Abstract
Purpose
Radiofrequency ablation (RFA) therapy has proven to be effective and feasible for early-stage hepatocellular carcinoma (HCC); however, rapid progression of residual tumor cells after RFA has been confirmed, but the molecular mechanisms of this phenomenon are poorly understood. This study evaluated the effect of the lipid raft proteins known as flotillins on the invasive and metastatic potential of residual HCC.
Methods
The human HCC cell line HCCLM3 was used to establish insufficient RFA models in vivo and in vitro. Changes in cellular morphology, soft agar colony formation, motility, metastasis, and epithelial–mesenchymal transition (EMT) markers after insufficient RFA intervention in vitro and in vivo were detected by real-time PCR, western blotting, immunohistochemistry and transwell assays.
Results
The results showed that flotillin-1 and flotillin-2 expression were upregulated in HCCLM3 cells following 45 °C heat treatment and in residual HCCLM3 xenografts cells after insufficient RFA. Knocking down flotillin-1 or flotillin-2 in HCCLM3 cells by shRNA significantly lowered insufficient RFA-induced tumor growth, EMT changes, and metastasis in vitro and in vivo. Furthermore, mechanism studies indicated that flotillins altered the EMT status and metastatic potential of heat-treated HCCLM3 cells by activating the Akt/Wnt/β-catenin signaling pathway.
Conclusions
Our findings present new evidence that flotillins play a key role in the aggressive behaviors of residual cancer cells after insufficient RFA and provide new insights into the regulatory mechanism of Wnt/β-catenin signaling.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC) is one of the most common malignancies and the third leading cause of cancer-related deaths worldwide (Siegel et al. 2015). Although a variety of therapies such as surgical resection, liver transplantation, interventional treatment, and systemic therapies are employed, HCC patients have low 5-year survival rates and high recurrence rates due to invasion and metastasis (Au and Frenette 2015; Forner et al. 2012; Schlachterman et al. 2015). A better understanding of the molecular mechanisms that regulate HCC invasion and metastasis is essential for developing novel prognostic and of therapeutic strategies for HCC.
Radiofrequency ablation (RFA) is a local therapy that is an alternative to surgery for HCC < 3 cm and is widely used due to its simplicity, safety, minimal invasiveness, repeatability, and short hospitalization (Lee et al. 2017; Tsukamoto et al. 2018). However, cases of rapidly progressing residual HCC after insufficient RFA have been reported to be on the rise, with post-RFA recurrent rates ranging from 3.2 to 26% (Lam et al. 2008; Shiina et al. 2012). Moreover, local recurrences after RFA show more invasive growth and vascular invasion, and less differentiation compared with tumors from patients without RFA. Until now, the underlying mechanisms behind this phenomenon were still less unknown.
Lipid rafts function as physical platforms for various molecules that are involved in a variety of biological processes by serving as organizing centers for assembling signaling molecules into functional complexes (Babuke and Tikkanen 2007). Flotillins are key components of lipid rafts and include two members: flotillin-1 (FLOT1) and flotillin-2 (FLOT2). Previous studies have shown that flotillins are closely associated with tumor development, invasion, and metastasis (Bodin et al. 2014; Deng et al. 2018; Liu et al. 2018). Moreover, FLOT1 and FLOT2 were recently reported to be overexpressed and associated with progression and poor survival in HCC, suggesting the possibility of their use as prognostic markers or therapeutic targets in HCC (Wang et al. 2017; Zhang et al. 2013). Nevertheless, the biological role and molecular mechanism through which flotillins alter the biology of residual HCC after insufficient RFA remains unclear.
In this study, we investigated FLOT1 and FLOT2 expression in insufficient RFA tumor tissues in vivo and in heat-treated HCC cells in vitro, and further explored the roles of flotillins in altering the metastatic potential of residual HCC cells.
Materials and methods
Cell culture
The human HCC cell lines HCCLM3 (a highly metastatic HCC line established at the Liver Cancer Institute, Zhongshan Hospital, Fudan University, Shanghai, China) and HepG2 cells (a modestly metastatic HCC line obtained from the Cell Bank of the Chinese Academy of Sciences, Shanghai, China) were maintained in Dulbecco’s modified Eagle’s medium (DMEM; Gibco BRL, Rockville, MD, USA) with 10% fetal bovine serum (FBS; Life Technologies, Carlsbad, CA, USA) and 100 U/mL penicillin at 37 °C in a humidified atmosphere containing 5% CO2 (Li et al. 2004).
