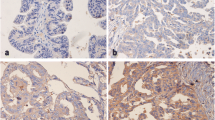
Abstract
Purpose
Fibroblast Growth Factor Receptor 4 (FGFR4) was proposed to hold prognostic significance in high-grade serous ovarian carcinoma (HGSOC). However, information on this deriving from large, representative patient panels is still missing, though such data would be indispensable to validate suitability of FGFR4 as prognostic marker or even pharmacological target.
Methods
1063 ovarian cancer cases were included in this study. Immunohistochemistry (IHC) was performed using two different anti-FGFR4 specific antibodies (HPA027273, sc-124) on an automated staining system. IHC data of both FGFR4 antibodies were available from 995 cases. FGFR4 immunostaining was correlated to prognostic factors including survival using uni- and multivariate proportional hazard models.
Results
FGFR4 was positively associated with advanced FIGO stage, high grade and presence of residual disease. When progression free (PFS) of FGFR4 negative vs. positive patients was compared, patients scored as FGFR4 positive had significantly shortened PFS as compared to those that stained negative. All associations of FGFR4 and shortened PFS were lost during multivariate testing. No significant associations were found in terms of OS.
Conclusions
We were not able to confirm FGFR4 as an independent negative prognosticator as described before. However, FGFR4 was highly prevalent in those cases harboring residual disease after debulking surgery. Since especially patients that could only be debulked sub-optimally may benefit from targeted adjuvant treatment, tyrosine kinase inhibitors targeting FGFRs might turn out to be an interesting future treatment option.




Similar content being viewed by others
Availability of data and material
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
References
Birrer MJ, Johnson ME, Hao K, Wong KK, Park DC, Bell A et al (2007) Whole genome oligonucleotide-based array comparative genomic hybridization analysis identified fibroblast growth factor 1 as a prognostic marker for advanced-stage serous ovarian adenocarcinomas. J Clin Oncol 25(16):2281–2287. https://doi.org/10.1200/jco.2006.09.0795 (Epub 2007/06/01. PubMed PMID: 17538174)
Cancer Genome Atlas Research Network (2011) Integrated genomic analyses of ovarian carcinoma. Nature 474(7353):609–615. https://doi.org/10.1038/nature10166 (Epub 2011/07/02. PubMed PMID: 21720365; PubMed Central PMCID: PMCPMC3163504)
Cho SH, Hong CS, Kim HN, Shin MH, Kim KR, Shim HJ et al (2016) FGFR4 Arg388 is correlated with poor survival in resected colon cancer promoting epithelial to mesenchymal transition. Cancer Res Treat. https://doi.org/10.4143/crt.2016.457 (Epub 2016/11/20. PubMed PMID: 27857023)
du Bois A, Reuss A, Pujade-Lauraine E, Harter P, Ray-Coquard I, Pfisterer J (2009) Role of surgical outcome as prognostic factor in advanced epithelial ovarian cancer: a combined exploratory analysis of 3 prospectively randomized phase 3 multicenter trials: by the Arbeitsgemeinschaft Gynaekologische Onkologie Studiengruppe Ovarialkarzinom (AGO-OVAR) and the Groupe d’Investigateurs Nationaux Pour les Etudes des Cancers de l’Ovaire (GINECO). Cancer 115(6):1234–1244. https://doi.org/10.1002/cncr.24149 (Epub 2009/02/04. PubMed PMID: 19189349)
du Bois A, Herrstedt J, Hardy-Bessard AC, Muller HH, Harter P, Kristensen G et al (2010) Phase III trial of carboplatin plus paclitaxel with or without gemcitabine in first-line treatment of epithelial ovarian cancer. J Clin Oncol 28(27):4162–4169. https://doi.org/10.1200/jco.2009.27.4696 (Epub 2010/08/25. PubMed PMID: 20733132)
