Abstract
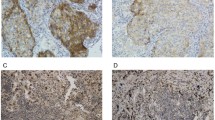
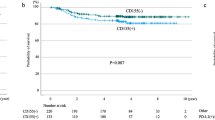
Programmed cell death-ligand 1 (PD-L1) expression on tumor cells is induced by interferon-gamma, suggesting the induction of an anti-tumor immune response. In turn, binding of PD-L1 to programmed cell death 1 (PD-1) triggers an immune checkpoint pathway that contributes to tumor growth. Though it remains to be elucidated, the clinical significance of PD-L1 expression might vary with tumor progression in non–small-cell lung cancer (NSCLC). Immunohistochemical analysis of PD-L1 was done in tumor specimens from patients who underwent radical surgery for stage I–IIIA NSCLC (n = 228). Tumor PD-L1 expression intensity was semi-quantitatively scored and its correlation with various clinicopathological features and postoperative relapse-free survival (RFS) was assessed relative to pathological stage. In stage I, postoperative RFS was significantly prolonged in patients with a high PD-L1 score compared with a low PD-L1 score, exhibiting 5-year relapse-free probabilities of 94.1% and 75.1%, respectively (P = 0.031). A multivariate analysis revealed that a high PD-L1 score was a prognostic factor of longer postoperative RFS (hazard ratio: 0.111, P = 0.033). Conversely, in stages II and IIIA, patients with a high PD-L1 score tended to suffer from postoperative tumor recurrence. In early-stage NSCLC, high tumor PD-L1 expression status represents a biomarker to predict good prognosis after radical surgery and may reflect the induction of an antitumor immune response. However, in locally advanced stage NSCLC, tumor PD-L1 expression status may reflect the execution of an immune checkpoint pathway and predicts the incidence of postoperative tumor recurrence.





Similar content being viewed by others
Abbreviations
- NSCLC:
-
Non–small-cell lung cancer
- OS:
-
Overall survival
- PD-L1:
-
Programmed cell death-ligand 1
- RFS:
-
Relapse-free survival
- 5-y RFP:
-
5-Year relapse-free probability
References
Dong H, Strome SE, Salomao DR, Tamura H, Hirano F, Flies DB, Roche PC, Zhu G, Tamada K, Lennon VA, Celis E, Chen L (2002) Tumor-associated B7–H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med 8(8):793–800
Hansen JD, Du Pasquier L, Lefranc MP, Lopez V, Benmansour A, Boudinot P (2009) The B7 family of immunoregulatory receptors: a comparative and evolutionary perspective. Mol Immunol 46(3):457–472
Brahmer J, Reckamp KL, Baas P, Crinò L, Eberhardt WEE, Antonia S, Pluzanski A, Vokes EE, Holgado E, Waterhouse D, Ready N, Gainor J, Frontera OA, Havel L, Steins M, Garassino M, Aerts JG, Domine M, Paz-Ares L, Reck M, Baudelet C, Harbison C, Lestini B, Spigel D (2015) Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N Engl J Med 373(2):123–135
Borghaei H, Paz-Ares L, Horn L, Spigel D, Steins M, Ready NE, Chow LQ, Voles EE, Felip E, Holgado E, Barlesi F, Kohlhäufl M, Arrieta O, Burgio MA, Fayette J, Lena H, Poddubskaya E, Gerber DE, Gettinger SN, Rudin CM, Rizvi N, Crinò L, Blumenschein GR, Antonia SJ, Dorange C, Harbison CT, Finckenstein FG (2015) Brahmer JR (2015) Nivulumab versus docetaxel nonsquamous non-small-cell lung cancer. N Engl J Med 373(17):1627–1639
Reck M, Rodriguez-Abreu D, Robinson AG, Hui R, Csőszi T, Fülöp A, Gottfried M, Peled N, Tafreshi A, Cuffe S, O’Brien M, Rao S, Hotta K, Leiby MA, Lubiniecki GM, Shentu Y, Rangwala R, Brahmer JR (2016) Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med 375(19):1823–1833
Igarashi T, Teramoto K, Ishida M, Hanaoka J, Daigo Y (2016) Scoreing of PD-L1 expression intensity on pulmonary adenocarcinomas and the correlations with clinicopathological factors. ESMO Open 1(4):e000083
McLaughlin J, Han G, Schalper KA, Carvajal-Hausdorfet D, Pelekanou V, Rehman J, Velcheti V, Herbst R, LoRusso P, Rimm DL (2016) Quantitative assesment of the heterogeneity of PD-L1 expression in non-small-cell lung cancer. JAMA Oncol 2(1):46–54
