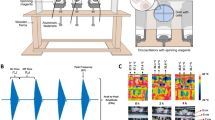
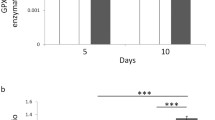
Abstract
Purpose
The mechanisms underlying anticancer effects of electromagnetic fields are poorly understood. An alternating electric field-generating therapeutic device called Optune™ device has been approved for the treatment of glioblastoma (GBM). We have developed a new device that generates oscillating magnetic fields (OMF) by rapid rotation of strong permanent magnets in specially designed patterns of frequency and timing and have used it to treat an end-stage recurrent GBM patient under an expanded access/compassionate use treatment protocol. Here, we ask whether OMF causes selective cytotoxic effects in GBM and whether it is through generation of reactive oxygen species (ROS).
Methods
We stimulated patient derived GBM cells, lung cancer cells, normal human cortical neurons, astrocytes, and bronchial epithelial cells using OMF generators (oncoscillators) of our Oncomagnetic Device and compared the results to those obtained under unstimulated or sham-stimulated control conditions. Quantitative fluorescence microscopy was used to assess cell morphology, viability, and ROS production mechanisms.
Results
We find that OMF induces highly selective cell death of patient derived GBM cells associated with activation of caspase 3, while leaving normal tissue cells undamaged. The cytotoxic effect of OMF is also seen in pulmonary cancer cells. The underlying mechanism is a marked increase in ROS in the mitochondria, possibly in part through perturbation of the electron flow in the respiratory chain.
Conclusion
Rotating magnetic fields produced by a new noninvasive device selectively kill cultured human glioblastoma and non-small cell lung cancer cells by raising intracellular reactive oxygen species, but not normal human tissue cells.








Similar content being viewed by others
Availability of data and materials
Data and material will be made available to all researchers upon request.
Code availability
MATLAB scripts will be made available to all researchers upon request.
References
Arthaut LD, Jourdan N, Mteyrek A, Procopio M, El-Esawi M, d’Harlingue A, Bouchet PE, Witczak J, Ritz T, Klarsfeld A, Birman S, Usselman RJ, Hoecker U, Martino CF, Ahmad M (2017) Blue-light induced accumulation of reactive oxygen species is a consequence of the Drosophila cryptochrome photocycle. PLoS ONE 12:e0171836. https://doi.org/10.1371/journal.pone.0171836
Baskin DS, Sharpe MA, Nguyen L, Helekar SA (2021) Case report: end-stage recurrent glioblastoma treated with a new noninvasive non-contact oncomagnetic device. Front Oncol. https://doi.org/10.3389/fonc.2021.708017
Chernyak BV, Izyumov DS, Lyamzaev KG, Pashkovskaya AA, Pletjushkina OY, Antonenko YN, Sakharov DV, Wirtz KW, Skulachev VP (2006) Production of reactive oxygen species in mitochondria of HeLa cells under oxidative stress. Biochim Biophys Acta 1757:525–534. https://doi.org/10.1016/j.bbabio.2006.02.019
Closs G (1969) A mechanism explaining nuclear spin polarizations in radical combination reactions. J Am Chem Soc 91:4552–4554
Cohen GM (1997) Caspases: the executioners of apoptosis. Biochem J 326(Pt 1):1–16. https://doi.org/10.1042/bj3260001
Dong Z, Zhang G, Qu M, Gimple RC, Wu Q, Qiu Z, Prager BC, Wang X, Kim LJY, Morton AR, Dixit D, Zhou W, Huang H, Li B, Zhu Z, Bao S, Mack SC, Chavez L, Kay SA, Rich JN (2019) Targeting glioblastoma stem cells through disruption of the circadian clock. Cancer Discov 9:1556–1573. https://doi.org/10.1158/2159-8290.CD-19-0215
Englezou PC, Esposti MD, Wiberg M, Reid AJ, Terenghi G (2012) Mitochondrial involvement in sensory neuronal cell death and survival. Exp Brain Res 221:357–367. https://doi.org/10.1007/s00221-012-3179-4
Fabian D, Guillermo Prieto Eibl MDP, Alnahhas I, Sebastian N, Giglio P, Puduvalli V, Gonzalez J, Palmer JD (2019) Treatment of glioblastoma (GBM) with the addition of tumor-treating fields (TTF): a review. Cancers (basel). https://doi.org/10.3390/cancers11020174
Galindo MF, Jordan J, Gonzalez-Garcia C, Cena V (2003) Reactive oxygen species induce swelling and cytochrome c release but not transmembrane depolarization in isolated rat brain mitochondria. Br J Pharmacol 139:797–804. https://doi.org/10.1038/sj.bjp.0705309
Gera N, Yang A, Holtzman TS, Lee SX, Wong ET, Swanson KD (2015) Tumor treating fields perturb the localization of septins and cause aberrant mitotic exit. PLoS ONE 10:e0125269. https://doi.org/10.1371/journal.pone.0125269
