Abstract
Objective
To analyze the relationship between dual-energy spectral CT and epidermal growth factor receptor (EGFR) mutation status in patients with lung adenocarcinoma.
Methods
The quantitative parameters of spectral CT were analyzed in 208 patients with lung adenocarcinoma. The quantitative parameters including CT40keV and CT70keV values, effective atomic number (Zeff), iodine concentration (IC), water concentration (WC), and the slope of the spectral curve (λ HU) were calculated. Statistical analysis was used to determine the clinical characteristics and quantitative parameters for the diagnosis of EGFR-mutation status. The ROC curves were used to calculate diagnostic efficiency.
Results
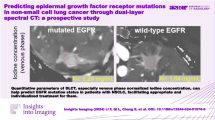
Sex (p = 0.027) and smoking history (p = 0.019) differed significantly according to the EGFR-mutation status. Spectral CT quantitative parameters (CT40keV and CT70keV values, λ HU, Zeff and IC) differed significantly between the EGFR mutant and the EGFR wild-type groups (p < 0.05) during the arterial phase (AP) and venous phase (VP). However, WC was not statistically different between the two groups (p > 0.05). ROC curve analysis revealed the combination of the significantly different quantitative parameters provided the best diagnostic performance for determining the EGFR-mutation status (AUC: 76.0%) in the AP, while the AUC during the VP was 75.6%.
Conclusion
The quantitative parameters of dual-energy spectral CT have potential value for identifying the EGFR-mutation status.





Similar content being viewed by others
References
An N, Zhang Y, Niu H, Li Z, Cai J, Zhao Q, Li Q (2016) EGFR-TKIs versus taxanes agents in therapy for nonsmall-cell lung cancer patients: a PRISMA-compliant systematic review with meta-analysis and meta-regression. Medicine (Baltimore) 95:e5601
Bruns CJ et al (2000) Blockade of the epidermal growth factor receptor signaling by a novel tyrosine kinase inhibitor leads to apoptosis of endothelial cells and therapy of human pancreatic carcinoma. Cancer Res 60:2926–2935
Chae EJ, Song J-W, Seo JB, Krauss B, Jang YM, Song K-S (2008) Clinical utility of dual-energy CT in the evaluation of solitary pulmonary nodules: initial experience. Radiology 249:671–681. https://doi.org/10.1148/radiol.2492071956
Chen Z, Fillmore CM, Hammerman PS, Kim CF, Wong K-K (2014) Non-small-cell lung cancers: a heterogeneous set of diseases. Nat Rev Cancer 14:535–546. https://doi.org/10.1038/nrc3775
Choi C-M, Kim MY, Hwang HJ, Lee JB, Kim WS (2015) Advanced adenocarcinoma of the lung: comparison of CT characteristics of patients with anaplastic lymphoma kinase gene rearrangement and those with epidermal growth factor receptor mutation. Radiology 275:272–279. https://doi.org/10.1148/radiol.14140848
Choi C-M, Kim MY, Lee JC, Kim HJ (2014) Advanced lung adenocarcinoma harboring a mutation of the epidermal growth factor receptor: CT findings after tyrosine kinase inhibitor therapy. Radiology 270:574–582. https://doi.org/10.1148/radiol.13121824
Dearden S, Stevens J, Wu YL, Blowers D (2013) Mutation incidence and coincidence in non small-cell lung cancer: meta-analyses by ethnicity and histology (mutMap). Ann Oncol 24:2371–2376. https://doi.org/10.1093/annonc/mdt205
Global Burden of Disease Cancer C et al (2018) Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 29 cancer groups, 1990 to 2016: a systematic analysis for the global burden of disease study. JAMA Oncol 4:1553–1568. https://doi.org/10.1001/jamaoncol.2018.2706
González-Pérez V et al (2016) Differentiation of benign and malignant lung lesions: dual-energy computed tomography findings. Eur J Radiol 85:1765–1772. https://doi.org/10.1016/j.ejrad.2016.07.019
