Abstract
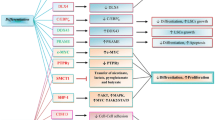
Acute myeloid leukemia (AML) is a malignant clonal disorder affecting myeloid differentiation through mechanisms that include epigenetic dysregulation. Abnormal changes in DNA methylation and gene expression profiles of pathways involved in hematopoietic development, such as Wnt/β-catenin, contribute to the transformation, development, and maintenance of leukemic cells. This review summarizes the alterations of Wnt signaling-related genes at the epigenetic and transcriptional level and their implications for AML prognosis. Among the implications of epigenetic alterations in AML, methylation of Wnt antagonists is related to poor prognosis, whereas their upregulation has been associated with a better clinical outcome. Furthermore, Wnt target genes c-Myc and LEF-1 present distinct implications. LEF-1 expression positively influences the patient overall survival. c-Myc upregulation has been associated with treatment resistance in AML, although c-Myc expression is not exclusively dependent of Wnt signaling. Understanding the signaling abnormalities could help us to further understand leukemogenesis, improve the current risk stratification for AML patients, and even serve to propose novel therapeutic targets.
Similar content being viewed by others
References
Albano F et al (2014) Lymphoid enhancer binding factor-1 (LEF1) expression as a prognostic factor in adult acute promyelocytic leukemia. Oncotarget 5(3):649–658. https://doi.org/10.18632/oncotarget.1619
Alvarez S et al (2010) DNA methylation profiles and their relationship with cytogenetic status in adult acute myeloid leukemia. PLoS ONE. https://doi.org/10.1371/journal.pone.0012197
Aly RM, Taalab MM, Abdsalam EM (2015) Prognostic significance of secreted frizzled-related protein 2 expression in cytogenetically normal primary acute myeloid leukemia. Am J Med Sci 350(5):369–373. https://doi.org/10.1097/MAJ.0000000000000567
An C et al (2015) Clinical significance of reduced SFRP1 expression in acute myeloid leukemia. Leukemia Lymphoma 56(7):2056–2060. https://doi.org/10.3109/10428194.2014.977883
Anastas JN, Moon RT (2013) WNT signalling pathways as therapeutic targets in cancer. Nat Rev Cancer 13(1):11–26. https://doi.org/10.1038/nrc3419
Andrea K et al (2015) Downregulation of the Wnt inhibitor CXXC5 predicts a better prognosis in acute myeloid leukemia. Blood 125(19):2985–2995. https://doi.org/10.1182/blood-2014-12-613703.These
Ashihara E, Takada T, Maekawa T (2015) Targeting the canonical Wnt/β-catenin pathway in hematological malignancies. Cancer Sci 106(6):665–671. https://doi.org/10.1111/cas.12655
Ballabio E, Milne TA (2012) Molecular and epigenetic mechanisms of MLL in human leukemogenesis. Cancers 4(3):904–944. https://doi.org/10.3390/cancers4030904
Bănescu C et al (2016) Influence of XPC, XPD, XPF, and XPG gene polymorphisms on the risk and the outcome of acute myeloid leukemia in a Romanian population. Tumor Biol 37(7):9357–9366. https://doi.org/10.1007/s13277-016-4815-6
Bigas A, Guiu J, Gama-Norton L (2013) Notch and Wnt signaling in the emergence of hematopoietic stem cells. Blood Cells Mol Dis 51(4):264–270. https://doi.org/10.1016/j.bcmd.2013.07.005
Bowen D (2006) Etiology of acute myeloid leukemia in the elderly. Semin Hematol 43(2):82–88
Brembeck FH, Rosa M, Birchmeier W (2006) Balancing cell adhesion and Wnt signaling, the key role of b-catenin. Curr Opin Genet Dev 16:51–59. https://doi.org/10.1016/j.gde.2005.12.007
