Abstract
Purpose
This study examined the expression of programmed cell death-ligand 1 (PD-L1), programmed cell death-ligand 2 (PD-L2), and indoleamine 2,3-dioxygenase-1 (IDO1) in tumor cells and cluster of differentiation 8 (CD8)-positive tumor-infiltrating lymphocytes (TILs) in early-stage lung adenocarcinoma according to histological subtypes.
Methods
We evaluated PD-L1, PD-L2, and IDO1 expression in tumor cells and CD8-positive TILs in surgically resected specimens from 196 stage 0 or I lung adenocarcinoma patients by immunohistochemical staining. We also examined the relationships between the expression of PD-L1, PD-L2, and IDO1 in tumor cells and the density of CD8-positive TILs and clinical factors. Patients were divided into three groups: A, adenocarcinoma in situ and minimally invasive adenocarcinoma (N = 32); B, lepidic predominant invasive adenocarcinoma (IAD; LPA; N = 66); and C, IAD except for LPA (N = 98).
Results
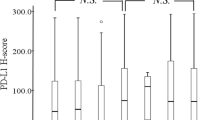
PD-L1 was expressed only in Group C, but not in Groups A or B. The positive ratio of PD-L2 was significantly higher in Group C (63.3%), and that of IDO1 was also significantly higher in Group C (65.3%). The density of CD8-positive TILs was significantly higher in Group C (45 ± 2.4). There was no significant difference between the positive ratios of PD-L2 and IDO1 and the density of CD8-positive TILs in Group A (50.0%, 21.9%, and 36 ± 4.1, respectively) or Group B (60.6%, 25.8%, and 44 ± 3.0, respectively).
Conclusions
No cases in Groups A and B expressed PD-L1. The expression of immune-related factors, especially PD-L1 and IDO1, was significantly associated with Group C. This is the first report of the detailed examination of PD-L1, PD-L2, IDO1, and CD8 expression in lung adenocarcinoma subtypes with lepidic predominant components. Our results could help identify patients who would benefit from perioperative immunotherapy.



Similar content being viewed by others
References
Antonia SJ et al (2017) Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med 377:1919–1929. https://doi.org/10.1056/NEJMoa1709937
Antonia SJ et al (2018) Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N Engl J Med 379:2342–2350. https://doi.org/10.1056/NEJMoa1809697
Borghaei H et al (2015) Nivolumab versus docetaxel in advanced nonsquamous non-small-cell lung cancer. N Engl J Med 373:1627–1639. https://doi.org/10.1056/NEJMoa1507643
Brahmer J et al (2015) Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med 373:123–135. https://doi.org/10.1056/NEJMoa1504627
Erdag G et al (2012) Immunotype and immunohistologic characteristics of tumor-infiltrating immune cells are associated with clinical outcome in metastatic melanoma. Can Res 72:1070–1080. https://doi.org/10.1158/0008-5472.can-11-3218
Fehrenbacher L et al (2016) Atezolizumab versus docetaxel for patients with previously treated non-small-cell lung cancer (POPLAR): a multicentre, open-label, phase 2 randomised controlled trial. Lancet 387:1837–1846
Forde PM, Craft JE, Felip E et al (2017) Checkmate 816: a phase 3, randomized, open-label trial of nivolumab plus ipilimumab vs platinum-doublet chemotherapy as neoadjuvant treatment for early-stage NSCLC. J Clin Oncol 35:TPS8577
Gatalica Z et al (2014) Programmed cell death 1 (PD-1) and its ligand (PD-L1) in common cancers and their correlation with molecular cancer type. Cancer Epidemiol Biomark Prev 23:2965–2970. https://doi.org/10.1158/1055-9965.epi-14-0654
Herbst RS et al (2016) Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet 387:1540–1550
Hirsch FR et al (2017) PD-L1 immunohistochemistry assays for lung cancer: results from phase 1 of the blueprint PD-L1 IHC assay comparison project. J Thorac Oncol 12:208–222. https://doi.org/10.1016/j.jtho.2016.11.2228