Hong SJ, Kim TJ, Choi YW, Park J-S, Chung J-H, Lee KW (2016) Radiogenomic correlation in lung adenocarcinoma with epidermal growth factor receptor mutations: Imaging features and histological subtypes. Eur Radiol 26:3660–3668. https://doi.org/10.1007/s00330-015-4196-z
Hsu WH, Yang JCH, Mok TS, Loong HH (2018) Overview of current systemic management of EGFR-mutant NSCLC. Ann Oncol 29:i3–i9. https://doi.org/10.1093/annonc/mdx702
Jeon JH, Kang CH, Kim HS, Seong YW, Park IK, Kim YT (2015) Prognostic and predictive role of epidermal growth factor receptor mutation in recurrent pulmonary adenocarcinoma after curative resection. Eur J Cardiothorac Surg 47:556–562. https://doi.org/10.1093/ejcts/ezu177
Jia T-Y et al (2019) Identifying EGFR mutations in lung adenocarcinoma by noninvasive imaging using radiomics features and random forest modeling. Eur Radiol 29:4742–4750. https://doi.org/10.1007/s00330-019-06024-y
Kerr KM (2009) Pulmonary adenocarcinomas: classification and reporting. Histopathology 54:12–27. https://doi.org/10.1111/j.1365-2559.2008.03176.x
Kim TJ, Lee C-T, Jheon SH, Park J-S, Chung J-H (2016) Radiologic characteristics of surgically resected non-small cell lung cancer with ALK rearrangement or EGFR mutations. Ann Thorac Surg 101:473–480. https://doi.org/10.1016/j.athoracsur.2015.07.062
Kim YT, Seong YW, Jung YJ, Jeon YK, Park IK, Kang CH, Kim JH (2013) The presence of mutations in epidermal growth factor receptor gene is not a prognostic factor for long-term outcome after surgical resection of non-small-cell lung cancer. J Thorac Oncol 8:171–178. https://doi.org/10.1097/JTO.0b013e318277a3bb
Klapper LN, Kirschbaum MH, Sela M, Yarden Y (2000) Biochemical and clinical implications of the ErbB/HER signaling network of growth factor receptors. Adv Cancer Res 77:25–79
Knöss N, Hoffmann B, Krauss B, Heller M, Biederer J (2011) Dual energy computed tomography of lung nodules: differentiation of iodine and calcium in artificial pulmonary nodules in vitro. Eur J Radiol 80:e516–e519. https://doi.org/10.1016/j.ejrad.2010.11.001
Koc G, Courtier JL, Phelps A, Marcovici PA, MacKenzie JD (2014) Computed tomography depiction of small pediatric vessels with model-based iterative reconstruction. Pediatr Radiol 44:787–794. https://doi.org/10.1007/s00247-014-2899-y
Kris MG et al (2003) Efficacy of gefitinib, an inhibitor of the epidermal growth factor receptor tyrosine kinase, in symptomatic patients with non-small cell lung cancer: a randomized trial. JAMA 290:2149–2158
Lee H-J et al (2013) Epidermal growth factor receptor mutation in lung adenocarcinomas: relationship with CT characteristics and histologic subtypes. Radiology 268:254–264. https://doi.org/10.1148/radiol.13112553
Li M, Zhang L, Tang W, Jin Y-J, Qi L-L, Wu N (2019) Identification of epidermal growth factor receptor mutations in pulmonary adenocarcinoma using dual-energy spectral computed tomography. Eur Radiol 29:2989–2997. https://doi.org/10.1007/s00330-018-5756-9
Li M et al (2019) Quantitative features of dual-energy spectral computed tomography for solid lung adenocarcinoma with and mutations, and rearrangement: a preliminary study. Transl Lung Cancer Res 8:401–412. https://doi.org/10.21037/tlcr.2019.08.13
Liu Y et al (2016a) Radiomic features are associated with EGFR mutation status in lung adenocarcinomas. Clin Lung Cancer. https://doi.org/10.1016/j.cllc.2016.02.001
Liu Y et al (2016b) CT features associated with epidermal growth factor receptor mutation status in patients with lung adenocarcinoma. Radiology 280:271–280. https://doi.org/10.1148/radiol.2016151455
Maemondo M et al (2010) Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med 362:2380–2388. https://doi.org/10.1056/NEJMoa0909530