Brondfield S et al (2015) Direct and indirect targeting of MYC to treat acute myeloid leukemia. Cancer Chemother Pharmacol 76(1):35–46. https://doi.org/10.1007/s00280-015-2766-z
Burgess AW, Faux MC, Layton MJ, Ramsay RG (2011) Wnt signaling and colon tumorigenesis: a view from the periphery. Exp Cell Res 317(19):2748–2758. https://doi.org/10.1016/j.yexcr.2011.08.010
Cardona A, Prada-Arismendy J, Castillo E, Arroyave JC (2018) The reduction of the expression of B-catenin and c-Myc is related to a better outcome in patients with AML. Ann Oncol 29:626. https://doi.org/10.1093/annonc/mdy314.018
Cardona Echeverry AH, Uribe Yunda DF, Cortés-Mancera FM (2016) Actividad antitumoral de la curcumina asociada a la regulación de mecanismos epigenéticos: implicaciones en la vía Wnt/-catenina. Revista Cubana Plantas Med 21(4):1–22
Cavallo RA et al (1998) Drosophila Tcf and Groucho interact to repress wingless signalling activity. Nature 395(6702):604–608. https://doi.org/10.1038/26982
Chen J, Odenike O, Rowley JD (2010) Leukaemogenesis: more than mutant genes. Nat Rev Cancer 10(1):23–36. https://doi.org/10.1038/nrc2765
Chen L et al (2016) Differential regulation of the c-Myc/Lin28 axis discriminates subclasses of rearranged MLL leukemia. Oncotarget 7(18):25208–25223. https://doi.org/10.18632/oncotarget.8199
Cheng CK et al (2012) Secreted-frizzled related protein 1 is a transcriptional repression target of the t (8; 21) fusion protein in acute myeloid leukemia. Blood 118(25):6638–6648. https://doi.org/10.1182/blood-2011-05-354712
Chuang M, Chiu Y, Chou W, Hou H (2015) An mRNA expression signature for prognostication in de novo acute myeloid leukemia patients with normal karyotype. Oncotarget 3(36):39098
Chung EJ et al (2002) Regulation of leukemic cell adhesion, proliferation, and survival by B-catenin. Blood 100(3):982–991
Clements W et al (2011) A somitic Wnt16/Notch pathway specifies haematopoietic stem cells. Nature 474:220–224
Conway O’Brien E, Prideaux S, Chevassut T (2014) The epigenetic landscape of acute myeloid leukemia. Adv Hematol. https://doi.org/10.1155/2014/103175
D’Alò F et al (2004) Polymorphisms of CYP1A1 and glutathione S-transferase and susceptibility to adult acute myeloid leukemia. Haematologica 89:664–670
Dang CV (2012) MYC on the path to cancer. Cell 149(1):22–35. https://doi.org/10.1016/j.cell.2012.03.003
Daniels DL, Weis WI (2005) β-Catenin directly displaces Groucho/TLE repressors from Tcf/Lef in Wnt-mediated transcription activation. Nat Struct Mol Biol 12(4):364–371. https://doi.org/10.1038/nsmb912
Deschler B, Lübbert M (2006) Acute myeloid leukemia: epidemiology and etiology. Cancer 107(9):2099–2107. https://doi.org/10.1002/cncr.22233
Döhner H et al (2010) Diagnosis and management of acute myeloid leukemia in adults: recommendations from an international expert panel, on behalf of the European LeukemiaNet. Blood 115(3):453–474. https://doi.org/10.1182/blood-2009-07-235358
Döhner H, Weisdorf DJ, Bloomfield CD (2015) Acute myeloid leukemia. N Engl J Med 373(12):1136–1152. https://doi.org/10.1056/NEJMra1406184
Döhner H et al (2017) Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 129(4):424–447. https://doi.org/10.1182/blood-2016-08-733196
Dombret H (2011) Gene mutations and AML pathogenesis. Blood 118(20):5366–5367. https://doi.org/10.1182/blood-2011-09-379081
Eaves CJ, Humphries RK (2010) Acute myeloid leukemia and the Wnt pathway. N Engl J Med 362(24):2326–2327. https://doi.org/10.1056/NEJMcibr1003522
Edmaier KE et al (2014) Expression of the lymphoid enhancer factor 1 is required for normal hematopoietic stem and progenitor cell function. Leukemia 28(1):227–230. https://doi.org/10.1038/leu.2013.238