Ferlay J, Autier P, Boniol M, Heanue M, Colombet M, Boyle P (2007) Estimates of the cancer incidence and mortality in Europe in 2006. Ann Oncol 18(3):581–592. https://doi.org/10.1093/annonc/mdl498 (Epub 2007/02/09. PubMed PMID: 17287242)
Heinzle C, Erdem Z, Paur J, Grasl-Kraupp B, Holzmann K, Grusch M et al (2014) Is fibroblast growth factor receptor 4 a suitable target of cancer therapy? Curr Pharm Des 20(17):2881–2898 (Epub 2013/08/16. PubMed PMID: 23944363; PubMed Central PMCID: PMCPMC5453246)
Holzmann K, Grunt T, Heinzle C, Sampl S, Steinhoff H, Reichmann N et al (2012) Alternative splicing of fibroblast growth factor receptor IgIII loops in cancer. J Nucleic Acid 2012:950508. https://doi.org/10.1155/2012/950508 (Epub 2011/12/29. PubMed PMID: 22203889; PubMed Central PMCID: PMCPMC3238399)
Hu L, Cong L (2015) Fibroblast growth factor 19 is correlated with an unfavorable prognosis and promotes progression by activating fibroblast growth factor receptor 4 in advanced-stage serous ovarian cancer. Oncol Rep 34(5):2683–2691. https://doi.org/10.3892/or.2015.4212 (Epub 2015/09/02. PubMed PMID: 26323668)
Jänicke F (1999) Das fortgeschrittene Ovarialkarzinom: Entwicklungen der operativen und systemischen Therapie. Dtsch Arztebl Int 96(5):281–287
Jiang XP, Rui XH, Guo CX, Huang YQ, Li Q, Xu Y (2017) A network meta-analysis of eight chemotherapy regimens for treatment of advanced ovarian cancer. Oncotarget 8(12):19125–19136. https://doi.org/10.18632/oncotarget.13253 (Epub 2016/11/12. PubMed PMID: 27835912; PubMed Central PMCID: PMC5386673)
Jonker DJ, Rosen LS, Sawyer MB, de Braud F, Wilding G, Sweeney CJ et al (2011) A phase I study to determine the safety, pharmacokinetics and pharmacodynamics of a dual VEGFR and FGFR inhibitor, brivanib, in patients with advanced or metastatic solid tumors. Ann Oncol 22(6):1413–1419. https://doi.org/10.1093/annonc/mdq599 (Epub 2010/12/07. PubMed PMID: 21131369; PubMed Central PMCID: PMC3139984)
Kalloger SE, Kobel M, Leung S, Mehl E, Gao D, Marcon KM et al (2011) Calculator for ovarian carcinoma subtype prediction. Mod Pathol 24(4):512–521. https://doi.org/10.1038/modpathol.2010.215 (Epub 2010/12/07. PubMed PMID: 21131918)
Knights V, Cook SJ (2010) De-regulated FGF receptors as therapeutic targets in cancer. Pharmacol Ther 125(1):105–117. https://doi.org/10.1016/j.pharmthera.2009.10.001 (Epub 2009/10/31. PubMed PMID: 19874848)
Kobel M, Kalloger SE, Boyd N, McKinney S, Mehl E, Palmer C et al (2008) Ovarian carcinoma subtypes are different diseases: implications for biomarker studies. PLoS Med 5(12):e232. https://doi.org/10.1371/journal.pmed.0050232 (Epub 2008/12/05. PubMed PMID: 19053170; PubMed Central PMCID: PMCPMC2592352)
Li J, Ye Y, Wang M, Lu L, Han C, Zhou Y et al (2016) The over-expression of FGFR4 could influence the features of gastric cancer cells and inhibit the efficacy of PD173074 and 5-fluorouracil towards gastric cancer. Tumour Biol 37(5):6881–6891. https://doi.org/10.1007/s13277-015-4411-1 (Epub 2015/12/15. PubMed PMID: 26662569)
Marme F, Hielscher T, Hug S, Bondong S, Zeillinger R, Castillo-Tong DC et al (2012) Fibroblast growth factor receptor 4 gene (FGFR4) 388Arg allele predicts prolonged survival and platinum sensitivity in advanced ovarian cancer. Int J Cancer 131(4):E586–E591. https://doi.org/10.1002/ijc.27329 (Epub 2011/10/29. PubMed PMID: 22034009)
Perren TJ, Swart AM, Pfisterer J, Ledermann JA, Pujade-Lauraine E, Kristensen G et al (2011) A phase 3 trial of bevacizumab in ovarian cancer. N Engl J Med 365(26):2484–2496. https://doi.org/10.1056/nejmoa1103799 (Epub 2011/12/30. PubMed PMID: 22204725)