Vectors, retroviral infections, and transfection
The lentiviral vectors pGCSIL-GFP-shRNA-FLOT1, pGCSIL-GFP-shRNA-FLOT2 and pGC-FU-GFP-CTNNB1 were purchased from GeneChem Co., Ltd. (Shanghai, China). pGC-FU-GFP-CTNNB1 was transfected into HCCLM3 cells to overexpress β-catenin, and pGCSIL-GFP-shRNA-FLOT1 and pGCSIL-GFP-shRNA-FLOT2 were transfected into HCCLM3 cells to silence FLOT1 and FLOT2, respectively. pGCSIL-GFP and pGC-FU-GFP were used as controls.
Soft agar colony formation assay
Soft agar colony formation was performed using the CytoSelect 96-Well Cell Transformation Assay (Cell Biolabs Inc., San Diego, CA, USA) according the manufacturer’s protocol. Briefly, a bottom layer of 1.2% agar solution was plated and solidified, and then a top layer of equal volumes 1.2% agar solution, culture medium, and cell suspension (1 × 103 cells/well) was added. Culture medium (100 µL) was added to the top layer of the soft agar and replaced with fresh medium every 3 d. The cells were incubated for 6–8 days at 37 °C. Numbers of microscopically visible colonies (> 0.1 mm) were counted.
In vitro heat treatment
HCCLM3 and HepG2 cells were seeded into 6-well plates at 5 × 104 cells/well. After 24 h, the plates were sealed with Parafilm and submerged in a water bath set to the target temperature for 10 min. The target temperatures for HCCLM3 and HepG2 cells were 39 °C, 42 °C, 45 °C and 41 °C, 44 °C, 47 °C, respectively; the control temperature was 37 °C. The soft agar colony formation, and cell migration and invasion assays were performed 48 h after heat treatments.
Cell migration and invasion assays
Cell migration and invasion were assessed by transwell assays (Corning Inc., Corning, NY, USA). Briefly, 8 × 104 cells in serum-free DMEM were seeded into the upper chamber of each well of 24-well plates containing 8.0-µm pore size membranes. DMEM containing 10% FBS was added to the lower chamber of each well. After 48 h, cells that had reached the underside of the membrane were stained with Giemsa (Sigma-Aldrich, St. Louis, MO, USA), and then five randomly selected areas (200× magnification) per well were counted. Cell invasion assays were performed similarly, except 80 µL of Matrigel (0.8 mg/mL, BD Biosciences, Franklin Lakes, NJ, USA) was added to each well 6 h before cells were seeded onto the membranes.
RNA extraction and real-time quantitative (RT-q)PCR
Total RNA was isolated using TRIzol reagent (Invitrogen, Carlsbad, CA, USA) and reverse transcribed to cDNA using the PrimeScript RT reagent kit (Takara, Shiga, Japan). SYBR Premix Ex Taq (Takara) was used for RT-qPCR according to the manufacturer’s instructions (Livak and Schmittgen 2001). Relative mRNA expression levels were calculated by the 2− ΔΔCt method after normalizing to β-actin as an internal control. (16) Sequences of the RT-qPCR primers were as follows: FLOT1, forward: 5′-CCCATCTCAGTCACTGGCATT-3′ and reverse: 5′-CCGCCAACATCTCCTTGTTC-3′; FLOT2, forward: 5′-CCCCAGATTGCTGCCAAA-3′ and reverse: 5′-TCCACTGAGGACCACAATCTCA-3′; E-cadherin, forward: 5′-CCCGGGACAACGTTTATTACTATG-3′ and reverse: 5′-TCAGCCGCTTTCAGATTTTCA-3′; N-cadherin, forward: 5′-CACGCCGAGCCCCAGTAT-3′ and reverse: 5′-GCCCCCAGTCGTTCAGGTAA-3′; Vimentin, forward: 5′-CTCTCAAAGATGCCCAGGAG-3′ and reverse: 5′-GCACGATCCAACTCTTCCTC-3′; Snail, forward: 5′-TGCAGGACTCTAATCCAAGTTTACC-3′ and reverse: 5′-GTGGGATGGCTGCCAGC-3′; β-actin, forward: 5′-GCTCCTCCTGAGCGCAAG-3′ and reverse: 5′-CATCTGCTGGAAGGTGGACA-3′.
Western blotting
Western blotting was performed as in previous studies using anti-FLOT1, anti-FLOT2 (Sigma-Aldrich), anti-E-cadherin, anti-N-cadherin, anti-Vimentin, anti-Snail, anti-β-catenin and anti-Cyclin-D1 antibodies (Abcam, Cambridge, UK) (Zhang et al. 2014a). Membranes were stripped and re-probed with anti-β-actin (Sigma-Aldrich) as a loading control.