Kim H et al (2019) Increased CD3+ T cells with a low FOXP3+/CD8+ T cell ratio can predict anti-PD-1 therapeutic response in non-small cell lung cancer patients. Mod pathol 32:367–375. https://doi.org/10.1038/s41379-018-0142-3
Kinoshita T et al (2017) Determination of poor prognostic immune features of tumour microenvironment in non-smoking patients with lung adenocarcinoma. Eur J Cancer 86:15–27. https://doi.org/10.1016/j.ejca.2017.08.026
Kozuma Y et al (2018) Indoleamine 2,3-dioxygenase 1 and programmed cell death-ligand 1 co-expression correlates with aggressive features in lung adenocarcinoma. Eur J Cancer 101:20–29. https://doi.org/10.1016/j.ejca.2018.06.020
Miyazawa T, Marushima H, Saji H, Kojima K, Hoshikawa M, Takagi M, Nakamura H (2019) PD-L1 expression in non-small-cell lung cancer including various adenocarcinoma subtypes. Ann Thorac Cardiovasc Surg 25:1–9. https://doi.org/10.5761/atcs.oa.18-00163
Nagai Y et al (2005) Genetic heterogeneity of the epidermal growth factor receptor in non-small cell lung cancer cell lines revealed by a rapid and sensitive detection system, the peptide nucleic acid-locked nucleic acid PCR clamp. Cancer Res 65:7276–7282. https://doi.org/10.1158/0008-5472.can-05-0331
Ng Kee Kwong F, Laggner U, McKinney O, Croud J, Rice A, Nicholson AG (2018) Expression of PD-L1 correlates with pleomorphic morphology and histological patterns of non-small-cell lung carcinomas. Histopathology 72:1024–1032. https://doi.org/10.1111/his.13466
Pardoll DM (2012) The blockade of immune checkpoints in cancer immunotherapy. Nat Rev Cancer 12:252–264. https://doi.org/10.1038/nrc3239
Patel SP, Kurzrock R (2015) PD-L1 expression as a predictive biomarker in cancer immunotherapy. Mol Cancer Ther 14:847–856. https://doi.org/10.1158/1535-7163.mct-14-0983
Ratcliffe MJ et al (2017) Agreement between programmed cell death ligand-1 diagnostic assays across multiple protein expression cutoffs in non-small cell lung cancer. Clin Cancer Res 23:3585–3591. https://doi.org/10.1158/1078-0432.ccr-16-2375
Reck M et al (2016a) Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med 375:1823–1833. https://doi.org/10.1056/NEJMoa1606774
Reck M et al (2016b) Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med. https://doi.org/10.1056/NEJMoa1606774
Rimm DL et al (2017) A prospective, multi-institutional, pathologist-based assessment of 4 immunohistochemistry assays for PD-L1 expression in non-small cell lung cancer. JAMA Oncol 3:1051–1058. https://doi.org/10.1001/jamaoncol.2017.0013
Rittmeyer A et al (2016) Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet. https://doi.org/10.1016/s0140-6736(16)32517-x
Rittmeyer A et al (2017) Atezolizumab versus docetaxel in patients with previously treated non-small-cell lung cancer (OAK): a phase 3, open-label, multicentre randomised controlled trial. Lancet 389:255–265. https://doi.org/10.1016/s0140-6736(16)32517-x
Santabarbara G, Maione P, Rossi A, Palazzolo G, Gridelli C (2016) The role of pembrolizumab in the treatment of advanced non-small cell lung cancer. Ann Transl Med 4
Scheel AH et al (2016) Harmonized PD-L1 immunohistochemistry for pulmonary squamous-cell and adenocarcinomas. Mod Pathol 29:1165–1172. https://doi.org/10.1038/modpathol.2016.117
Takada K et al (2016) Clinical significance of PD-L1 protein expression in surgically resected primary lung adenocarcinoma. J Thorac Oncol 11:1879–1890. https://doi.org/10.1016/j.jtho.2016.06.006
Takada K, Toyokawa G, Shoji F, Okamoto T, Maehara Y (2018) The significance of the PD-L1 expression in non-small-cell lung cancer: trenchant double swords as predictive and prognostic markers. Clin Lung Cancer 19:120–129. https://doi.org/10.1016/j.cllc.2017.10.014
