Abstract
Purpose
Human epidermal growth factor 2 (HER2) alterations are found in approximately 2%–5% of non-small cell lung cancer (NSCLC). This study aimed to evaluate the clinical characteristics of patients with NSCLC having HER2 alterations in China and the differences compared with Western counterparts and also perform a prognostic analysis.
Material and Methods
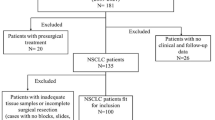
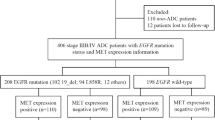
A total of 1300 patients diagnosed with NSCLC from January 2017 to December 2020 were included. Their clinical characteristics were retrospectively recorded. The gene expression profiles and clinical information of 20 patients having altered HER2 were downloaded from the Cancer Genome Atlas database and compared, and the prognostic factors affecting the Chinese population were analyzed. If tissues were sufficient, the overexpression was assessed by immunohistochemical staining.
Results
Among 39 (3.0%) patients with HER2 alterations, 31 patients (79.5%) had HER2 mutations. HER2 insertion mutation in exon 20 was the most common type (A775_G776 ins YVMA). Seven patients (17.9%) had amplification, and one had both. The HER2 kinase domain was most commonly mutated. A majority of patients in the study were young-aged with no smoking history; 66.7% had stage III/IV adenocarcinoma. Compared with Chinese patients, HER2 alterations in Western counterparts were mostly associated with old age, previous smoking, and stages I and II at diagnosis. The most common type of HER2 alteration was HER2 amplification; one patient had coexistence of HER2 gene amplification and fusion. The furin-like cysteine-rich region was most commonly mutated. The median overall survival (OS) of the Chinese patients was 41 months. The univariate analysis showed that age > 60 years, no surgical treatment, no liver or renal cysts on imaging, and maximum tumor diameter ≥ 4.25 cm were significantly associated with poor OS. The multivariate analysis showed that age, presence of surgery, and no hepatic or renal cysts were independent prognostic factors for OS. Chemotherapy achieved better outcomes, and HER2 mutations were not associated with HER2 amplification and overexpression.
Conclusions
This study was novel in comprehensively investigating the clinical and molecular characteristics of patients in Chinese and Western populations, and in analyzing the factors affecting the prognosis of Chinese patients. It provided critical data for future therapies against HER2-altered NSCLC.




Similar content being viewed by others
Data availability
All data used in our study can be found online: https://dataview.ncbi.nlm.nih.gov/object/PRJNA818860?reviewer=eqe2cmevim73qpfkjm0msh0q5l.
References
Arcila ME, Chaft JE, Nafa K et al (2012) Prevalence, clinicopathologic associations, and molecular spectrum of ERBB2 (HER2) tyrosine kinase mutations in lung adenocarcinomas. Clin Cancer Res 18:4910–4918. https://doi.org/10.1158/1078-0432.CCR-12-0912
Auliac JB, Do P, Bayle S et al (2019a) Non-small cell lung cancer patients harboring HER2 mutations: clinical characteristics and management in a real-life setting. Cohort HER2 EXPLORE GFPC 02–14. Adv Ther 36:2161–2166. https://doi.org/10.1007/s12325-019-01001-9
Auliac JB, Dô P, Bayle S et al (2019b) Non-small cell lung cancer patients harboring HER2 mutations: clinical characteristics and management in a real-life setting. Cohort HER2 EXPLORE GFPC 02–14. Adv Ther 36:2161–2166. https://doi.org/10.1007/s12325-019-01001-9
Bu S, Wang R, Pan Y et al (2017) Clinicopathologic characteristics of patients with HER2 insertions in non-small cell lung cancer. Ann Surg Oncol 24:291–297. https://doi.org/10.1245/s10434-015-5044-8
De Greve J, Moran T, Graas MP et al (2015) Phase II study of afatinib, an irreversible ErbB family blocker, in demographically and genotypically defined lung adenocarcinoma. Lung Cancer 88:63–69. https://doi.org/10.1016/j.lungcan.2015.01.013
Dziadziuszko R, Smit EF, Dafni U et al (2019) Afatinib in NSCLC with HER2 mutations: results of the prospective, open-label phase II NICHE trial of European Thoracic Oncology Platform (ETOP). J Thorac Oncol 14:1086–1094. https://doi.org/10.1016/j.jtho.2019.02.017
Fan Y, Chen J, Zhou C et al (2020) Afatinib in patients with advanced non-small cell lung cancer harboring HER2 mutations, previously treated with chemotherapy: a phase II trial. Lung Cancer 147:209–213. https://doi.org/10.1016/j.lungcan.2020.07.017
Gao J, Aksoy BA, Dogrusoz U et al (2013) Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal 6:pl1. https://doi.org/10.1126/scisignal.2004088
Gao Q, Liang WW, Foltz SM et al (2018) Driver fusions and their implications in the development and treatment of human cancers. Cell Rep 23:227-238.e223. https://doi.org/10.1016/j.celrep.2018.03.050
Gao S, Li N, Wang S et al (2020) Lung cancer in People’s Republic of China. J Thorac Oncol 15:1567–1576. https://doi.org/10.1016/j.jtho.2020.04.028
Gatzemeier U, Groth G, Butts C et al (2004) Randomized phase II trial of gemcitabine-cisplatin with or without trastuzumab in HER2-positive non-small-cell lung cancer. Ann Oncol 15:19–27. https://doi.org/10.1093/annonc/mdh031
