Abstract
Purpose
Programmed death ligand 1 (PD-L1) and indoleamine 2,3-dioxygenase 1 (IDO1) are immunosuppressive proteins known to be associated with poor prognosis in various cancers. However, their expression and clinical relevance in osteosarcoma remain unknown. In this study, the relationships of PD-L1 and IDO1 expression with clinicopathological features and prognosis were explored.
Methods
The expression of PD-L1, IDO1, CD3, CD4, and CD8 in 112 formalin-fixed, paraffin-embedded tumor tissues collected by biopsy or surgical resection from 56 osteosarcoma patients was evaluated immunohistochemically. Moreover, four osteosarcoma cell lines were evaluated for the effects of IFNγ on PD-L1 and IDO1 mRNA expression by real-time reverse-transcription polymerase chain reaction.
Results
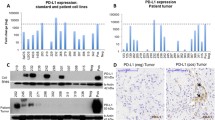
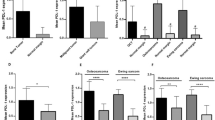
In pre-neoadjuvant chemotherapy (NAC) primary specimens, 10 cases (17%) showed PD-L1 expression and 12 (21%) showed IDO1 expression. Six of ten cases (60%) with PD-L1 positivity co-expressed IDO1. In post-NAC metastatic lesions, the frequency of immunoexpression of PD-L1 and IDO1 was increased compared with that in pre-NAC specimens. PD-L1 and/or IDO1 expression was not associated with poor prognosis. PD-L1 immunoexpression was significantly associated with the infiltration of CD3+ T cells, CD4+ T cells, and CD8+ T cells; while, IDO1 immunoexpression was significantly associated with the infiltration of CD3+ T cells and CD4+ T cells. In all osteosarcoma cell lines, PD-L1 and IDO1 expression was upregulated by stimulation with IFNγ.
Conclusion
Our results suggest that the PD-L1 and IDO1 immune checkpoint inhibitors may provide clinical benefit in osteosarcoma patients with metastatic lesions after conventional chemotherapy.








Similar content being viewed by others
Abbreviations
- PD-L1:
-
Programmed death ligand 1
- IDO1:
-
Indoleamine 2,3-dioxygenase 1
- NAC:
-
Neoadjuvant chemotherapy
- TIL:
-
Tumor-infiltrating lymphocyte
- MTX:
-
Methotrexate
- CDDP:
-
Cisplatin
- ADR:
-
Adriamycin
- OS:
-
Overall survival
- EFS:
-
Event-free survival
References
Abiko K, Mandai M, Hamanishi J et al (2013) PD-L1 on tumor cells is induced in ascites and promotes peritoneal dissemination of ovarian cancer through CTL dysfunction. Clin Cancer Res 19:1363–1374. https://doi.org/10.1158/1078-0432.CCR-12-2199
Abiko K, Matsumura N, Hamanishi J et al (2015) IFN-gamma from lymphocytes induces PD-L1 expression and promotes progression of ovarian cancer. Br J Cancer 112:1501–1509. https://doi.org/10.1038/bjc.2015.101
Angela Y, Haferkamp S, Weishaupt C et al (2019) Combination of denosumab and immune checkpoint inhibition: experience in 29 patients with metastatic melanoma and bone metastases. Cancer Immunol Immunother 68:1187–1194. https://doi.org/10.1007/s00262-019-02353-5
Dunn GP, Koebel CM, Schreiber RD (2006) Interferons, immunity and cancer immunoediting. Nat Rev Immunol 2006:836–848
Castro F, Cardoso AP, Gonçalves RM, Serre K, Oliveira MJ (2018) Interferon-gamma at the crossroads of tumor immune surveillance or evasion. Front Immunol 9:847. https://doi.org/10.3389/fimmu.2018.00847
Deng C, Li Z, Guo S et al (2017) Tumor PD-L1 expression is correlated with increased TILs and poor prognosis in penile squamous cell carcinoma. Oncoimmunology 6:e1269047. https://doi.org/10.1080/2162402X.2016.1269047
Fournel L, Wu Z, Stadler N et al (2019) Cisplatin increases PD-L1 expression and optimizes immune check-point blockade in non-small cell lung cancer. Cancer Lett 464:5–14. https://doi.org/10.1016/j.canlet.2019.08.005
Gao F, Zhang C, Qiu WX et al (2018) PD-1 blockade for improving the antitumor efficiency of polymer-doxorubicin nanoprodrug. Small 14:e1802403. https://doi.org/10.1002/smll.201802403