Matsuda I et al (2012) Precision of the measurement of CT numbers: comparison of dual-energy CT spectral imaging with fast kVp switching and conventional CT with phantoms. Jpn J Radiol 30:34–39. https://doi.org/10.1007/s11604-011-0004-0
Ohana M, Jeung MY, Labani A, El Ghannudi S, Roy C (2014) Thoracic dual energy CT: acquisition protocols, current applications and future developments. Diagn Interv Imaging 95:1017–1026. https://doi.org/10.1016/j.diii.2014.01.001
Rosell R et al (2012) Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol 13:239–246. https://doi.org/10.1016/S1470-2045(11)70393-X
Rosell R et al (2009) Screening for epidermal growth factor receptor mutations in lung cancer. N Engl J Med 361:958–967. https://doi.org/10.1056/NEJMoa0904554
Sekine A et al (2013) Prevalence of underlying lung disease in smokers with epidermal growth factor receptor-mutant lung cancer. Oncol Rep 29:2005–2010. https://doi.org/10.3892/or.2013.2320
Shepherd FA et al (2005) Erlotinib in previously treated non-small-cell lung cancer. N Engl J Med 353:123–132
Shi Y et al (2014) A prospective, molecular epidemiology study of EGFR mutations in Asian patients with advanced non-small-cell lung cancer of adenocarcinoma histology (PIONEER). J Thorac Oncol 9:154–162. https://doi.org/10.1097/JTO.0000000000000033
Song Z, Zhu H, Guo Z, Wu W, Sun W, Zhang Y (2013) Correlation of EGFR mutation and predominant histologic subtype according to the new lung adenocarcinoma classification in Chinese patients. Med Oncol 30:645. https://doi.org/10.1007/s12032-013-0645-1
Suh YJ, Lee H-J, Kim YJ, Kim KG, Kim H, Jeon YK, Kim YT (2018) Computed tomography characteristics of lung adenocarcinomas with epidermal growth factor receptor mutation: a propensity score matching study. Lung Cancer 123:52–59. https://doi.org/10.1016/j.lungcan.2018.06.030
Sun PL et al (2012) High incidence of EGFR mutations in Korean men smokers with no intratumoral heterogeneity of lung adenocarcinomas: correlation with histologic subtypes, EGFR/TTF-1 expressions, and clinical features. J Thorac Oncol 7:323–330. https://doi.org/10.1097/JTO.0b013e3182381515
Travis WD (2009) Reporting lung cancer pathology specimens. Impact of the anticipated 7th Edition TNM classification based on recommendations of the IASLC Staging Committee. Histopathology. https://doi.org/10.1111/j.1365-2559.2008.03179.x
Usui K et al (2011) The frequency of epidermal growth factor receptor mutation of nonsmall cell lung cancer according to the underlying pulmonary diseases. Pulm Med 2011:290132. https://doi.org/10.1155/2011/290132
Zhang Y et al (2012) Frequency of driver mutations in lung adenocarcinoma from female never-smokers varies with histologic subtypes and age at diagnosis. Clin Cancer Res 18:1947–1953. https://doi.org/10.1158/1078-0432.CCR-11-2511
Funding
This study has received funding from Talent Innovation and Entrepreneurship Project of Lanzhou (2016-RC-58). Open Fund project of Key Laboratory of Medical Imaging of Gansu Province (GSYX202010).
Author information
Authors and Affiliations
Contributions
GJZ and YTC contributed equally to this study. All authors had access to the study data. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declares that there is no conflict of interest.
Ethical approval
The Ethics Committee of the Second Hospital of Lanzhou University (Lanzhou, China) approved the use of patient materials in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, G., Cao, Y., Zhang, J. et al. Epidermal growth factor receptor mutations in lung adenocarcinoma: associations between dual-energy spectral CT measurements and histologic results. J Cancer Res Clin Oncol 147, 1169–1178 (2021). https://doi.org/10.1007/s00432-020-03402-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03402-8