Abstract
Purpose
It is important for hepatocellular carcinoma (HCC) treatment that the targets related to its progression are identified. Clustered regularly interspaced short palindromic repeat (CRISPR)-associated nuclease 9 (Cas9)-based genetic screening is a powerful tool for identifying genes with loss-of-function mutations that are critical for tumour growth and metastasis.
Methods
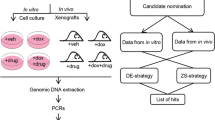
We transduced the human SMMC7721 HCC cell line expressing Cas9 with a human genome-scale CRISPR-Cas9 knockout (GeCKO) lentiviral library A (hGeCKOa) of 65,383 single-guide RNAs (sgRNAs) targeting 19,050 human genes; we then subcutaneously transplanted the transduced cells into nude mice.
Results
The transduced cells were found to proliferate and metastasize faster than the untransduced cells. Through next-generation sequencing, the genes potentially related to HCC proliferation and metastasis were identified. The sgRNAs targeting the ADAMTSL3 and PTEN genes appeared twice on the list of genes related to HCC proliferation and metastasis, respectively. Analysis based on the data mining of Oncomine revealed that the ADAMTSL3 and PTEN genes were expressed at lower levels in HCC cells than they were in normal liver cells, indicating their tumour-suppressive roles. Downregulation of ADAMTSL3 and PTEN displayed poor overall survival (OS) and predicted poor relapse-free survival (RFS), further supporting their tumour-suppressive roles. Moreover, knocking out either the ADAMTSL3 or PTEN genes promoted either the proliferation or metastasis of HCC cells, respectively.
Conclusions
Using both in vitro and in vivo approaches, we described the profound role of the ADAMTSL3 and PTEN genes. This study indicates novel candidate targets for use in HCC treatment and therapy.




Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request.
Abbreviations
- DMEM:
-
Dulbecco’s modified Eagle’s medium
- PBS:
-
Phosphate-buffered saline
- HCC:
-
Hepatocellular carcinoma
- DENA:
-
Diethylnitrosamine
- GeCKO:
-
Genome-scale CRISPR- Cas9 knockout
- sgRNAs:
-
Single-guide RNAs
- CRISPR-Cas9:
-
Clustered regularly interspaced short palindromic repeat/CRISPR-associated nuclease 9
- FBS:
-
Fetal bovine serum
- MOI:
-
Multiplicity of infection
- DAB:
-
3, 3′- Diaminobenzidine
- EMT:
-
Epithelial–mesenchymal transition
- BSD:
-
Blasticidin
- H&E staining:
-
Hematoxylin–eosin staining
- PAM:
-
Protospacer adjacent motif
- CCK-8:
-
Cell counting kit-8
References
Belghiti J, Kianmanesh R (2005) Surgical treatment of hepatocellular carcinoma. HPB 7:42–49. https://doi.org/10.1080/13651820410024067
Boyault S et al (2007) Transcriptome classification of HCC is related to gene alterations and to new therapeutic targets. Hepatology 45:42–52. https://doi.org/10.1002/hep.21467
Chen S et al (2015) Genome-wide CRISPR screen in a mouse model of tumor growth and metastasis. Cell 160:1246–1260. https://doi.org/10.1016/j.cell.2015.02.038
Chen M-n et al (2019) Performing data mining and integrative analysis of biomarker in breast cancer using multiple publicly accessible databases. J Vis Exp. https://doi.org/10.3791/59238
Cong L et al (2013) Multiplex genome engineering using CRISPR/Cas systems. Science 339:819–823. https://doi.org/10.1126/science.1231143
Deng Z, Yang C, Wang G, Guo S, Liu Y, Jia J, Zhao J (2009) Gene therapy targeted to telomerase in HCC by AF-hTERT-TK/GCV The Chinese–German. J Clin Oncol 8:415–419. https://doi.org/10.1007/s10330-009-0036-4
El-Serag HB (2012) Epidemiology of viral hepatitis and hepatocellular carcinoma. Gastroenterology 142(1264–1273):e1261. https://doi.org/10.1053/j.gastro.2011.12.061
Erstad DJ, Tanabe KK (2017) Hepatocellular carcinoma: early-stage management challenges. J Hepatocell Carcinoma 4:81–92. https://doi.org/10.2147/JHC.S107370
Francia G, Cruz-Munoz W, Man S, Xu P, Kerbel RS (2011) Mouse models of advanced spontaneous metastasis for experimental therapeutics. Nat Rev Cancer 11:135–141. https://doi.org/10.1038/nrc3001
Gaj T, Gersbach CA, Barbas CF 3rd (2013) ZFN, TALEN, and CRISPR/Cas-based methods for genome engineering. Trends Biotechnol 31:397–405. https://doi.org/10.1016/j.tibtech.2013.04.004