El-Tokhy M, Hussein N, Bedewy A, Barakat M (2014) XPD gene polymorphisms and the effects of induction chemotherapy in cytogenetically normal de novo acute myeloid leukemia patients. Hematology 19(7):397–403
Emami KH et al (2004) A small molecule inhibitor of B-catenin/CREB-binding protein transcription. Proc Natl Acad Sci 101(34):12682–12687
Engelke CG, Chinnaiyan AM (2015) aBETting therapeutic resistance by Wnt signaling. Cell Res 25(11):1187–1188. https://doi.org/10.1038/cr.2015.127
Estey E (2016) Acute myeloid leukemia: 2016 update on risk-stratification and management. Am J Hematol 91(8):824–846. https://doi.org/10.1002/ajh.09108
Fagnocchi L et al (2016) A Myc-driven self-reinforcing regulatory network maintains mouse embryonic stem cell identity. Nat Commun. https://doi.org/10.1038/ncomms11903
Falantes JF et al (2015) Overexpression of GYS1, MIF, and MYC is associated with adverse outcome and poor response to azacitidine in myelodysplastic syndromes and acute myeloid leukemia. Clin Lymphoma Myeloma Leukemia 15(4):236–244. https://doi.org/10.1016/j.clml.2014.10.003
Fauriat C, Olive D (2014) AML drug resistance: C-Myc comes into play. Blood 123(23):3528–3530. https://doi.org/10.1182/blood-2014-04-566711
Ferrara F, Schiffer CA (2013) Acute myeloid leukaemia in adults. Lancet 381(9865):484–495
Figueroa ME et al (2010) DNA methylation signatures identify biologically distinct subtypes in acute myeloid leukemia. Cancer Cell 17(1):13–27. https://doi.org/10.1016/j.ccr.2009.11.020
Fisher D et al (2006) Three Dact gene family members are expressed during embryonic development and in the adult brains of mice. Dev Dyn 235(9):2620–2630
Fiskus W et al (2015) Pre-clinical efficacy of combined therapy with novel β-catenin antagonist BC2059 and histone deacetylase inhibitor against AML cells. Leukemia 29(6):1267–1278. https://doi.org/10.1038/leu.2014.340.Pre-clinical
Florean C et al (2011) Epigenomics of leukemia: from mechanisms to therapeutic applications. Epigenomics 3(5):581–609. https://doi.org/10.2217/epi.11.73
Frame JM et al (2016) Definitive hematopoiesis in the yolk sac emerges from Wnt-responsive hemogenic endothelium independently of circulation and arterial identity. Stem Cells 34(2):431–444. https://doi.org/10.1002/stem.2213
Fu Y et al (2014) Clinical significance of lymphoid enhancer-binding factor 1 expression in acute myeloid leukemia. Leukemia Lymphoma 55(2):371–377. https://doi.org/10.3109/10428194.2013.805759
Gang EJ et al (2014) Small molecule inhibition of CBP/catenin interactions eliminates drug resistant clones in acute lymphoblastic leukemia Eun. Oncogene 33(17):2169–2178. https://doi.org/10.1038/onc.2013.169.Small
Gehrke I, Gandhirajan RK, Kreuzer K (2009) Targeting the WNT/b-catenin/TCF/LEF1 axis in solid and haematological cancers: multiplicity of therapeutic options. Eur J Cancer 45(16):2759–2767. https://doi.org/10.1016/j.ejca.2009.08.003
Genovese G et al (2014) Clonal hematopoiesis and blood-cancer risk inferred from blood DNA sequence. N Engl J Med 371(26):2477–2487. https://doi.org/10.1056/NEJMoa1409405
Griffiths EA et al (2010) Acute myeloid leukemia is characterized by Wnt pathway inhibitor promoter hypermethylation. Leukemia Lymphoma 51(9):1711–1719. https://doi.org/10.3109/10428194.2010.496505
Grimwade D et al (1998) The importance of diagnostic cytogenetics on outcome in AML: analysis of 1612 patients entered into the MRC AML 10 trial. Blood 92(7):2322–2333
