Abstract
Objectives
This study investigates whether lead-time bias contributes to the excellent survival of AIS and MIA.
Methods
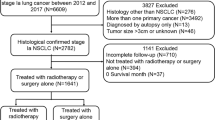
We enrolled patients with resected adenocarcinoma from 2008 to 2012. Age, sex, smoke history, surgical approach, radiological features, invasive stage and postoperative follow-up data were documented. 1:1 PSM was performed to balance the influence of sex and smoking status on survival. After matching, the average age of the two groups was compared to calculate the lead time of diagnosis. The gain in life years for adenocarcinoma diagnosed at pre-/minimally invasive stage was estimated by subtracting the “lead time” and “median survival year of IAC” from “the life expectancy of AIS/MIA patients” referring to the Centre for Health and Information.
Results
There were 124 AIS/MIA patients and 1148 IAC patients. The frequency of female and never-smoking patients in AIS/MIA group was much higher than that in IAC group. PSM analysis identified 124 patient pairs. No cancer-related death and recurrence were observed among AIS/MIA patients 5 years after surgery. For IAC patients, the 5-year disease-specific survival rate was 73.5% and the median survival is 13.5 years. The average age of AIS/MIA group and IAC group are 53.6 years and 58.2 years, respectively. The lead time between diagnosis of AIS/MIA and IAC is 4.6 years. Referring to the Centre for Health and Information, the life expectancy of patients with AIS/MIA diagnosed at 53.6 years old is 28.9 years. With adjustment for the lead time, the gain in life years for adenocarcinoma diagnosed at pre-/minimally invasive stage is 10.8 years.
Conclusions
With adjustment for the lead time between diagnosis of AIS/MIA and IAC, resecting lung adenocarcinoma at pre-/minimally invasive stage can improve life expectancy. The excellent survival of AIS/MIA is not lead-time bias.



Similar content being viewed by others
Abbreviations
- CT:
-
Computed tomography
- AIS:
-
Adenocarcinoma in situ
- MIA:
-
Minimally invasive adenocarcinoma
- IAC:
-
Invasive adenocarcinoma
- IASLC:
-
International Association for the Study of Lung Cancer
- ATS:
-
American thoracic society
- ERS:
-
European respiratory society
- GGO:
-
Ground-glass opacity
- PSM:
-
Propensity score matching
References
Aberle D, Adams A, Berg C et al (2011a) Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 365:395–409
Aberle D, Adams A, Berg C et al (2011b) Reduced lung-cancer mortality with low-dose computed tomographic screening. New Engl J Med 365:395–409
de Koning H, van der Aalst C, de Jong P et al (2020a) Reduced lung-cancer mortality with volume CT screening in a randomized trial. N Engl J Med 382:503–513
de Koning HJ, van der Aalst CM, de Jong PA et al (2020b) Reduced lung-cancer mortality with volume CT screening in a randomized trial. New Engl J Med 382:503–513
Gill RR, Jaklitsch MT, Jacobson FL (2013) Controversies in lung cancer screening. J Am Coll Radiol 10:931–936
Ishida H, Shimizu Y, Sakaguchi H et al (2019) Distinctive clinicopathological features of adenocarcinoma in situ and minimally invasive adenocarcinoma of the lung: a retrospective study. Lung Cancer 129:16–21
Kadota K, Villena-Vargas J, Yoshizawa A et al (2014) Prognostic significance of adenocarcinoma in situ, minimally invasive adenocarcinoma, and nonmucinous lepidic predominant invasive adenocarcinoma of the lung in patients with stage I disease. Am J Surg Pathol 38:448–460
Lee KH, Goo JM, Park SJ et al (2014) Correlation between the size of the solid component on thin-section CT and the invasive component on pathology in small lung adenocarcinomas manifesting as ground-glass nodules. J Thorac Oncol 9:74–82
Liu S, Wang R, Zhang Y et al (2016) Precise diagnosis of intraoperative frozen section is an effective method to guide resection strategy for peripheral small-sized lung adenocarcinoma. J Clin Oncol 34:307–313
Murakami S, Ito H, Tsubokawa N et al (2015) Prognostic value of the new IASLC/ATS/ERS classification of clinical stage IA lung adenocarcinoma. Lung Cancer 90:199–204
National Lung Screening Trial Research T (2019) Lung cancer incidence and mortality with extended follow-up in the national lung screening trial. J Thorac Oncol 14:1732–1742
Noguchi M (2010) Stepwise progression of pulmonary adenocarcinoma–clinical and molecular implications. Cancer Metastasis Rev 29:15–21
Okami J, Shintani Y, Okumura M et al (2019) Demographics, safety and quality, and prognostic information in both the seventh and eighth editions of the TNM classification in 18,973 surgical cases of the Japanese Joint Committee of Lung Cancer Registry Database in 2010. J Thorac Oncol 14:212–222