Pujade-Lauraine E, Selle F, Weber B, Ray-Coquard IL, Vergote I, Sufliarsky J et al (2016) Volasertib versus chemotherapy in platinum-resistant or -refractory ovarian cancer: a randomized phase II groupe des Investigateurs Nationaux pour l’Etude des Cancers de l’Ovaire Study. J Clin Oncol 34(7):706–713. https://doi.org/10.1200/jco.2015.62.1474 (Epub 2016/01/13. PubMed PMID: 26755507)
Riester M, Wei W, Waldron L, Culhane AC, Trippa L, Oliva E, Kim SH, Michor F, Huttenhower C, Parmigiani G, Birrer MJ (2014) Risk Prediction for Late-Stage Ovarian Cancer by Meta-analysis of 1525 Patient Samples. J Natl Cancer Inst. https://doi.org/10.1093/jnci/dju048
Tabernero J, Bahleda R, Dienstmann R, Infante JR, Mita A, Italiano A et al (2015) Phase I dose-escalation study of JNJ-42756493, an oral pan-fibroblast growth factor receptor inhibitor, in patients with advanced solid tumors. J Clin Oncol 33(30):3401–3408. https://doi.org/10.1200/jco.2014.60.7341 (Epub 2015/09/02. PubMed PMID: 26324363)
Tolcher AW, Khan K, Ong M, Banerji U, Papadimitrakopoulou V, Gandara DR et al (2015) Antitumor activity in RAS-driven tumors by blocking AKT and MEK. Clin Cancer Res 21(4):739–748. https://doi.org/10.1158/1078-0432.ccr-14-1901 (Epub 2014/12/18. PubMed PMID: 25516890; PubMed Central PMCID: PMC4335074)
Wei W, Mok SC, Oliva E, Kim SH, Mohapatra G, Birrer MJ (2013) FGF18 as a prognostic and therapeutic biomarker in ovarian cancer. J Clin Invest 123(10):4435–4448
Weinstein IB (2002) Cancer. Addiction to oncogenes—the Achilles heal of cancer. Science 297(5578):63–64. https://doi.org/10.1126/science.1073096 (Epub 2002/07/06. PubMed PMID: 12098689)
Yamagami W, Nagase S, Takahashi F, Ino K, Hachisuga T, Aoki D et al (2017) Clinical statistics of gynecologic cancers in Japan. J Gynecol Oncol 28(2):e32. https://doi.org/10.3802/jgo.2017.28.e32 (Epub 2017/02/16. PubMed PMID: 28198168; PubMed Central PMCID: PMC5323288)
Zaid TM, Yeung TL, Thompson MS, Leung CS, Harding T, Co NN et al (2013) Identification of FGFR4 as a potential therapeutic target for advanced-stage, high-grade serous ovarian cancer. Clin Cancer Res 19(4):809–820. https://doi.org/10.1158/1078-0432.ccr-12-2736 (Epub 2013/01/25. PubMed PMID: 23344261; PubMed Central PMCID: PMC3577959)
Funding
No specific funding was received.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Sabine Heublein received research support from Novartis, Ferring, Apceth and Addex; Honoria from Roche, travel expenses from Astra Zeneca. Frederik Marmé received honoraria from Roche, AstraZeneca, Pfizer, Clovis, Tesaro, Amgen, Celgene, Eisai, MSD, Genomic Health, Curevac; Travel expenses from Roche, Pfizer, Astra Zeneca, Pharma-Mar, Amgen, Celgene; Advisory Boards for Roche, Astra Zeneca, Pfizer, MSD, Amgen, Celgene, Genomic Health, Curevac. Stefan Kommoss and Michael S. Anglesio declare no competing interests.
Ethics approval
Collection of patient specimens and data from the British Columbia cohort was done under approved research protocols reviewed by the British Columbia Cancer Agency and University of British Columbia research ethics board (H05-60119).
Informed consent
No written informed consent of the participants is needed given the circumstances described above.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Frederik Marmé and Stefan Kommoss share senior authorship.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Heublein, S., Anglesio, M.S., Marmé, F. et al. Fibroblast growth factor receptor 4 (FGFR4) as detected by immunohistochemistry is associated with postoperative residual disease in ovarian cancer. J Cancer Res Clin Oncol 145, 2251–2259 (2019). https://doi.org/10.1007/s00432-019-02986-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-019-02986-0