Grimwade D et al (2001) The predictive value of hierarchical cytogenetic classification in older adults with acute myeloid leukemia (AML): analysis of 1065 patients entered into the United Kingdom Medical Research Council AML11 trial. Blood 98(5):1312–1320. https://doi.org/10.1182/blood.V98.5.1312
Guillamot M, Cimmino L, Aifantis I (2016) The impact of DNA methylation in hematopoietic malignancies. Trends Cancer 2(2):70–83. https://doi.org/10.1016/j.trecan.2015.12.006
Guo H et al (2014) Decreased SFRP2 expression is associated with intermediate and poor karyotypes in de novo acute myeloid leukemia. Int J Clin Exp Pathol 7(8):4695–4703. https://www.scopus.com/inward/record.url?eid=2-s2.0-84906962467&partnerID=tZOtx3y1
Hamdy M et al (2011) RAD51 and XRCC3 gene polymorphisms and the risk of developing acute myeloid leukemia. J Investig Med 59(7):1121–1130
Hou HA et al (2011) Distinct association between aberrant methylation of Wnt inhibitors and genetic alterations in acute myeloid leukaemia. Br J Cancer 105(12):1927–1933. https://doi.org/10.1038/bjc.2011.471
Huang S et al (2009) Tankyrase inhibition stabilizes axin and antogonizes Wnt signalling. Nature 461:614–620
Hur E-M, Zhou F-Q (2010) GSK3 signaling in neural development. Nat Rev Neurosci 11(8):539–551. https://doi.org/10.1038/nrn2870.GSK3
Jagannathan-Bogdan M, Zon LI (2013) Hematopoiesis. Development 140(12):2463–2467. https://doi.org/10.1242/dev.083147
Jaiswal S et al (2014) Age-related clonal hematopoiesis associated with adverse outcomes. N Engl J Med 371(26):2488–2498. https://doi.org/10.1056/NEJMoa1408617
Jho E et al (2002) Wnt/B-catenin/Tcf signaling induces the transcription of Axin2, a negative regulator of the signaling pathway. Mol Cell Biol 22(4):1172–1183. https://doi.org/10.1128/MCB.22.4.1172
Jiang X et al (2008) DACT3 is an epigenetic regulator of Wnt/beta-catenin signaling in colorectal cancer and is a therapeutic target of histone modifications. Cancer Cell 13(6):529–541. https://doi.org/10.1016/j.ccr.2008.04.019
Kajiguchi T et al (2007) FLT3 regulates beta-catenin tyrosine phosphorylation, nuclear localization, and transcriptional activity in acute myeloid leukemia cells. Leukemia 21(12):2476–2484. https://doi.org/10.1038/sj.leu.2404923
Kao HW et al (2014) Gene mutation patterns in patients with minimally differentiated acute myeloid leukemia. Neoplasia 16(6):481–488. https://doi.org/10.1016/j.neo.2014.06.002
Kawano Y, Kypta R (2003) Secreted antagonists of the Wnt signalling pathway. J Cell Sci 116(13):20627–22634. https://doi.org/10.1242/jcs.00623
Kim Y, Thanendrarajan S (2011) Wnt/ß-Catenin: a new therapeutic approach to acute myeloid leukemia. Leukemia Res Treat. https://doi.org/10.4061/2011/428960
Lancrin C et al (2009) The haemangioblast generates haematopoietic cells through a haemogenic endothelium stage. Nature 457(7231):892–895. https://doi.org/10.1038/nature07679
Lazenby M et al (2014) The prognostic relevance of flt3 and npm1 mutations on older patients treated intensively or non-intensively: a study of 1312 patients in the UK NCRI AML16 trial. Leukemia. https://doi.org/10.1038/leu.2014.90
Lento W et al (2013) Wnt signaling in normal and malignant hematopoiesis. Cold Spring Harbor Perspect Biol. https://doi.org/10.1101/cshperspect.a008011
Levanon D et al (1998) Transcriptional repression by AML1 and LEF-1 is mediated by the TLE/Groucho corepressors. Proc Natl Acad Sci USA 95(20):11590–11595. https://doi.org/10.1073/pnas.95.20.11590
Ley TJ et al (2010) DNMT3A mutations in acute myeloid leukemia. N Engl J Med 363(25):2424–2433. https://doi.org/10.1056/NEJMoa1005143