Gill D, Hahn AW, Sonpavde G, Agarwal N (2016) Immunotherapy of advanced renal cell carcinoma: current and future therapies. Hum Vaccin Immunother 12:2997–3004. https://doi.org/10.1080/21645515.2016.1212794
Gu L, Chen M, Guo D et al (2017) PD-L1 and gastric cancer prognosis: a systematic review and meta-analysis. PLoS ONE 12:e0182692. https://doi.org/10.1371/journal.pone.0182692
Harrison DJ, Schwartz CL (2017) Osteogenic sarcoma: systemic chemotherapy options for localized disease. Curr Treat Options Oncol 18:24. https://doi.org/10.1007/s11864-017-0464-2
Iwamoto Y, Tanaka K, Isu K et al (2009) Multiinstitutional phase II study of neoadjuvant chemotherapy for osteosarcoma (NECO study) in Japan: NECO-93J and NECO-95J. J Orthop Sci 14:397–404. https://doi.org/10.1007/s00776-009-1347-6
Jie HB, Srivastava RM, Argiris A, Bauman JE, Kane LP, Ferris RL (2017) Increased PD-1(+) and TIM-3(+) TILs during cetuximab therapy inversely correlate with response in head and neck cancer patients. Cancer Immunol Res 5:408–416. https://doi.org/10.1158/2326-6066.CIR-16-0333
Jung HI, Jeong D, Ji S, Ahn TS et al (2017) Overexpression of PD-L1 and PD-L2 is associated with poor prognosis in patients with hepatocellular carcinoma. Cancer Res Treat 49:246–254. https://doi.org/10.4143/crt.2016.066
Kim HS, Kim JY, Lee YJ et al (2018) Expression of programmed cell death ligand 1 and immune checkpoint markers in residual tumors after neoadjuvant chemotherapy for advanced high-grade serous ovarian cancer. Gynecol Oncol 151:414–421. https://doi.org/10.1016/j.ygyno.2018.08.023
Kiyozumi Y, Baba Y, Okadome K et al (2019) IDO1 expression is associated with immune tolerance and poor prognosis in patients with surgically resected esophageal cancer. Ann Surg 269:1101–1108. https://doi.org/10.1097/SLA.0000000000002754
Koirala P, Roth ME, Gill J et al (2016) Immune infiltration and PD-L1 expression in the tumor microenvironment are prognostic in osteosarcoma. Sci Rep 6:30093. https://doi.org/10.1038/srep30093
Kozuma Y, Takada K, Toyokawa G et al (2018) Indoleamine 2,3-dioxygenase 1 and programmed cell death-ligand 1 co-expression correlates with aggressive features in lung adenocarcinoma. Eur J Cancer 101:20–29. https://doi.org/10.1016/j.ejca.2018.06.020
Liao Y, Chen L, Feng Y et al (2017) Targeting programmed cell death ligand 1 by CRISPR Cas9 in osteosarcoma cells. Oncotarget 8:30276–30287. https://doi.org/10.18632/oncotarget.16326
Lin YM, Sung WW, Hsieh M et al (2015) High PD-L1 expression correlates with metastasis and poor prognosis in oral squamous cell carcinoma. PLoS ONE 10:e0142656. https://doi.org/10.1371/journal.pone.0142656
Lo CS, Sanii S, Kroeger DR et al (2017) Neoadjuvant chemotherapy of ovarian cancer results in three patterns of tumor-infiltrating lymphocyte response with distinct implications for immunotherapy. Clin Cancer Res 23:925–934. https://doi.org/10.1158/1078-0432.CCR-16-1433
Ma WJ, Wang X, Yan WT et al (2018) Indoleamine-2,3-dioxygenase 1/cyclooxygenase 2 expression prediction for adverse prognosis in colorectal cancer. World J Gastroenterol 24:2181–2190. https://doi.org/10.3748/wjg.v24.i20.2181
Mandai M, Hamanishi J, Abiko K, Matsumura N, Baba T, Konishi I (2016) Dual faces of IFNgamma in cancer progression: a role of PD-L1 induction in the determination of pro- and antitumor immunity. Clin Cancer Res 22:2329–2334. https://doi.org/10.1158/1078-0432.CCR-16-0224
Meng Y, Liang H, Hu J et al (2018) PD-L1 expression correlates with tumor infiltrating lymphocytes and response to neoadjuvant chemotherapy in cervical cancer. J Cancer 9:2938–2945. https://doi.org/10.7150/jca.22532
Mesnage SJL, Auguste A, Genistie C et al (2017) Neoadjuvant chemotherapy (NACT) increases immune infiltration and programmed death-ligand 1 (PD-L1) expression in epithelial ovarian cancer (EOC). Ann Oncol 28:651–657. https://doi.org/10.1093/annonc/mdw625
Miyoshi H, Kiyasu J, Kato T et al (2016) PD-L1 expression on neoplastic or stromal cells is respectively a poor or good prognostic factor for adult T-cell leukemia/lymphoma. Blood 128:1374–1381. https://doi.org/10.1182/blood-2016-02-698936