Pastorino U, Silva M, Sestini S et al (2019) Prolonged lung cancer screening reduced 10-year mortality in the MILD trial: new confirmation of lung cancer screening efficacy. Ann Oncol 30:1162–1169
Rollison DE, Sabel MS (2007) Basic epidemiologic methods for cancer investigations. In: Sabel MS, Sondak VK, Sussman JJ (eds) Essentials of surgical oncology. Mosby, Philadelphia, pp 21–38
Saji H, Matsubayashi J, Akata S et al (2015) Correlation between whole tumor size and solid component size on high-resolution computed tomography in the prediction of the degree of pathologic malignancy and the prognostic outcome in primary lung adenocarcinoma. Acta Radiol 56:1187–1195
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Camcer J 71:209–249
Takahashi M, Shigematsu Y, Ohta M et al (2014) Tumor invasiveness as defined by the newly proposed IASLC/ATS/ERS classification has prognostic significance for pathologic stage IA lung adenocarcinoma and can be predicted by radiologic parameters. J Thorac Cardiovasc Surg 147:54–59
Travis WD, Brambilla E, Noguchi M et al (2011) International Association for the Study of Lung Cancer/American Thoracic Society/European Respiratory Society International Multidisciplinary Classification of Lung Adenocarcinoma. J Thorac Oncol 6:244–285
Travis WD, Brambilla E, Riely GJ (2013) New pathologic classification of lung cancer: relevance for clinical practice and clinical trials. J Clin Oncol 31:992–1001
Travis WD, Brambilla E, Nicholson AG et al (2015) The 2015 World Health organization classification of lung tumors: impact of genetic, clinical and radiologic advances since the 2004 classification. J Thorac Oncol 10:1243–1260
Tzung B, Julien PJ (2010) Lung cancer screening. In: Lewis MI, McKenna RJ, Falk JA et al (eds) Medical management of the thoracic surgery patient. W.B. Saunders, Philadelphia, pp 197–200
Yanagawa M, Johkoh T, Noguchi M et al (2017) Radiological prediction of tumor invasiveness of lung adenocarcinoma on thin-section CT. Med (baltim) 96:e6331
Yang S-C, Lai W-W, Lin C-C et al (2017a) Cost-effectiveness of implementing computed tomography screening for lung cancer in Taiwan. Lung Cancer 108:189–191
Yang S, Lai W, Lin C et al (2017b) Cost-effectiveness of implementing computed tomography screening for lung cancer in Taiwan. Lung Cancer 108:183–191
Yotsukura M, Asamura H, Motoi N et al (2021) Long-term prognosis of patients with resected adenocarcinoma in situ and minimally invasive adenocarcinoma of the lung. J Thorac Oncol 16:1312–1320
Yue C, Meng Q, Wang C et al (2016) The estimation of chinese life expectancy in 2015 and 2020 and influence factors. Chin J Health Stat 33(1):4
Zhang Y, Sun Y, Shen L et al (2013) Predictive factors of lymph node status in small peripheral non-small cell lung cancers: tumor histology is more reliable. Ann Surg Oncol 20:1949–1954
Zhang Y, Ma X, Shen X et al (2020a) Surgery for pre- and minimally invasive lung adenocarcinoma. J Thorac Cardiovasc Surg 163(2):456–464. https://doi.org/10.1016/j.jtcvs.2020.11.151
Zhang Y, Fu F, Chen H (2020b) Management of ground-glass opacities in the lung cancer spectrum. Ann Thorac Surg 110:1796–1804
Zhang Y, Deng C, Fu F et al (2021) Excellent prognosis of patients with invasive lung adenocarcinomas during surgery misdiagnosed as atypical adenomatous hyperplasia, adenocarcinoma in situ, or minimally invasive adenocarcinoma by frozen section. Chest 159:1265–1272
Zojwalla N, Fogelman D, Neugut AI (2004) Cancer screening. In: Legato MJ (ed) Principles of gender-specific medicine. Academic Press, San Diego, pp 693–702
Funding
This work was supported by the National Natural Science Foundation of China (81930073), Shanghai Municipal Science and Technology Major Project (Grant No. 2017SHZDZX01), Shanghai Shenkang Hospital Development Center City Hospital Emerging Cutting-edge Technology Joint Research Project (SHDC12017102) and Shanghai Municipal Health Commission Key Discipline Project (2017ZZ02025 and 2017ZZ01019).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no relevant conflicts of interest.
Ethical statement
The investigation was conducted based on the Declaration of Helsinki and approved by the Institutional Review Board of the Fudan University Shanghai Cancer Center (No. 090977-1). Each patient signed informed consent to allow biological samples to be pathologically examined.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ma, Z., Wang, Z., Li, Y. et al. Detection and treatment of lung adenocarcinoma at pre-/minimally invasive stage: is it lead-time bias?. J Cancer Res Clin Oncol 148, 2717–2722 (2022). https://doi.org/10.1007/s00432-022-04031-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04031-z