Ley TJ et al (2013) Genomic and epigenomic landscapes of adult de novo acute myeloid leukemia. N Engl J Med 368(22):2059–2074. https://doi.org/10.1056/NEJMoa1301689
Li VSW et al (2012) Wnt signaling through inhibition of β-catenin degradation in an intact axin1 complex. Cell 149(6):1245–1256. https://doi.org/10.1016/j.cell.2012.05.002
Li K et al (2014a) Sequential combination of decitabine and idarubicin synergistically enhances anti-leukemia effect followed by demethylating Wnt pathway inhibitor promoters and downregulating Wnt pathway nuclear target. J Transl Med 12(1):167. https://doi.org/10.1186/1479-5876-12-167
Li L et al (2014b) SIRT1 activation by a c-MYC oncogenic network promotes the maintenance and drug resistance of human FLT3-ITD acute myeloid leukemia stem cells. Cell Stem Cell 15(4):431–446. https://doi.org/10.1016/j.stem.2014.08.001
Liu T et al (2006) CpG island methylation and expression of the secreted frizzled-related protein gene family in chronic lymphocytic leukemia. Cancer Res 2:653–658. https://doi.org/10.1158/0008-5472.CAN-05-3712
Loeffler D, Kokkaliaris KD, Schroeder T (2011) Wnt to notch relay signaling induces definitive hematopoiesis. Stem Cell 9(1):2–4. https://doi.org/10.1016/j.stem.2011.06.011
Logan CY, Nusse R (2004) The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol 20:781–810. https://doi.org/10.1146/annurev.cellbio.20.010403.113126
Lu J et al (2015) Genetic polymorphisms of CYP1A1 and risk of leukemia: a meta-analysis. OncoTargets Ther 8:2883–2902. https://doi.org/10.2147/OTT.S92259
Luis TC et al (2011) Canonical Wnt signaling regulates hematopoiesis in a dosage-dependent fashion. Cell Stem Cell 9(4):345–356. https://doi.org/10.1016/j.stem.2011.07.017
Luis TC et al (2012) Wnt signaling strength regulates normal hematopoiesis and its deregulation is involved in leukemia development. Leukemia 26(3):414–421. https://doi.org/10.1038/leu.2011.387.Wnt
Lustig B, Behrens J (2003) The Wnt signaling pathway and its role in tumor development. J Cancer Res Clin Oncol 129(4):199–221. https://doi.org/10.1007/s00432-003-0431-0
Marie PJ, Haÿ E (2013) Cadherins and Wnt signalling: a functional link controlling bone formation. BoneKEy Rep 2(4):330. https://doi.org/10.1038/bonekey.2013.64
Martens JHA, Stunnenberg HG (2010) The molecular signature of oncofusion proteins in acute myeloid leukemia. FEBS Lett 584(12):2662–2669. https://doi.org/10.1016/j.febslet.2010.04.002
Martín V et al (2010) Epigenetic regulation of the non-canonical Wnt pathway in acute myeloid leukemia. Cancer Sci 101(2):425–432. https://doi.org/10.1111/j.1349-7006.2009.01413.x
Medvinsky A, Rybtsov S, Taoudi S, Glossary B (2011) Embryonic origin of the adult hematopoietic system: advances and questions. Development 1031:1017–1031. https://doi.org/10.1242/dev.040998
Mehdipour P, Santoro F, Minucci S (2014) Epigenetic alterations in acute myeloid leukemias. FEBS J 282(2015):1786–1800. https://doi.org/10.1111/febs.13142
Metcalfe C, Bienz M (2011) Inhibition of GSK3 by Wnt signalling-two contrasting models. J Cell Sci 124(21):3537–3544. https://doi.org/10.1242/jcs.091991
Metzeler KH et al (2012) High expression of lymphoid enhancer-binding factor-1 (LEF1) is a novel favorable prognostic factor in cytogenetically normal acute myeloid leukemia. Blood 120(10):2118–2126. https://doi.org/10.1182/blood-2012-02-411827
Metzeler KH et al (2016) Spectrum and prognostic relevance of driver gene mutations in acute myeloid leukemia. Blood 128(5):686–699. https://doi.org/10.1182/blood-2016-01-693879