Muenst S, Schaerli AR, Gao F et al (2014) Expression of programmed death ligand 1 (PD-L1) is associated with poor prognosis in human breast cancer. Breast Cancer Res Treat 146:15–24. https://doi.org/10.1007/s10549-014-2988-5
Nakanishi J, Wada Y, Matsumoto K, Azuma M, Kikuchi K, Ueda S (2007) Overexpression of B7–H1 (PD-L1) significantly associates with tumor grade and postoperative prognosis in human urothelial cancers. Cancer Immunol Immunother 56:1173–1182. https://doi.org/10.1007/s00262-006-0266-z
Negri GL, Grande BM, Delaidelli A et al (2019) Integrative genomic analysis of matched primary and metastatic pediatric osteosarcoma. J Pathol 249:319–331. https://doi.org/10.1002/path.5319
Okita R, Maeda A, Shimizu K, Nojima Y, Saisho S, Nakata M (2017) PD-L1 overexpression is partially regulated by EGFR/HER2 signaling and associated with poor prognosis in patients with non-small-cell lung cancer. Cancer Immunol Immunother 66:865–876. https://doi.org/10.1007/s00262-017-1986-y
Palmerini EAC, Picci P, Pileri S, Marafioti T, Lollini PL, Scotlandi K et al (2017) Tumoral immune-infiltrate (IF), PD-L1 expression and role of CD8 TIA-1 lymphocytes in localized osteosarcoma patients treated within protocol ISG-OS1. Oncotarget 8:111836–111846. https://doi.org/10.18632/oncotarget.22912
Parra ER, Villalobos P, Zhang J et al (2018) Immunohistochemical and image analysis-based study shows that several immune checkpoints are co-expressed in non-small cell lung carcinoma tumors. J Thorac Oncol 13:779–791. https://doi.org/10.1016/j.jtho.2018.03.002
Pelekanou V, Carvajal-Hausdorf DE, Altan M et al (2017) Effect of neoadjuvant chemotherapy on tumor-infiltrating lymphocytes and PD-L1 expression in breast cancer and its clinical significance. Breast Cancer Res 19:91. https://doi.org/10.1186/s13058-017-0884-8
Perea F, Bernal M, Sanchez-Palencia A et al (2017) The absence of HLA class I expression in non-small cell lung cancer correlates with the tumor tissue structure and the pattern of T cell infiltration. Int J Cancer 140:888–899. https://doi.org/10.1002/ijc.30489
Rosenbaum MW, Gigliotti BJ, Pai SI et al (2018) PD-L1 and IDO1 are expressed in poorly differentiated thyroid carcinoma. Endocr Pathol 29:59–67. https://doi.org/10.1007/s12022-018-9514-y
Schalper KA, Carvajal-Hausdorf D, McLaughlin J et al (2017) Differential expression and significance of PD-L1, IDO-1, and B7–H4 in human lung. Cancer Clin Cancer Res 23:370–378. https://doi.org/10.1158/1078-0432.CCR-16-0150
Seeber A, Klinglmair G, Fritz J et al (2018) High IDO-1 expression in tumor endothelial cells is associated with response to immunotherapy in metastatic renal cell carcinoma. Cancer Sci 109:1583–1591. https://doi.org/10.1111/cas.13560
Sharma P, Retz M, Siefker-Radtke A et al (2017) Nivolumab in metastatic urothelial carcinoma after platinum therapy (CheckMate 275): a multicentre, single-arm, phase 2 trial. Lancet Oncol 18:312–322. https://doi.org/10.1016/S1470-2045(17),30065-7
Shen JK, Cote GM, Choy E et al (2014) Programmed cell death ligand 1 expression in osteosarcoma. Cancer Immunol Res 2:690–698. https://doi.org/10.1158/2326-6066.CIR-13-0224
Shimizu T, Fuchimoto Y, Fukuda K, Okita H, Kitagawa Y, Kuroda T (2017) The effect of immune checkpoint inhibitors on lung metastases of osteosarcoma. J Pediatr Surg 52:2047–2050. https://doi.org/10.1016/j.jpedsurg.2017.08.030
Sundara YT, Kostine M, Cleven AH, Bovee JV, Schilham MW, Cleton-Jansen AM (2017) Increased PD-L1 and T-cell infiltration in the presence of HLA class I expression in metastatic high-grade osteosarcoma: a rationale for T-cell-based immunotherapy. Cancer Immunol Immunother 66:119–128. https://doi.org/10.1007/s00262-016-1925-3