Ghiaseddin AP, Shin D, Melnick K, Tran DD (2020) Tumor treating fields in the management of patients with malignant gliomas. Curr Treat Options Oncol 21:76. https://doi.org/10.1007/s11864-020-00773-5
Gillams AR (2005) The use of radiofrequency in cancer. Br J Cancer 92:1825–1829. https://doi.org/10.1038/sj.bjc.6602582
Helekar SA, Voss HU (2016) Transcranial brain stimulation with rapidly spinning high-field permanent magnets. IEEE Access 4:2520–2528
Helekar SA, Convento S, Nguyen L, John BS, Patel A, Yau JM, Voss HU (2018) The strength and spread of the electric field induced by transcranial rotating permanent magnet stimulation in comparison with conventional transcranial magnetic stimulation. J Neurosci Methods 309:153–160. https://doi.org/10.1016/j.jneumeth.2018.09.002
Hore PJ, Mouritsen H (2016) The radical-pair mechanism of magnetoreception. Annu Rev Biophys 45:299–344. https://doi.org/10.1146/annurev-biophys-032116-094545
Jimenez H, Blackman C, Lesser G, Debinski W, Chan M, Sharma S, Watabe K, Lo HW, Thomas A, Godwin D, Blackstock W, Mudry A, Posey J, O’Connor R, Brezovich I, Bonin K, Kim-Shapiro D, Barbault A, Pasche B (2018) Use of non-ionizing electromagnetic fields for the treatment of cancer. Front Biosci (landmark Ed) 23:284–297
Kaptein R, Oosterhoff L (1969) Chemically induced dynamic nuclear polarization III (anomalous multiplets of radical coupling and disproportionation products). Chem Phys Lett 4:214–216
Kovacic P, Somanathan R (2010) Electromagnetic fields: mechanism, cell signaling, other bioprocesses, toxicity, radicals, antioxidants and beneficial effects. J Recept Signal Transduct Res 30:214–226. https://doi.org/10.3109/10799893.2010.488650
Mullen AR, Wheaton WW, Jin ES, Chen PH, Sullivan LB, Cheng T, Yang Y, Linehan WM, Chandel NS, DeBerardinis RJ (2011) Reductive carboxylation supports growth in tumour cells with defective mitochondria. Nature 481:385–388. https://doi.org/10.1038/nature10642
Mun EJ, Babiker HM, Weinberg U, Kirson ED, Von Hoff DD (2018) Tumor-treating fields: a fourth modality in cancer treatment. Clin Cancer Res 24:266–275. https://doi.org/10.1158/1078-0432.CCR-17-1117
Sabharwal SS, Schumacker PT (2014) Mitochondrial ROS in cancer: initiators, amplifiers or an Achilles’ heel? Nat Rev Cancer 14:709–721. https://doi.org/10.1038/nrc3803
Saliev T, Begimbetova D, Masoud AR, Matkarimov B (2019) Biological effects of non-ionizing electromagnetic fields: two sides of a coin. Prog Biophys Mol Biol 141:25–36. https://doi.org/10.1016/j.pbiomolbio.2018.07.009
Sherrard RM, Morellini N, Jourdan N, El-Esawi M, Arthaut LD, Niessner C, Rouyer F, Klarsfeld A, Doulazmi M, Witczak J, d’Harlingue A, Mariani J, McLure I, Martino CF, Ahmad M (2018) Low-intensity electromagnetic fields induce human cryptochrome to modulate intracellular reactive oxygen species. PLoS Biol 16:e2006229. https://doi.org/10.1371/journal.pbio.2006229
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, R European Organisation for, T Treatment of Cancer Brain, G Radiotherapy, and G National Cancer Institute of Canada Clinical Trials (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996. https://doi.org/10.1056/NEJMoa043330
Stupp R, Taillibert S, Kanner A, Read W, Steinberg D, Lhermitte B, Toms S, Idbaih A, Ahluwalia MS, Fink K, Di Meco F, Lieberman F, Zhu JJ, Stragliotto G, Tran D, Brem S, Hottinger A, Kirson ED, Lavy-Shahaf G, Weinberg U, Kim CY, Paek SH, Nicholas G, Bruna J, Hirte H, Weller M, Palti Y, Hegi ME, Ram Z (2017) Effect of tumor-treating fields plus maintenance temozolomide vs maintenance temozolomide alone on survival in patients with glioblastoma: a randomized clinical trial. JAMA 318:2306–2316. https://doi.org/10.1001/jama.2017.18718
Taphoorn MJB, Dirven L, Kanner AA, Lavy-Shahaf G, Weinberg U, Taillibert S, Toms SA, Honnorat J, Chen TC, Sroubek J, David C, Idbaih A, Easaw JC, Kim CY, Bruna J, Hottinger AF, Kew Y, Roth P, Desai R, Villano JL, Kirson ED, Ram Z, Stupp R (2018) Influence of treatment with tumor-treating fields on health-related quality of life of patients with newly diagnosed glioblastoma: a secondary analysis of a randomized clinical trial. JAMA Oncol 4:495–504. https://doi.org/10.1001/jamaoncol.2017.5082
Thallinger C, Fureder T, Preusser M, Heller G, Mullauer L, Holler C, Prosch H, Frank N, Swierzewski R, Berger W, Jager U, Zielinski C (2018) Review of cancer treatment with immune checkpoint inhibitors : Current concepts, expectations, limitations and pitfalls. Wien Klin Wochenschr 130:85–91. https://doi.org/10.1007/s00508-017-1285-9