Meyer SC, Levine RL (2014) Translational implications of somatic genomics in acute myeloid leukaemia. Lancet Oncol 15(9):e382–e394. https://doi.org/10.1016/S1470-2045(14)70008-7
Mikesch J-H et al (2007) The emerging role of Wnt signaling in the pathogenesis of acute myeloid leukemia. Leukemia 21(8):1638–1647. https://doi.org/10.1038/sj.leu.2404732
Minke KS et al (2009) Small molecule inhibitors of WNT signaling effectively induce apoptosis in acute myeloid leukemia cells. Eur J Haematol 82(3):165–175. https://doi.org/10.1111/j.1600-0609.2008.01188.x
Moorman AV et al (2001) Karyotype and age in acute myeloid leukemia. Are they linked? Cancer Genet Cytogenet 126:155–161
Mughal MK et al (2017) Acute myeloid leukaemia: expression of MYC protein and its association with cytogenetic risk profile and overall survival. Hematol Oncol 35(3):350–356. https://doi.org/10.1002/hon.2279
Muller-Tidow C et al (2004) Translocation products of AML activate Wnt signaling pathway in hematopoietic cells. Mol Cell Biol 24(7):2890–2904. https://doi.org/10.1128/MCB.24.7.2890
Nagao R et al (2011) Growth inhibition of imatinib-resistant CML cells with the T315I mutation and hypoxia-adaptation by AV65—a novel Wnt/b-catenin signaling inhibitor. Cancer Lett 312(1):91–100. https://doi.org/10.1016/j.canlet.2011.08.002
Neusch M, Kreipe H, Bock O (2004) Aberrant expression of b-catenin discriminates acute myeloid leukaemia from acute lymphoblastic leukaemia. Br J Haematol 126:313–319. https://doi.org/10.1111/j.1365-2141.2004.05049.x
Niida A et al (2004) DKK1, a negative regulator of Wnt signaling, is a target of the b-catenin/TCF pathway. Oncogene 23:8520–8526. https://doi.org/10.1038/sj.onc.1207892
Nostro MC, Cheng X, Keller GM, Gadue P (2008) Wnt, activin, and BMP signaling regulate distinct stages in the developmental pathway from embryonic stem cells to blood. Cell Stem Cell 2(1):60–71. https://doi.org/10.1016/j.stem.2007.10.011
O’Brien EC, Brewin J, Chevassut T (2014) DNMT3A: the DioNysian MonsTer of acute myeloid leukaemia. Ther Adv Hematol 5(6):187–196. https://doi.org/10.1177/2040620714554538
Papaemmanuil E et al (2016) Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med. https://doi.org/10.1056/NEJMoa1516192
Patel J et al (2012) Prognostic relevance of integrated genetic profiling in acute myeloid leukemia. N Engl J Med 366(12):1079–1089. https://doi.org/10.1056/NEJMoa1406617
Paul F et al (2015) Transcriptional heterogeneity and lineage commitment in myeloid progenitors. Cell 163(7):1663–1677. https://doi.org/10.1016/j.cell.2015.11.013
Petropoulos K et al (2008) A novel role for Lef-1, a central transcription mediator of Wnt signaling, in leukemogenesis. J Exp Med 205(3):515–522. https://doi.org/10.1084/jem.20071875
Prada-Arismendy J, Arroyave JC, Röthlisberger S (2017) Molecular biomarkers in acute myeloid leukemia. Blood Rev 31(1):63–76. https://doi.org/10.1016/j.blre.2016.08.005
Roman-Gomez J et al (2007) WNT5A, a putative tumour suppressor of lymphoid malignancies, is inactivated by aberrant methylation in acute lymphoblastic leukaemia. Eur J Cancer 43(18):2736–2746. https://doi.org/10.1016/j.ejca.2007.10.004
Russler-Germain DA et al (2014) The R882H DNMT3A mutation associated with AML dominantly inhibits WT DNMT3A by blocking its ability to form active tetramers. Cancer Cell 25(4):442–454. https://doi.org/10.1038/nature11130.Reduced
Salvatori B et al (2011) Critical role of c-Myc in acute myeloid leukemia involving direct regulation of miR-26a and histone methyltransferase EZH2. Genes Cancer 2(5):585–592. https://doi.org/10.1177/1947601911416357
