato D, De Palma M (2011) The interplay between macrophages and angiogenesis in development, tissue injury and regeneration. Int J Dev Biol 55:495–503. https://doi.org/10.1387/ijdb.103227sn
Pelekanou V, Villarroel-Espindola F, Schalper KA, Pusztai L, Rimm DL (2018) CD68, CD163, and matrix metalloproteinase 9 (MMP-9) co-localization in breast tumor microenvironment predicts survival differently in ER-positive and-negative cancers. Breast Cancer Res 20:1–10. https://doi.org/10.1186/s13058-018-1076-x
Raggi F, Pelassa S, Pierobon D, Penco F, Gattorno M, Novelli F, Eva A, Varesio L, Giovarelli M, Bosco MC (2017) Regulation of human macrophage M1–M2 polarization balance by hypoxia and the triggering receptor expressed on myeloid cells-1. Front Immunol 8:1097. https://doi.org/10.3389/fimmu.2017.01097
Rawla P, Sunkara T, Gaduputi V (2019) Epidemiology of pancreatic cancer: global trends, etiology and risk factors. J Oncol 10:10. https://doi.org/10.14740/wjon1166
Ribatti D (2008) Judah Folkman, a pioneer in the study of angiogenesis. Angiogenesis 11:3–10. https://doi.org/10.1007/s10456-008-9092-6
Ryan DP, Hong TS, Bardeesy N (2014) Pancreatic adenocarcinoma. N Engl J Med 371:1039–1049. https://doi.org/10.1056/NEJMra1404198
Siegel RL, Miller KD, Fuchs HE, Jemal A (2022) Cancer statistics, 2022. CA Cancer J Clin. https://doi.org/10.3322/caac.21708
Stefanowski J, Lang A, Rauch A, Aulich L, Köhler M, Fiedler AF, Buttgereit F, Schmidt-Bleek K, Duda GN, Gaber T (2019) Spatial distribution of macrophages during callus formation and maturation reveals close crosstalk between macrophages and newly forming vessels. Front Immunol. https://doi.org/10.3389/fimmu.2019.02588
Wang L, Xie D, Wei D (2019) Pancreatic acinar-to-ductal metaplasia and pancreatic cancer. Pancreatic cancer. Springer, New York, pp 299–308
Wynn TA, Chawla A, Pollard JW (2013) Macrophage biology in development, homeostasis and disease. Nature 496:445–455. https://doi.org/10.1038/nature12034
Xiang H, Yu H, Zhou Q, Wu Y, Ren J, Zhao Z, Tao X, Dong D (2022) Macrophages: a rising star in immunotherapy for chronic pancreatitis. Pharmacol Res. https://doi.org/10.1016/j.phrs.2022.106508
Yan Z, Ohuchida K, Fei S, Zheng B, Guan W, Feng H, Kibe S, Ando Y, Koikawa K, Abe T (2019a) Inhibition of ERK1/2 in cancer-associated pancreatic stellate cells suppresses cancer—stromal interaction and metastasis. J Exp Clin Cancer Res 38:1–16. https://doi.org/10.1186/s13046-019-1226-8
Yan Z, Ohuchida K, Zheng B, Okumura T, Takesue S, Nakayama H, Iwamoto C, Shindo K, Moriyama T, Nakata K (2019b) CD110 promotes pancreatic cancer progression and its expression is correlated with poor prognosis. J Cancer Res Clin Oncol 145:1147–1164. https://doi.org/10.1007/s00432-019-02860-z
Zhang Z, Ji S, Zhang B, Liu J, Qin Y, Xu J, Yu X (2018) Role of angiogenesis in pancreatic cancer biology and therapy. Biomed Pharmacother 108:1135–1140. https://doi.org/10.1016/j.biopha.2018.09.136
Zhao Y-L, Tian P-X, Han F, Zheng J, Xia X-X, Xue W-J, Ding X-M, Ding C-G (2017) Comparison of the characteristics of macrophages derived from murine spleen, peritoneal cavity, and bone marrow. J Zhejiang Univ Sci B 18:1055–1063. https://doi.org/10.1631/jzus.B1700003
Acknowledgements
We appreciate the technical support provided by Emiko Manabe and Shoko Sadatomi (Department of Surgery and Oncology, Kyushu University). Shuang Fei is the recipient of Otsuka Toshimi (2021-2022) [http://www.otsukafoundation.org/index.html] and Rotary Yoneyama Memorial Foundation scholarship (2022-2023) [http://www.rotary-yoneyama.or.jp]. We thank Gabrielle White Wolf, PhD, from Edanz (https://jp.edanz.com/ac) for editing a draft of this manuscript.
Funding
This work was supported by Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Numbers JP19K22663, JP19K22664, JP22H02922, JP22K15560, JP22K16490.
Author information
Authors and Affiliations
Contributions
Shuang Fei: conceptualization, methodology, formal analysis, investigation, project administration, writing review and editing. Kenoki Ohuchida: conceptualization, investigation, methodology, project administration, writing review and editing. Shin Kibe and Zilong Yan: conceptualization and investigation. Chika Iwamoto and Shinkawa Tomohiko: methodology. Bo Zhang: data curation. Jun Kawata: resources and investigation. Toshiya Abe and Noboru Ideno: investigation. Naoki Ikenaga: formal analysis. Kohei Nakata: methodology. Yoshinao Oda: conceptualization and resources. Masafumi Nakamura: project administration, supervision writing review and editing. The first draft of the manuscript was written by Shuang Fei and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
Approval of the research protocol by an Institutional Reviewer Board: The study was approved by the Ethics Committee of Kyushu University (22002-00) and performed in accordance with the Japanese Government’s Ethical Guidelines for Human Genome/Gene Research and the Helsinki Declaration.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Fei, S., Ohuchida, K., Kibe, S. et al. Involvement of angiogenesis in cancer-associated acinar-to-ductal metaplasia lesion of pancreatic cancer invasive front. J Cancer Res Clin Oncol 149, 5885–5899 (2023). https://doi.org/10.1007/s00432-022-04554-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04554-5