Takamori S, Takada K, Tagawa T et al (2018) Differences in PD-L1 expression on tumor and immune cells between lung metastases and corresponding primary tumors. Surg Oncol 27:637–641. https://doi.org/10.1016/j.suronc.2018.08.001
Tawbi HA, Burgess M, Bolejack V et al (2017) Pembrolizumab in advanced soft-tissue sarcoma and bone sarcoma (SARC028): a multicentre, two-cohort, single-arm, open-label, phase 2 trail. Lancet Oncol 18:1493–1501. https://doi.org/10.1016/S1470-2045(17),30624-1
Torabi A, Amaya CN, Wians FH Jr, Bryan BA (2017) PD-1 and PD-L1 expression in bone and soft tissue sarcomas. Pathology 49:506–513. https://doi.org/10.1016/j.pathol.2017.05.003
Troiano G, Caponio VCA, Zhurakivska K, Arena C et al (2018) High PD-L1 expression in the tumour cells did not correlate with poor prognosis of patients suffering for oral squamous cells carcinoma: a meta-analysis of the literature. Cell Prolif. https://doi.org/10.1111/cpr.12537
Uyttenhove C, Pilotte L, Theate IS et al (2003) Evidence for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine 2,3-dioxygenase. Nat Med 9:1269–1274. https://doi.org/10.1038/nm934
Wang Q, Liu F, Liu L (2017) Prognostic significance of PD-L1 in solid tumor: an updated meta-analysis. Medicine (Baltimore) 96:e6369. https://doi.org/10.1097/MD.0000000000006369
Wang D, Niu X, Wang Z et al (2019) Multiregion sequencing reveals the genetic heterogeneity and evolutionary history of osteosarcoma and matched pulmonary metastases. Cancer Res 79:7–20. https://doi.org/10.1158/0008-5472.CAN-18-1086
Yagi T, Baba Y, Ishimoto T et al (2019) PD-L1 expression, tumor-infiltrating lymphocytes, and clinical outcome in patients with surgically resected esophageal cancer. Ann Surg 269:471–478. https://doi.org/10.1097/SLA.0000000000002616
Yamaki S, Yanagimoto H, Tsuta K, Ryota H, Kon M (2017) PD-L1 expression in pancreatic ductal adenocarcinoma is a poor prognostic factor in patients with high CD8(+) tumor-infiltrating lymphocytes: highly sensitive detection using phosphor-integrated dot staining. Int J Clin Oncol 22:726–733. https://doi.org/10.1007/s10147-017-1112-3
Zhai L, Ladomersky E, Lauing KL et al (2017) Infiltrating T cells increase IDO1 expression in glioblastoma and contribute to decreased patient survival. Clin Cancer Res 23:6650–6660. https://doi.org/10.1158/1078-0432.CCR-17-0120
Zhou Y, Shi D, Miao J et al (2017) PD-L1 predicts poor prognosis for nasopharyngeal carcinoma irrespective of PD-1 and EBV-DNA load. Sci Rep 7:43627. https://doi.org/10.1038/srep43627
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflicts of interest.
Ethical approval
The institutional review board at Kyushu University approved this study (approval codes: 29-625, 29-429).
Informed consent
Informed consent was obtained from all participants included in this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
432_2020_3242_MOESM3_ESM.pptx
Supplementary Figure. S1. The flow chart of patients selection of process. We excluded parosteal osteosarcoma, extraskeletal osteosarcoma and low-grade central osteosarcoma. We performed immunohistochemical study and examined antigenicity by evaluating immunoexpression of endogenous control (CD3, CD4 and CD8) and contrasting with HE stain. 56 cases 99 tumors including, primary pre-NAC specimens (56 tumors), primary post-NAC specimens (11 tumors), metastatic specimens (31 tumors) and recurrence specimen (one tumor) were evaluable. Supplementary Figure. S2. Double-staining for interferon-gamma (red: arrow) and tumor-infiltrating lymphocytes (brown), CD3, CD4 and CD8. Scale bars shown 20μl (PPTX 433 kb)
Rights and permissions
About this article
Cite this article
Toda, Y., Kohashi, K., Yamada, Y. et al. PD-L1 and IDO1 expression and tumor-infiltrating lymphocytes in osteosarcoma patients: comparative study of primary and metastatic lesions. J Cancer Res Clin Oncol 146, 2607–2620 (2020). https://doi.org/10.1007/s00432-020-03242-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03242-6