Savvidou I et al (2017) Beta-catenin inhibitor BC2059 is efficacious as monotherapy or in combination with proteasome inhibitor bortezomib in multiple myeloma. Mol Cancer Ther. https://doi.org/10.1158/1535-7163.MCT-16-0624
Schnatter AR, Rosamilia K, Wojcik NC (2005) Review of the literature on benzene exposure and leukemia subtypes. Chem Biol Interact 153:9–21. https://doi.org/10.1016/j.cbi.2005.03.039
Schoch C et al (2001) Dependence of age-specific incidence of acute myeloid leukemia on karyotype. Blood 98(12):3500. https://doi.org/10.1182/blood.V98.12.3500
Schoch C et al (2004) The influence of age on prognosis of de novo acute myeloid leukemia differs according to cytogenetic subgroups. Haematologica 89:1082–1090
Scholtysek C et al (2013) PPARβ/δ governs Wnt signaling and bone turnover. Nature Med 19(5):608–613. https://doi.org/10.1038/nm.3146
Schoofs T, Berdel W, Muller-Tidow C (2014) Origins of aberrant DNA methylation in acute myeloid leukemia. Leukemia 33(11):2548–2553. https://doi.org/10.1038/nrg3980
Shen Y et al (2011) Gene mutation patterns and their prognostic impact in a cohort of 1185 patients with acute myeloid leukemia. Blood 118(20):5593–5603. https://doi.org/10.1182/blood-2011-03-343988
Staal FJT, Chhatta A, Mikkers H (2016) Caught in a Wnt storm: complexities of Wnt signaling in hematopoiesis. Exp Hematol 44:451
Stölzel F et al (2016) Karyotype complexity and prognosis in acute myeloid leukemia. Blood Cancer J 6(1):e386. https://doi.org/10.1038/bcj.2015.114
Sturgeon C et al (2014) Wnt signaling controls the specification of definitive and primitive hematopoiesis from human pluripotent stem cells. Nat Biotechnol 32(4):554–561. https://doi.org/10.1126/scisignal.2001449.Engineering
Sukhdeo K et al (2007) Targeting the B-catenin/TCF transcriptional complex in the treatment of multiple myeloma. Proc Natl Acad 104(18):7516–7521
Suknuntha K et al (2017) Wnt signaling inhibitor FH535 selectively inhibits cell proliferation and potentiates imatinib-induced apoptosis in myeloid leukemia cell lines. Int J Hematol 105(2):196–205. https://doi.org/10.1007/s12185-016-2116-x
Taskesen E, Staal FJT, Reinders MJT (2015) An integrated approach of gene expression and DNA-methylation profiles of WNT signaling genes uncovers novel prognostic markers in acute myeloid leukemia. BMC Bioinform. https://doi.org/10.1186/1471-2105-16-S4-S4
Tickenbrock L et al (2005) Flt3 tandem duplication mutations cooperate with Wnt signaling in leukemic signal transduction. Blood 105(9):3699–3707. https://doi.org/10.1182/blood-2004-07-2924.Supported
Tran HT et al (2010) Wnt/-catenin signaling is involved in the induction and maintenance of primitive hematopoiesis in the vertebrate embryo. Proc Natl Acad Sci 107(37):16160–16165. https://doi.org/10.1073/pnas.1007725107
Undi RB et al (2015) Wnt signaling: role in regulation of haematopoiesis. Indian J Hematol Blood Transf 32(2):123–134. https://doi.org/10.1007/s12288-015-0585-3
Uribesalgo I, di Croce L (2011) Dynamics of epigenetic modifications in leukemia. Brief Funct Genom 10(1):18–29. https://doi.org/10.1093/bfgp/elr002
Valencia A et al (2009) Wnt signaling pathway is epigenetically regulated by methylation of Wnt antagonists in acute myeloid leukemia. Leukemia 23(9):1658–1666. https://doi.org/10.1038/leu.2009.86
Visnjic D et al (2004) Hematopoiesis is severely altered in mice with an induced osteoblast deficiency. Blood 103(9):3258–3264. https://doi.org/10.1182/blood-2003-11-4011