Xenograft mouse model
Male BALB/c nude mice (5-week-old, 18–20 g) were purchased from SLAC Laboratory Animal Co., Ltd. (Shanghai, China) and housed under specific pathogen-free conditions. The experimental protocol was approved by the Ethical Committee on Animal Experiments of the Animal Care Committee of Fudan University. Xenograft HCC models were established by orthotopic implantation of histologically intact tumor tissue into the nude mouse liver according to previous protocols (Sun et al. 1996). Two weeks after orthotopic implantation, the insufficient RFA operation was performed as follows: insufficient RFA mice underwent laparotomy to expose the tumor, then, considering the weight and volume of nude mice, RFA was performed at a lower energy protocol, in which the out power was 5 W and the duration was 30 s. This ensured the presence of residual cancer. Mice in the control group were sham-operated by inserting a needle electrode into the tumor without performing ablation. 5 weeks later, all mice were euthanized and tumor volumes were measured. Tumor volume was calculated according to the formula: volume (mm3) = (largest diameter × shortest diameter2)/2 (Zhang et al. 2014a).Then, tumors and lungs were placed in a 4% paraformaldehyde solution. Lung tissues were serially sectioned and stained with hematoxylin and eosin.
Immunohistochemistry
Tumor tissues were fixed, embedded, and sliced into 5-µm thick sections. FLOT1, FLOT2, E-cadherin, N-cadherin, Vimentin and β-catenin immunohistochemical staining were performed as previously described. Staining results were viewed under a light microscope (Olympus, Tokyo, Japan) (Zhang et al. 2014a). Stained slides were scored by two investigators according to values obtained from IRS systems.
Statistical analysis
Experimental data are presented as mean ± SD. First, the Kolmogorov–Smirnov test was used to determine the normality of the data in each group. Differences between two normal distribution groups were estimated using Student’s t test. Differences among three or more normal distribution groups were analyzed using ANOVA. Differences between non-normal distribution groups were analyzed using nonparametric analyses of Chi-square tests. All analyses were performed using SPSS v20.0 software (IBM. Armonk, NY, USA); a two-tailed P value < 0.05 was considered statistically significant.
Results
Insufficient RFA increased FLOT1 and FLOT2 expression in HCCLM3 cells in vivo and in vitro
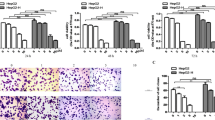
To examine the effects of insufficient RFA on FLOT1 and FLOT2 expression in vitro, expression was determined in HCC cells 24 h after 10 min heat intervention. Western blot results showed that FLOT1 and FLOT2 were significantly upregulated in heat-treated cells compared with the control group, especially at 45 °C for HCCLM3 cells. However, FLOT1 and FLOT2 expression were similar in heat-treated and control HepG2 cells (Fig. 1). Consistent with these in vitro results, western blot (Fig. 2a), RT-qPCR (Fig. 2b) and immunohistochemistry (Fig. 2c, d) results demonstrated that both FLOT1 and FLOT2 were significantly upregulated in insufficient RFA-treated HCCLM3 tumors compared with controls.
Insufficient RFA increased FLOT1 and FLOT2 levels in HCC cells in vitro. a Western blot analysis of FLOT1 and FLOT2 levels in HCC cells. HepG2 cells were lysed 24 h after 10-min heat treatments at 41 °C, 44 °C, and 47 °C, HCCLM3 cells were lysed 24 h after 10-min heat treatments at 39 °C, 42 °C, and 45 °C. b Densitometry analyses depict relative changes in FLOT1 and FLOT2 expression. Data are presented as mean ± SD. Experiments were independently conducted three times; *P < 0.05 and **P < 0.01
Insufficient RFA increased FLOT1 and FLOT2 levels in residual cancer in vivo. a, b FLOT1 and FLOT2 mRNA and protein levels in heat-treated HCCLM3 and HepG2 residual cancer and sham-operated tissue were detected by western blot and RT-qPCR. c, d Immunohistochemical staining confirmed increased FLOT1 and FLOT2 protein levels in heat-treated HCCLM3 residual cancer, but not in heat-treated HepG2 residual cancer. Representative images are shown at 200× magnification. Data are presented as mean ± SD. Experiments were independently conducted three times; *P < 0.05 and **P < 0.01
Silencing FLOT1 and FLOT2 decreased the aggressiveness of heat-treated HCCLM3 cells in vitro
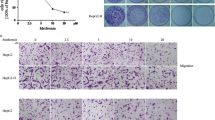
We further investigated whether FLOT1 and FLOT2 played a role in the increased metastatic capacity of HCCLM3 cells after insufficient RFA. When HCCLM3 cells were examined 24 h after 45 °C heat treatment for 10 min, soft agar colony formation assays showed that heat-treated HCCLM3 cells displayed higher anchorage-independent growth compared with control cells (Fig. 3a, d). Moreover, transwell migration (Fig. 3b, e) and matrigel invasion (Fig. 3c, f) assays revealed that heat treatment significantly increased the mobility and invasive capacity of HCCLM3 cells. Strikingly, silencing FLOT1, FLOT2, or with shRNA significantly reduced anchorage-independent growth, mobility, and invasion (Fig. 3a–f). Furthermore, the silencing FLOT1 or FLOT2 or both also reduced the anchorage-independent growth, mobility and invasive capacity of HCCLM3 cultured in 37 °C (Supplemental Fig. 1). Collectively, the above findings demonstrated that up-regulated expression of FLOT1 and FLOT2 was functionally relevant to invasion and metastasis of HCCLM3 cells mediated by heat intervention. Interestingly, the expression of FLOT1 and FLOT2 also differs in clinical specimens with different metastatic potential (Supplemental Fig. 2).