Takamori S et al (2018) PD-L2 expression as a potential predictive biomarker for the response to anti-PD-1 drugs in patients with non-small cell lung cancer. Anticancer Res 38:5897–5901. https://doi.org/10.21873/anticanres.12933
Takamori S et al (2019) prognostic impact of programmed death-ligand 2 expression in primary lung adenocarcinoma patients. Ann Surg Oncol 26:1916–1924. https://doi.org/10.1245/s10434-019-07231-z
Teraoka S et al (2017) Early immune-related adverse events and association with outcome in advanced non-small cell lung cancer patients treated with nivolumab: a prospective cohort study. J Thorac Oncol 12:1798–1805. https://doi.org/10.1016/j.jtho.2017.08.022
Theate I et al (2015) Extensive profiling of the expression of the indoleamine 2,3-dioxygenase 1 protein in normal and tumoral human tissues. Cancer Immunol res 3:161–172. https://doi.org/10.1158/2326-6066.cir-14-0137
Travis WD et al (2011) International association for the study of lung cancer/american thoracic society/european respiratory society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 6:244–285. https://doi.org/10.1097/JTO.0b013e318206a221
Travis WD et al (2015) The 2015 World Health Organization classification of lung tumors: impact of genetic, clinical and radiologic advances since the classification. J Thorac Oncol 10:1243–1260. https://doi.org/10.1097/jto.0000000000000630
Tsutsumi S et al (2017) Programmed death-ligand 1 expression at tumor invasive front is associated with epithelial-mesenchymal transition and poor prognosis in esophageal squamous cell carcinoma. Cancer Sci 108:1119–1127. https://doi.org/10.1111/cas.13237
Uryvaev A, Passhak M, Hershkovits D, Sabo E, Bar-Sela G (2018) The role of tumor-infiltrating lymphocytes (TILs) as a predictive biomarker of response to anti-PD1 therapy in patients with metastatic non-small cell lung cancer or metastatic melanoma. Med Oncol 35:25. https://doi.org/10.1007/s12032-018-1080-0
Yang H et al (2018) Prognostic value of PD-L1 expression in combination with CD8(+) TILs density in patients with surgically resected non-small cell lung cancer. Cancer Med 7:32–45. https://doi.org/10.1002/cam4.1243
Yu R et al (2017) Clinical characteristics and programmed cell death ligand-1 expression in adenocarcinoma in situ and minimally invasive adenocarcinoma of lung. Oncotarget 8:97801–97810. https://doi.org/10.18632/oncotarget.22082
Zhai L, Spranger S, Binder DC, Gritsina G, Lauing KL, Giles FJ, Wainwright DA (2015) Molecular pathways: targeting IDO1 and other tryptophan dioxygenases for cancer immunotherapy. Clin Cancer Res 21:5427–5433. https://doi.org/10.1158/1078-0432.ccr-15-0420
Zhang C et al (2019) Genomic landscape and immune microenvironment features of preinvasive and early invasive lung adenocarcinoma. J Thorac Oncol 14:1912–1923. https://doi.org/10.1016/j.jtho.2019.07.031
Acknowledgements
We appreciate technical support from M. Nakamizo (Department of Anatomic Pathology, Graduate School of Medical Sciences, Kyushu University), Y. Kubota and M. Nakajima (Department of Surgery and Science, Graduate School of Medical Sciences, Kyushu University). We thank Edanz Group (https://en-author-services.edanzgroup.com/) for editing a draft of this manuscript.
Funding
This work was not supported by any funding sources.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Takada, K., Toyokawa, G., Kinoshita, F. et al. Expression of PD-L1, PD-L2, and IDO1 on tumor cells and density of CD8-positive tumor-infiltrating lymphocytes in early-stage lung adenocarcinoma according to histological subtype. J Cancer Res Clin Oncol 146, 2639–2650 (2020). https://doi.org/10.1007/s00432-020-03250-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03250-6