Abstract
Background
Mesenchymal epithelial transition (MET) overexpression has been reported in approximately 50–60% of epidermal growth factor receptor (EGFR)-mutant non-small-cell lung cancers. However, the prognostic significance of MET overexpression has not been established in advanced lung adenocarcinoma (ADC) patients with EGFR-sensitive mutations.
Methods
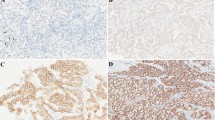
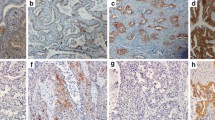
A retrospective study was performed on a total of 406 treatment-naïve advanced ADC patients with EGFR mutation detection and MET expression information. EGFR mutations were detected by next‐generation sequencing or amplification refractory mutation system-polymerase chain reaction. Immuno-histochemistry staining of MET expression was evaluated by H-score and overexpression was defined as an H-score ≥ 200. Overall survival (OS) and progression-free survival (PFS) were analyzed according to MET expression.
Results
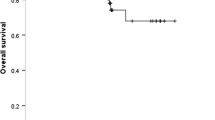
Among the 406 patients, 208 patients had EGFR mutations, including 102 exon 19_del mutations, 94 L858R mutations and 12 other types of mutations. Of 110 patients with concomitant EGFR mutations and MET overexpression, 61 (59.8%) patients had 19_del mutations, 44 (46.8%) patients had L858R mutations and five (41.7%) patients had others. Patients with MET overexpression had a markedly shorter PFS and OS than patients with MET H-score < 200 in the EGFR L858R mutation subgroup (median PFS: 12 versus 26 months, p = 0.001; median OS: 24 versus 32 months, p = 0.038), whereas no significant difference was observed in 19_del mutation subgroup. Multivariate Cox analysis showed that MET overexpression was an independent poor prognostic factor for PFS and OS in patients with the L858R mutations (HR = 3.064, 95% CI 1.705–5.507, p < 0.001; HR = 2.043, 95% CI 1.000–4.172; p = 0.049), rather than 19_del.
Conclusions
MET overexpression is a poor prognostic factor for advanced ADC patients with the EGFR L858R mutation.




Similar content being viewed by others
Data availability
Please contact author for data requests.
References
Bauml J, Cho BC, Park K, Lee KH, Cho EK, Kim D-W et al (2021) Amivantamab in combination with lazertinib for the treatment of osimertinib-relapsed, chemotherapy-naïve EGFR mutant (EGFRm) non-small cell lung cancer (NSCLC) and potential biomarkers for response. J Clin Oncol 39(15):9006. https://doi.org/10.1200/JCO.2021.39.15_suppl.9006
Guo R, Berry LD, Aisner DL, Sheren J, Boyle T, Bunn PA Jr et al (2019) MET IHC is a poor screen for MET amplification or MET Exon 14 mutations in lung adenocarcinomas: data from a tri-institutional cohort of the lung cancer mutation consortium. J Thorac Oncol 14(9):1666–1671. https://doi.org/10.1016/j.jtho.2019.06.009
Hirsch FR, Scagliotti GV, Mulshine JL, Kwon R, Curran WJ Jr, Wu YL et al (2017) Lung cancer: current therapies and new targeted treatments. Lancet 389(10066):299–311. https://doi.org/10.1016/S0140-6736(16)30958-8
Hong S, Gao F, Fu S, Wang Y, Fang W, Huang Y et al (2018) Concomitant genetic alterations with response to treatment and epidermal growth factor receptor tyrosine Kinase inhibitors in patients with EGFR-mutant advanced non-small cell lung cancer. JAMA Oncol 4(5):739–742. https://doi.org/10.1001/jamaoncol.2018.0049
Huang L, An SJ, Chen ZH, Su J, Yan HH, Wu YL (2014) MET expression plays differing roles in non-small-cell lung cancer patients with or without EGFR mutation. J Thorac Oncol 9(5):725–728. https://doi.org/10.1097/JTO.0000000000000105
Kumar A, Petri ET, Halmos B, Boggon TJ (2008) Structure and clinical relevance of the epidermal growth factor receptor in human cancer. J Clin Oncol 26(10):1742–1751. https://doi.org/10.1200/JCO.2007.12.1178
Li A, Niu FY, Han JF, Lou NN, Yang JJ, Zhang XC et al (2015) Predictive and prognostic value of de novo MET expression in patients with advanced non-small-cell lung cancer. Lung Cancer 90(3):375–380. https://doi.org/10.1016/j.lungcan.2015.10.021
Liang H, Li C, Zhao Y, Zhao S, Huang J, Cai X et al (2020) Concomitant mutations in EGFR 19Del/L858R mutation and their association with response to EGFR-TKIs in NSCLC patients. Cancer Manag Res 12:8653–8662. https://doi.org/10.2147/CMAR.S255967