Silencing FLOT1 and FLOT2 decreased the in vitro aggressiveness of heat-treated HCCLM3 cells. a, d Representative images (a) and quantification (d) of HCCLM3 colony numbers from anchorage-independent growth assays. Colonies > 0.1 mm in diameter were scored. b, e Representative images (b) and quantification (e) of cell migration results of transwell assays; scale bar = 50 µm. c, f Representative images (c) and quantification (f) cell invasion results of transwell matrix penetration assays; scale bar = 50 µm. Data are presented as mean ± SD. Experiments were independently conducted three times; *P < 0.05, **P < 0.01 vs. the HCCLM3-WT group; #P < 0.05, ##P < 0.01 vs. the heat-treated HCCLM3-shVector group
Silencing FLOT1 and FLOT2 attenuated epithelial–mesenchymal transition (EMT)-like changes in heat-treated HCCLM3 cells in vitro
We further elucidate whether the upregulation of FLOT1 and FLOT2 was functionally associated with the EMT-like changes in heat-treated HCCLM3 cells. At 48 h after 45 °C heat treatment, HCCLM3 cells showed an irregular fibroblast-like shape instead of their typical epithelial/cobblestone appearance (Fig. 4a). Meanwhile, RT-qPCR (Fig. 4b) and western blot (Fig. 4c) results demonstrated that E-cadherin expression (epithelial marker) was reduced in heat-treated cells compared with controls, while N-cadherin, Vimentin, and Snail were all increased. Silencing FLOT1 and FLOT2 influenced cell morphology and the expression of EMT markers. RT-PCR and western blot data showed that N-cadherin, Vimentin, and Snail were decreased in heat-treated HCCLM3-shFLOT1-, HCCLM3-shFLOT2- and HCCLM3-shFLOT1 + 2-expressing cells compared with heat-treated HCCLM3-shVector cells (Fig. 4a–c). Furthermore, the silencing FLOT1 or FLOT2 or both also reduced the expression of N-cadherin, vimentin and Snail in HCCLM3 cultured in 37 °C (Supplemental Fig. 3).