Galuppo R, Ramaiah D, Ponte OM, Gedaly R (2014) Molecular therapies in hepatocellular carcinoma: what can we target? Dig Dis Sci 59:1688–1697. https://doi.org/10.1007/s10620-014-3058-x
Gao H, Le Y, Wu X, Silberstein LE, Giese RW, Zhu Z (2010) VentX, a novel lymphoid-enhancing factor/T-cell factor-associated transcription repressor, is a putative tumor suppressor. Cancer Res 70:202–211. https://doi.org/10.1158/0008-5472.CAN-09-2668
Goonesekere NCW, Andersen W, Smith A, Wang X (2018) Identification of genes highly downregulated in pancreatic cancer through a meta-analysis of microarray datasets: implications for discovery of novel tumor-suppressor genes and therapeutic targets. J Cancer Res Clin Oncol 144:309–320. https://doi.org/10.1007/s00432-017-2558-4
Hu T-H et al (2003) Expression and prognostic role of tumor suppressor gene PTEN/MMAC1/TEP1 in hepatocellular carcinoma. Cancer 97:1929–1940. https://doi.org/10.1002/cncr.11266
Kan Z et al (2013) Whole-genome sequencing identifies recurrent mutations in hepatocellular carcinoma. Genome Res 23:1422–1433. https://doi.org/10.1101/gr.154492.113
Koo B-H, Hurskainen T, Mielke K, Aung PP, Casey G, Autio-Harmainen H, Apte SS (2007) ADAMTSL3/punctin-2, a gene frequently mutated in colorectal tumors, is widely expressed in normal and malignant epithelial cells, vascular endothelial cells and other cell types, and its mRNA is reduced in colon cancer. Int J Cancer 121:1710–1716. https://doi.org/10.1002/ijc.22882
Laurent-Puig P et al (2001) Genetic alterations associated with hepatocellular carcinomas define distinct pathways of hepatocarcinogenesis. Gastroenterology 120:1763–1773. https://doi.org/10.1053/gast.2001.24798
Lin S, Hoffmann K, Schemmer P (2012) Treatment of hepatocellular carcinoma: a systematic review. Liver Cancer 1:144–158. https://doi.org/10.1159/000343828
Magnani CF, Tettamanti S, Maltese F, Turazzi N, Biondi A, Biagi E (2013) Advanced targeted, cell and gene-therapy approaches for pediatric hematological malignancies: results and future perspectives. Front Oncol 3:106. https://doi.org/10.3389/fonc.2013.00106
Mali P et al (2013) RNA-guided human genome engineering via Cas9. Science 339:823–826. https://doi.org/10.1126/science.1232033
Mashimo T (2014) Gene targeting technologies in rats: zinc finger nucleases, transcription activator-like effector nucleases, and clustered regularly interspaced short palindromic repeats. Dev Growth Differ 56:46–52. https://doi.org/10.1111/dgd.12110
Mazurenko NNBIS, Bliyev AY et al (2003) Genetic alterations at chromosome 6 associated with cervical cancer progression. Mol Biol 37:404–411
Ooki A, Yamashita K, Yamaguchi K, Mondal A, Nishimiya H, Watanabe M (2013) DNA damage-inducible gene, reprimo functions as a tumor suppressor and is suppressed by promoter methylation in gastric cancer. Mol Cancer Res MCR 11:1362–1374. https://doi.org/10.1158/1541-7786.MCR-13-0091
Rhodes DR et al (2004) ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia 6:1–6. https://doi.org/10.1016/s1476-5586(04)80047-2
Rhodes DR et al (2007) Oncomine 3.0: genes, pathways, and networks in a collection of 18,000 cancer gene expression profiles. Neoplasia 9:166–180. https://doi.org/10.1593/neo.07112
Richter H, Randau L, Plagens A (2013) Exploiting CRISPR/Cas: interference mechanisms and applications. Int J Mol Sci 14:14518–14531. https://doi.org/10.3390/ijms140714518
Roessler S et al (2010) A unique metastasis gene signature enables prediction of tumor relapse in early-stage hepatocellular carcinoma patients. Can Res 70:10202–10212. https://doi.org/10.1158/0008-5472.CAN-10-2607
Sanjana NE, Shalem O, Zhang F (2014) Improved vectors and genome-wide libraries for CRISPR screening. Nat Methods 11:783–784. https://doi.org/10.1038/nmeth.3047
Shalem O et al (2014) Genome-scale CRISPR-Cas9 knockout screening in human cells. Science 343:84–87. https://doi.org/10.1126/science.1247005
Steven Leary WU, Raymond A, Samuel C, Douglas C, Temple G, Cheryl G, Sharon GB, Mary Ann M, Robert M, David M, Jan S, Roy Y (2013) %3cAVMA Guidelines for the Euthanasia of Animals 2013 Edition.pdf%3e. In: American Veterinary Medical Association.