Mignard X, Ruppert AM, Antoine M, Vasseur J, Girard N, Mazieres J et al (2018) c-MET overexpression as a poor predictor of MET amplifications or exon 14 mutations in lung sarcomatoid carcinomas. J Thorac Oncol 13(12):1962–1967. https://doi.org/10.1016/j.jtho.2018.08.008
Paik PK, Felip E, Veillon R, Sakai H, Cortot AB, Garassino MC et al (2020) Tepotinib in non-small-cell lung cancer with MET exon 14 skipping mutations. N Engl J Med 383(10):931–943. https://doi.org/10.1056/NEJMoa2004407
Park K, Haura EB, Leighl NB, Mitchell P, Shu CA, Girard N et al (2021) Amivantamab in EGFR Exon 20 insertion-mutated non-small-cell lung cancer progressing on platinum chemotherapy: initial results from the CHRYSALIS phase I study. J Clin Oncol 39(30):3391–3402. https://doi.org/10.1200/JCO.21.00662
Pilotto S, Carbognin L, Karachaliou N, Ma PC, Rosell R, Tortora G et al (2017) Tracking MET de-addiction in lung cancer: a road towards the oncogenic target. Cancer Treat Rev 60:1–11. https://doi.org/10.1016/j.ctrv.2017.08.002
Salgia R (2017) MET in lung cancer: biomarker selection based on scientific rationale. Mol Cancer Ther 16(4):555–565. https://doi.org/10.1158/1535-7163.MCT-16-0472
Schuler M, Berardi R, Lim WT, de Jonge M, Bauer TM, Azaro A et al (2020) Molecular correlates of response to capmatinib in advanced non-small-cell lung cancer: clinical and biomarker results from a phase I trial. Ann Oncol 31(6):789–797. https://doi.org/10.1016/j.annonc.2020.03.293
Spigel DR, Edelman MJ, O’Byrne K, Paz-Ares L, Mocci S, Phan S et al (2017) Results from the phase III randomized trial of onartuzumab plus erlotinib versus erlotinib in previously treated stage IIIB or IV non-small-cell lung cancer: METLung. J Clin Oncol 35(4):412–420. https://doi.org/10.1200/JCO.2016.69.2160
Tong JH, Yeung SF, Chan AW, Chung LY, Chau SL, Lung RW et al (2016) MET amplification and exon 14 splice site mutation define unique molecular subgroups of non-small cell lung carcinoma with poor prognosis. Clin Cancer Res 22(12):3048–3056. https://doi.org/10.1158/1078-0432.CCR-15-2061
Wolf J, Seto T, Han JY, Reguart N, Garon EB, Groen HJM et al (2020) Capmatinib in MET exon 14-mutated or MET-amplified non-small-cell lung cancer. N Engl J Med 383(10):944–957. https://doi.org/10.1056/NEJMoa2002787
Wu YL, Cheng Y, Zhou J, Lu S, Zhang Y, Zhao J et al (2020) Tepotinib plus gefitinib in patients with EGFR-mutant non-small-cell lung cancer with MET overexpression or MET amplification and acquired resistance to previous EGFR inhibitor (INSIGHT study): an open-label, phase 1b/2, multicentre, randomised trial. Lancet Respir Med 8(11):1132–1143. https://doi.org/10.1016/S2213-2600(20)30154-5
Xu Y, Fan Y (2019) Responses to crizotinib can occur in c-MET overexpressing nonsmall cell lung cancer after developing EGFR-TKI resistance. Cancer Biol Ther 20(2):145–149. https://doi.org/10.1080/15384047.2018.1523851
Funding
This work was supported by the National Natural Science Foundation of China [grant number 82072333 and 81773022]; and the Natural Science Foundation of Hubei Province [grant number 2020CFB808].
Author information
Authors and Affiliations
Contributions
NW contributed to methodology, writing—original draft. YZ contributed to investigation, data curation, formal analysis. YW contributed to methodology, investigation. BH contributed to resources, project administration. JW contributed to resources, project administration. RZ contributed to resources. JF contributed to conceptualization, supervision, project administration. XN contributed to conceptualization, supervision, funding acquisition.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
All authors have declared no conflicts of interest.
Ethical approval
Approval was obtained from the ethics committee of Tongji Medical College of Huazhong University of Science and Technology, and all patients signed informed consent forms. The procedures used in this study adhere to the tenets of the Declaration of Helsinki.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, N., Zhu, Y., Wu, Y. et al. MET overexpression in EGFR L858R mutant treatment-naïve advanced lung adenocarcinoma correlated with poor prognosis: a real-world retrospective study. J Cancer Res Clin Oncol 149, 3219–3228 (2023). https://doi.org/10.1007/s00432-022-04225-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04225-5