Nakamura S, Hayashi K, Imaoka Y, Kitamura Y, Akazawa Y, Tabata K, Groen R, Tsuchiya T, Yamasaki N, Nagayasu T, Fukuoka J (2017) Intratumoral heterogeniety of programmed cell death ligand-1 expression is common in lung cancer. PLoS ONE 12(10):e0186192
Li H, Xu Y, Ban D, Song Y, Zhan P, Hu Y, Zhang Q, Zhang F, Liu H, Li T, Sugiura H, Cappuzzo F, lin D, Lv T, (2019) The clinicopathological and prognostic significance of PD-L1 expression assessed by immunohistochemistry in lung cancer: a meta-analysis of 50 studies with 11,383 patients. Transl Lung Cancer Res 8(4):429–449
Mu C-Y, Huang J-A, Chen Y, Chen C, Zhang X-G (2011) High expression of PD-L1 in lung cancer may contribute to poor prognosis and tumor cells immune escape through suppressing tumor infiltrating dendritic cells maturation. Med Oncol 28(3):682–688
Shimoji M, Shimizu S, Sato K, Suda K, Kobayashi Y, Tomizawa K, Takemoto T, Mitsudomi T (2016) Clinical and pathologic features of lung cancer expressing programmed cell death ligand (PD-L1). Lung Cancer 98:69–75
Sun J-M, Zhou W, Choi Y-L, Choi S-J, Kim SE, Wang Z, Dolled-Filhart M, Emancipator K, Wu D, Weiner R, Frisman D, Kim HK, Choi YS, Shim YM, Kim J (2016) Prognostic significance of PD-L1 in patients with non-small cell lung cancer: A large cohort study of surgically resected cases. J Thorac Oncol 11(7):1003–1011
Zhou C, Tang J, Sun H, Zheng X, Li Z, Sun T, Li J, Wang S, Zhou X, Sun H, Cheng Z, Zhang H, Ma H (2017) PD-L1 expression as poor prognostic factor in patients with non-squamous non-small cell lung cancer. Oncotarget 8(35):58457–58468
Takada K, Okamoto T, Toyokawa G, Kozuma Y, Matsubara T, Haratake N, Akamine T, Takamori S, Katsura M, Shoji F, Oda Y, Maehara Y (2017) The expression of PD-L1 protein as a prognostic factor in lung squamous cell carcinoma. Lung Cancer 104:7–15
Sepesi B, Cuentas EP, Canales JR, Behrens C, Correa AM, Vaporciyan A, Weissferdt A, Kalhor N, Maron C, Swisher S, Wistuba I (2017) Programmed death cell ligand 1 (PD-L1) is associated with survival in stage I non-small cell lung cancer. Semin Thorac Cardiovasc Surg 29(3):408–415
Yang C-Y, Lin M-W, Chang Y-L, Wu C-T, Yang P-C (2014) Programmedn cell death-ligand I expression in surgically resected stage I pulmonary adenocarcinoma and its correlation with driver mutations and clinical outcomes. Eur J Cancer 50(7):1361–1369
Cooper WA, Tran T, Vilain RE, Madore J, Selinger CI, Kohonen-Corish M, Yip PY, Yu B, O’Toole SA, McCaughan BC, Yearley JH, Horvath LG, Kao S, Boyer M, Scolyer RA (2015) PD-L1 expression is a favorable prognostic factor in early stage non-small cell carcinoma. Lung Cancer 89(2):181–188
Yang C-Y, Lin M-W, Chang Y-L, Wu C-T, Yang P-C (2016) Programmed cell death-ligand 1 expression is associated with a favourable immune microenvironment and better overall survival in stage I pulmonary squamous cell carcinoma. Eur J Cancer 57:91–103
Igarashi T, Teramoto K, Ishida M, Hanaoka J, Daigo Y (2017) The mechanism of de novo expression of programmed cell death-ligand 1 in squamous cell carcinoma of the lung. Oncol Rep 38(4):2189–2196
Abiko K, Matsumura N, Hamanishi J, Horikawa N, Murakami R, Yamaguchi K, Yoshida Y, Baba T, Konishi I, Mandai M (2015) IFN-γ from lymphocytes induces PD-L1 expression and promotes progression of ovarian cancer. Br J Cancer 112(9):1501–1509
Spranger S, Spaapen RM, Zha Y, Williams J, Meng Y, Ha TT, Gajewski TF (2013) Up-regulation of PD-L1, IDO, and T (regs) in the melanoma tumor microenvironment is driven by CD8(+) T cells. Sci Transl Med 5(200):200ra116
Teramoto K, Igarashi T, Kataoka Y, Ishida M, Hanaoka J, Sumimoto H, Daigo Y (2019) Clinical significance of PD-L1-positive cancer-associated fibroblasts in pN0M0 non-small cell lung cancer. Lung Cancer 173:56–63