Tofani S (2015) Electromagnetic energy as a bridge between atomic and cellular levels in the genetics approach to cancer treatment. Curr Top Med Chem 15:572–578. https://doi.org/10.2174/1568026615666150225104217
Tuszynski JA, Wenger C, Friesen DE, Preto J (2016) An overview of sub-cellular mechanisms involved in the action of TTFields. Int J Environ Res Public Health. https://doi.org/10.3390/ijerph13111128
Vadala M, Morales-Medina JC, Vallelunga A, Palmieri B, Laurino C, Iannitti T (2016) Mechanisms and therapeutic effectiveness of pulsed electromagnetic field therapy in oncology. Cancer Med 5:3128–3139. https://doi.org/10.1002/cam4.861
Waliszewski P, Skwarek R, Jeromin L, Minikowski H (1999) On the mitochondrial aspect of reactive oxygen species action in external magnetic fields. J Photochem Photobiol B 52:137–140. https://doi.org/10.1016/s1011-1344(99)90000-3
Wang H, Zhang X (2017) Magnetic fields and reactive oxygen species. Int J Mol Sci. https://doi.org/10.3390/ijms18102175
Wen PY, Weller M, Lee EQ, Alexander BM, Barnholtz-Sloan JS, Barthel FP, Batchelor TT, Bindra RS, Chang SM, Chiocca EA, Cloughesy TF, DeGroot JF, Galanis E, Gilbert MR, Hegi ME, Horbinski C, Huang RY, Lassman AB, Le Rhun E, Lim M, Mehta MP, Mellinghoff IK, Minniti G, Nathanson D, Platten M, Preusser M, Roth P, Sanson M, Schiff D, Short SC, Taphoorn MJB, Tonn JC, Tsang J, Verhaak RGW, von Deimling A, Wick W, Zadeh G, Reardon DA, Aldape KD, van den Bent MJ (2020) Glioblastoma in adults: a Society for Neuro-Oncology (SNO) and European Society of Neuro-Oncology (EANO) consensus review on current management and future directions. Neuro Oncol 22:1073–1113. https://doi.org/10.1093/neuonc/noaa106
Winterbourn CC (2018) Biological production, detection, and fate of hydrogen peroxide. Antioxid Redox Signal 29:541–551. https://doi.org/10.1089/ars.2017.7425
Wong ET, Toms SA, Ahluwalia MS (2015) Technological advances in the treatment of cancer: combining modalities to optimize outcomes. Clin Adv Hematol Oncol 13:1–18
Zorov DB, Juhaszova M, Sollott SJ (2014) Mitochondrial reactive oxygen species (ROS) and ROS-induced ROS release. Physiol Rev 94:909–950. https://doi.org/10.1152/physrev.00026.2013
Zugazagoitia J, Guedes C, Ponce S, Ferrer I, Molina-Pinelo S, Paz-Ares L (2016) Current challenges in cancer treatment. Clin Ther 38:1551–1566. https://doi.org/10.1016/j.clinthera.2016.03.026
Acknowledgements
The authors are grateful to the many patients and their families who have given permission to use their tissues in our studies. They thank Fabio Henrique Brasil da Costa for assistance with one of the experiments, and Lisa Nguyen, Blessy S. John, and Alvin Saldon for constructing the OMF delivering apparatuses used in all experiments.
Funding
This work was supported by a grant from the Translational Research Initiative of the Houston Methodist Research Institute to SAH and DSB; Funding from Kenneth R. Peak Foundation, John S. Dunn Foundation, Taub Foundation, Blanche Green Fund of the Pauline Sterne Wolff Memorial Foundation, Kelly Kicking Cancer Foundation, Methodist Hospital Foundation, Veralan Foundation, and many contributions in honor of Will McKone to DSB. The John S. Dunn Foundation also supports the Distinguished Professorship of MAS.
Author information
Authors and Affiliations
Contributions
Study design: SAH, MAS and KP; conduct of experiments: SH, MAS, and OBI; data analysis and presentation: SH, MAS, SAH and OBI; manuscript preparation: SAH, SH and OBI; interpretation of results and discussion: all authors.
Corresponding author
Ethics declarations
Conflict of interest
SAH, DSB, MAS and KP are listed as inventors on a U.S. patent application filed by Houston Methodist Hospital. The other authors declare no conflicts of interest.
Ethical approval
Informed consent for tumor tissue was obtained from the patients in accordance with an approved Houston Methodist Research Institute Institutional Review Board protocol.
Consent to participate
Not applicable.
Consent for publication
All authors have provided their consent for publication of this research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Helekar, S.A., Hambarde, S., Ijare, O.B. et al. Selective induction of rapid cytotoxic effect in glioblastoma cells by oscillating magnetic fields. J Cancer Res Clin Oncol 147, 3577–3589 (2021). https://doi.org/10.1007/s00432-021-03787-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-021-03787-0