Sucharita Bandyopadhyay SKP, Shigeru H, Sadahiro H, Taisei T, Kunio M, Yukio Takano KS, Therese C, David P, Misako W, Steven G, Ying W, Jodi HKW (2004) PTEN up-regulates the tumor metastasis suppressor gene drg-1 in prostate and breast cancer. Cancer Res 64:7655–7660
Thomassen M, Tan Q, Kruse TA (2009) Gene expression meta-analysis identifies chromosomal regions and candidate genes involved in breast cancer metastasis. Breast Cancer Res Treat 113:239–249. https://doi.org/10.1007/s10549-008-9927-2
Valastyan S, Weinberg RA (2011) Tumor metastasis: molecular insights and evolving paradigms. Cell 147:275–292. https://doi.org/10.1016/j.cell.2011.09.024
Vanharanta S, Massague J (2013) Origins of metastatic traits. Cancer Cell 24:410–421. https://doi.org/10.1016/j.ccr.2013.09.007
Vilarinho S, Calvisi DF (2014) New advances in precision medicine for hepatocellular carcinoma recurrence prediction and treatment. Hepatology 60:1812–1814. https://doi.org/10.1002/hep.27311
Wurmbach E et al (2007) Genome-wide molecular profiles of HCV-induced dysplasia and hepatocellular carcinoma. Hepatology 45:938–947. https://doi.org/10.1002/hep.21622
Xue W et al (2014) CRISPR-mediated direct mutation of cancer genes in the mouse liver. Nature 514:380–384. https://doi.org/10.1038/nature13589
Yutaka Midorikawa MM, Wei T, Hiroyuki A (2007) Microarray-based analysis for hepatocellular carcinoma from gene expression profiling to new challenges. World J Gastroenterol 13:1487–1492
Zeng K et al (2018) LACTB, a novel epigenetic silenced tumor suppressor, inhibits colorectal cancer progression by attenuating MDM2-mediated p53 ubiquitination and degradation. Oncogene 37:5534–5551. https://doi.org/10.1038/s41388-018-0352-7
Zhiqiang Zou AA, Hendrix MJC, Ann Thor MN, Shijie S, Kristina R, Elisabeth Seftor RS (1994) Maspin, a serpin with tumor-suppressing activity in human mammary epithelial cells. Science 263:526–529
Acknowledgements
We thank the Tianjin Medical University Cancer Hospital for the assistance with the HCC and adjacent tissue patient biopsies used in this study.
Funding
This study was funded by the National Natural Science Foundation of China (No. 81501531).
Author information
Authors and Affiliations
Contributions
XLZ and QZ conceived and designed the experiment. XLZ, RBL, and RWJ performed the in vitro and in vivo experiments, Western blot and immunohistochemistry experiments. RBL and BFZ completed the Transwell invasion assay and H&E staining, and XLZ, RBL, and QZ performed the bioinformatics analysis. XLZ, RBL, and RWJ contributed to the analysis of the results and to the draft of the manuscript. XLZ, RWJ, and QZ wrote the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethics approval and consent to participate
This animal study was reviewed and approved by the institutional ethical committee (Permit Number:SYXK 2009–0001). The HCC and adjacent tissue patient biopsies were approved by the Tianjin Medical University Cancer Hospital Ethics Committee.
Consent for publication
All authors reviewed and approved the final version of the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhou, X., Li, R., Jing, R. et al. Genome-wide CRISPR knockout screens identify ADAMTSL3 and PTEN genes as suppressors of HCC proliferation and metastasis, respectively. J Cancer Res Clin Oncol 146, 1509–1521 (2020). https://doi.org/10.1007/s00432-020-03207-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03207-9