Hotta K, Aoe K, Kozuki T et al (2018) A phase II study of Trastuzumab Emtansine in HER2-positive non-small cell lung cancer. J Thorac Oncol 13:273–279. https://doi.org/10.1016/j.jtho.2017.10.032
Hyman DM, Piha-Paul SA, Won H et al (2018) HER kinase inhibition in patients with HER2- and HER3-mutant cancers. Nature 554:189–194. https://doi.org/10.1038/nature25475
Langer CJ, Stephenson P, Thor A et al (2004) Trastuzumab in the treatment of advanced non-small-cell lung cancer: is there a role? Focus on Eastern Cooperative Oncology Group study 2598. J Clin Oncol 22:1180–1187. https://doi.org/10.1200/JCO.2004.04.105
Lee K, Jung HA, Sun JM et al (2020) Clinical characteristics and outcomes of non-small cell lung cancer patients with HER2 alterations in Korea. Cancer Res Treat 52:292–300. https://doi.org/10.4143/crt.2019.186
Li BT, Ross DS, Aisner DL et al (2016a) HER2 amplification and HER2 mutation are distinct molecular targets in lung cancers. J Thorac Oncol 11:414–419. https://doi.org/10.1016/j.jtho.2015.10.025
Li BT, Zheng T, Ni A et al (2016b) Identifying HER2 mutation, amplification, and HER2 protein overexpression as therapeutic targets in lung cancers. J Clin Oncol. https://doi.org/10.1200/JCO.2016.34.15_supll.e20666
Li BT, Shen R, Buonocore D et al (2018) Ado-trastuzumab emtansine for patients with HER2-Mutant lung cancers: results from a phase II basket trial. J Clin Oncol 36:2532–2537. https://doi.org/10.1200/jco.2018.77.9777
Li M, Liu Y, Jiang X et al (2021) Inhibition of miR-144-3p exacerbates non-small cell lung cancer progression by targeting CEP55. Acta Biochim Biophys Sin. https://doi.org/10.1093/abbs/gmab118
Ninomiya K, Hata T, Yoshioka H et al (2019) A prospective cohort study to define the clinical features and outcome of lung cancers harboring HER2 aberration in Japan (HER2-CS STUDY). Chest 156:357–366. https://doi.org/10.1016/j.chest.2019.01.011
Oh DY, Bang YJ (2020) HER2-targeted therapies—a role beyond breast cancer. Nat Rev Clin Oncol 17:33–48. https://doi.org/10.1038/s41571-019-0268-3
Pahuja KB, Nguyen TT, Jaiswal BS et al (2018) Actionable activating oncogenic ERBB2/HER2 transmembrane and juxtamembrane domain mutations. Cancer Cell 34:792-806.e795. https://doi.org/10.1016/j.ccell.2018.09.010
Peters S, Zimmermann S (2014) Targeted therapy in NSCLC driven by HER2 insertions. Transl Lung Cancer Res 3:84–88. https://doi.org/10.3978/j.issn.2218-6751.2014.02.06
Xu F, Yang G, Xu H et al (2020) Treatment outcome and clinical characteristics of HER2 mutated advanced non-small cell lung cancer patients in China. Thorac Cancer 11:679–685. https://doi.org/10.1111/1759-7714.13317
Yagishita S, Fujita Y, Kitazono S et al (2015) Chemotherapy-regulated microRNA-125-HER2 pathway as a novel therapeutic target for trastuzumab-mediated cellular cytotoxicity in small cell lung cancer. Mol Cancer Ther 14:1414–1423. https://doi.org/10.1158/1535-7163.Mct-14-0625
Yu HA, Planchard D, Lovly CM (2018) Sequencing therapy for genetically defined subgroups of non-small cell lung cancer. Am Soc Clin Oncol Educ Book Am Soc Clin Oncol Ann Meet 38:726–739. https://doi.org/10.1200/edbk_201331
Zhou C, Li X, Wang Q et al (2020a) Pyrotinib in HER2-mutant advanced lung adenocarcinoma after platinum-based chemotherapy: a multicenter, open-label, single-arm, phase II study. J Clin Oncol 38:2753–2761. https://doi.org/10.1200/jco.20.00297
Zhou J, Ding N, Xu X et al (2020b) Clinical outcomes of patients with HER2-mutant advanced lung cancer: chemotherapies versus HER2-directed therapies. Ther Adv Med Oncol 12:1758835920936090. https://doi.org/10.1177/1758835920936090
Funding
This study was supported in part by the National Natural Science Foundation of China (No. 81802284), the Taishan Scholar Foundation of Shandong Province (No. tsqn202103179), 2021 Shandong Medical Association Clinical Research Fund (No. YXH2022ZX02176). the Science and Technology Development Plans of Shandong Province (No. 2014GSF118157), and the Scientific Research Foundation of Shandong Province of Outstanding Young Scientists (No. BS2013YY058).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. LL, JL and JW made a contribution to data collection and analysis. XZ and HG analyzed and interpreted data and edited the manuscript. GF made substantial contributions to the study design and revision of the manuscript. All authors critically reviewed the manuscript, and all approved the final version submitted for publication.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
This study was performed in line with the principles of the Declaration of Helsinki. Approved was granted by the ethics committee of the Shandong Provincial Affiliated First Medical University (Approval number NSFC: No. 2020-5001). Signed consent forms were obtained before participants were enrolled.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhuo, X., Guo, H., Ma, J. et al. Clinical characteristics and prognostic factors of patients with non-small cell lung cancer having HER2 alterations. J Cancer Res Clin Oncol 149, 2029–2039 (2023). https://doi.org/10.1007/s00432-022-04196-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04196-7