Vitro I et al (2010) Small molecule inhibitors of Wnt/β-catenin/Lef-1 signaling induces apoptosis in chronic lymphocytic leukemia cells. Neoplasm 12(4):326–335. https://doi.org/10.1593/neo.91972
Voso MT et al (2007) Increased risk of acute myeloid leukaemia due to polymorphisms in detoxification and DNA repair enzymes. Ann Oncol 18(9):1523–1528. https://doi.org/10.1093/annonc/mdm191
Voso MT et al (2008) Prognostic role of glutathione S-transferase polymorphisms in acute myeloid leukemia. Leukemia 22(9):1685–1691. https://doi.org/10.1038/leu.2008.169
Wang Y et al (2010) The Wnt/-catenin pathway is required for the development of leukemia stem cells in AML. Science 327(5973):1650–1653. https://doi.org/10.1126/science.1186624
Wu D, Pan W (2010) GSK3: a multifaceted kinase in Wnt signaling. Trends Biochem Sci 35(3):161–168. https://doi.org/10.1016/j.tibs.2009.10.002.GSK3
Xia B et al (2015) C-Myc plays part in drug resistance mediated by bone marrow stromal cells in acute myeloid leukemia. Leuk Res 39(1):92–99. https://doi.org/10.1016/j.leukres.2014.11.004
Xiao Q et al (2014) GSTT1 and GSTM1 polymorphisms predict treatment outcome for acute myeloid leukemia: a systematic review and meta-analysis. Ann Hematol 93(8):1381–1390
Xie M et al (2014) Age-related mutations associated with clonal hematopoietic expansion and malignancies. Nat Med 20(12):1472–1478. https://doi.org/10.1038/nm.3733
Xu J et al (2008) Clinical significance of nuclear non-phosphorylated beta-catenin in acute myeloid leukaemia and myelodysplastic syndrome. Br J Haematol 140:394–401. https://doi.org/10.1111/j.1365-2141.2007.06914.x
Yang Y et al (2013) Wnt pathway contributes to the protection by bone marrow stromal cells of acute lymphoblastic leukemia cells and is a potential therapeutic target Yang. Cancer Lett 333(1):1–20. https://doi.org/10.1016/j.canlet.2012.11.056.Wnt
Yang K et al (2016) The evolving roles of canonical WNT signaling in stem cells and tumorigenesis: implications in targeted cancer therapies. Lab Investig 96(2):116–136. https://doi.org/10.1038/labinvest.2015.144
Ysebaert L et al (2006) Expression of beta-catenin by acute myeloid leukemia cells predicts enhanced clonogenic capacities and poor prognosis. Leukemia 20(7):1211–1216. https://doi.org/10.1038/sj.leu.2404239
Zhan T, Rindtorff N, Boutros M (2017a) Wnt signaling in cancer. Oncogene 36(11):1461–1473. https://doi.org/10.1038/onc.2016.304
Zhan T, Rindtorff N, Boutros M (2017b) Wnt signaling in cancer. Oncogene 36:1461–1473. https://doi.org/10.1038/onc.2016.304
Acknowledgements
Authors thank to “Departamento Administrativo de Ciencia, Tecnología e Innovación-Colciencias” and “Instituto Tecnológico Metropolitano-ITM” for financial support within the program Jovenes Investigadores en Inovadores Colciencias-ITM (Grant call 706-2015. This work was also supported by Colciencias (Project Code: 115065743926, Grant call 657-2014) and Instituto Tecnológico Metropolitano-ITM (Project Code: P17214).
Funding
This work was supported by Departamento Administrativo de Ciencia, Tecnología e Innovación-Colciencias under Grant 115065743926; and Instituto Tecnológico Metropolitano-ITM Grant P17214.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cardona-Echeverry, A., Prada-Arismendy, J. Deciphering the role of Wnt signaling in acute myeloid leukemia prognosis: how alterations in DNA methylation come into play in patients’ prognosis. J Cancer Res Clin Oncol 146, 3097–3109 (2020). https://doi.org/10.1007/s00432-020-03407-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03407-3