Chen Y-B, Mu C-Y, Huang J-A (2012) Clinical significance of programmed death-1 ligand-1 expression in patients with non-small cell lung cancer: a 5-year-follow-up study. Tumori 98(6):751–755
Azuma L, Ota K, Kawahara A, Hattori S, Ikawa E, Harada T, Matsumoto K, Takayama K, Takamori S, Kage M, Hoshimno T, Nakanishi Y, Okamoto I (2014) Association of PD-L1 overexpression with activating EGFR mutations in surgically resected non small-cell lung cancer. Ann Oncol 25(10):1935–1940
Mao Y, Li W, Chen K, Xie V, Liu Q, Duan W, Zhou X, Liang R, Tao M (2015) B7–H1 and B7–H3 are independent predictors of poor prognosis in patients with non-small cell lung cancer. Oncotarget 6(5):3452–3461
Ji M, Liu Y, Li Q, Li X, Ning Z, Zhao W, Shi H, Jiang J, Wu C (2016) PD-1/PD-L1 expression in non-small-cell lung cancer and its correlation with EGFR/KRAS mutations. Cancer Biol Ther 17(4):407–413
Okita R, Maeda A, Shimizu K, Nojima Y, Saisho S, Nakata M (2017) PD-L1 overexpression is partially regulated by EGFR/HER2 signaling and associated with poor prognosis in patients with non-small-cell lung cancer. Cancer Immunol Immunother 66(7):865–876
Zhang Y, Wang L, Li Y, Pan Y, Wang R, Hu H, Li H, Luo X, Ye T, Sun Y, Chen H (2014) Protein expression of programmed death 1 ligand 1 and ligand 2 independently predict poor prognosis in surgically resected lung adenocarcinoma. Onco Targets Ther 7:567–573
Cha YJ, Kim HR, Lee CY, Cho BC, Shim HS (2016) Clinicopathological and prognostic significance of programmed cell death ligand-1 expression in lung adenocarcinoma and its relationship with p53 status. Lung Cancer 297:73–80
Huynh TG, Morales-Oyarvide V, Campo MJ, Gainor JF, Bozkurlar E, Uruga H, Zhao L, Gomez-Caraballo M, Hata AN, Mark EJ, Lanuti M, Engelman JA, Mino-Kenudson M (2016) Programmed cell death ligand 1 expression in resected lung adenocarcinomas: association with immune microenvironment. J Thorac Oncol 11(11):1869–1918
Acknowledgements
This work was supported in part by Grant-in-Aid for Scientific Research from The Japan Society for the Promotion of Science (JSPS KAKENHI Grant Number: 16K10677, 16H06277, 17K10780, and 19H03559). K.T. and Y.D. are members of Shiga Cancer Treatment Project supported by Shiga Prefecture (Japan) and IMSUT (Institute of Medical Sciences, The University of Tokyo) Joint Research Project FY2016-2021.
Funding
This work was supported in part by Grant-in-Aid for Scientific Research from The Japan Society for the Promotion of Science (JSPS KAKENHI Grant Number: 16K10677, 16H06277, 17K10780, and 19H03559).
Author information
Authors and Affiliations
Contributions
KT: Conceptualization, Methodology, Formal analysis, Investigation, Data curation, Writing-Original draft, Project administration, Funding acquisition. TI: Validation, Investigation, Writing-Original draft, Funding acquisition. YK: Validation, Investigation. MI: Validation, Investigation. JH: Resources. HS: Supervision. YD: Supervision, Writing-Review and Editing, Funding acquisition.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Ethical approval
The study design was approved by the Ethical Committee of Shiga University of Medical Science (approval number: R2016-115).
Informed consent
Informed consent was obtained from all patients in the study. The patients agreed to the use of their specimens and clinical data for research and publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Teramoto, K., Igarashi, T., Kataoka, Y. et al. Biphasic prognostic significance of PD-L1 expression status in patients with early- and locally advanced-stage non-small cell lung cancer. Cancer Immunol Immunother 70, 1063–1074 (2021). https://doi.org/10.1007/s00262-020-02755-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-020-02755-w