Silencing FLOT1 and FLOT2 attenuated EMT-like changes in heat-treated HCCLM3 cells in vitro. a Morphologic changes observed in untreated and heat-treated HCCLM3 cells that were transfected with shVector, shFLOT1, shFLOT2, or shFLOT1 + 2. b, c RT-qPCR and western blot analyses of FLOT1, FLOT2, E-cadherin, N-cadherin, Vimentin, and Snail expression in control and heat-treated HCCLM3 cells that were transfected with shVector, shFLOT1, shFLOT2, or shFLOT1 + 2; scale bar = 50 µm. Data are presented as mean ± SD. Experiments were independently conducted three times; *P < 0.05, **P < 0.01 vs. the HCCLM3-WT group; #P < 0.05, ##P < 0.01 vs. the heat-treated HCCLM3-shVector group
The Akt/Wnt/β-catenin pathway plays a vital role in the change of metastatic potential of heat-treated HCCLM3 cells in vitro
We previously found that insufficient RFA enhanced the invasive and metastatic potential of residual HCCLM3 cells by activating β-catenin signaling (Zhang et al. 2014a). Therefore, in this study, we investigated whether β-catenin signaling is required for the FLOT1- and FLOT2-mediated increase in HCCLM3 aggressiveness. As shown in Fig. 5a, c, levels of phosphorylated (p)Akt, β-catenin and Cyclin-D1 were increased in heat-treated HCCLM3-shVector cells. Additionally, a marked translocation of intracellular translocation β-catenin in heat-treated HCCLM3-shVector cells was verified by western blot (Fig. 5b, c). In contrast, the phosphorylation level of AKT and the expression of β-catenin and Cyclin-D1 were decreased in the HCCLM3-shFLOT1, HCCLM3-shFLOT2 as well as HCCLM3-shFLOT1 + 2 cells (Fig. 5a–c). Similarly, the silencing of FLOT1 or FLOT2 or both reduced the expression of phosphorylation level of AKT, cytoplasmic and nuclear β-catenin in HCCLM3 cultured in 37 °C (Supplemental Fig. 4). Moreover, overexpressing β-catenin restored the increased migration and invasion following heat treatment of HCCLM3-shFLOT1 and HCCLM3-shFLOT2 cells (Fig. 5d). Taken together, these results demonstrate that AKT/β-catenin/Cyclin D1 signaling is essential for FLOT1 and FLOT2-induced migration and invasion in heat-treated HCCLM3 cells.
The Akt/Wnt/β-catenin pathway plays a vital role in the increased metastatic potential of residual HCCLM3 cells after insufficient RFA. a Western blot analysis of phosphorylated (p)Akt, and total β-catenin and Cyclin-D1 in HCCLM3 cells that were transfected with shVector, shFLOT1, shFLOT2, or shFLOT1 + 2. b Nuclear distribution of β-catenin in HCCLM3 cells was detected by western blot. c Densitometry showing relative changes in FLOT1 and FLOT2 expression. d Representative images of migration and invasion assays of HCCLM3 cells transfected with shVector, shFLOT1, and shFLOT2 ± CTNNB1 were analyzed using transwell assays; scale bar = 100 µm. Data are presented as mean ± SD. Experiments were independently conducted three times; *P < 0.05, **P < 0.01 vs. heat-untreated HCCLM3-WT group; #P < 0.05, ##P < 0.01 vs. heat-treated HCCLM3-shVector group
Silencing FLOT1 and FLOT2 inhibited tumor growth and EMT of HCCLM3 cells in vivo
Finally, we determined whether FLOT1 and FLOT2 influenced the behavior of residual tumor cells after insufficient RFA intervention in vivo. In insufficient RFA group, the tumor volume of HCCLM3-shVector was significantly larger than those in the control group (1476.7 ± 95.1 vs. 1827.2 ± 161.7 mm3, P < 0.05; Fig. 6a). Either HCCLM3-shFLOT1 or HCCLM3-shFLOT2 tumors were smaller than HCCLM3-shVector tumors in the control mice (575.7 ± 90.3 vs. 1476.7 ± 95.1 mm3, P < 0.01; 650.1 ± 91.9 vs. 1476.7 ± 95.1 mm3, P < 0.01) or insufficient RFA-treated mice (680.7 ± 97.8 vs. 1827.2 ± 161.7 mm3, P < 0.01; 769.3 ± 98.49 vs. 1827.2 ± 161.7 mm3, P < 0.01; Fig. 6a). To further evaluate the metastatic potential of residual cancer of HCCLM3 after insufficient RFA, serial lung paraffin sections were used. The pulmonary metastases in insufficient RFA group were significantly increased, compared with the control group; while FLOT1 or FLOT2 knockout can inhibit the pulmonary metastases in the control or insufficient RFA-treated mice (Fig. 6b). We further examined E-cadherin, N-cadherin, Vimentin, and β-catenin expression in orthotopic tumor samples after insufficient RFA. Immunohistochemistry revealed typical membranous E-cadherin expression at cell–cell contacts in the HCCLM3-shFLOT1 and HCCLM3-shFLOT2 groups, and low N-cadherin, Vimentin and β-catenin expression. In contrast, HCCLM3-shVector tumors showed significantly lower E-cadherin expression, and higher N-cadherin, Vimentin, and β-catenin expression (Fig. 6c). These results demonstrated that FLOT1 and FLOT2 promoted residual HCC tumor growth and metastasis after insufficient RFA in vivo.
Silencing FLOT1 and FLOT2 inhibited growth and EMT of HCCLM3 cells in vivo. a Tumor sizes in the HCCLM3-LV-shFLOT1 and HCCLM3-shFLO2 groups were smaller than in the HCCLM3-WT group. b Silencing FLOT1 and FLOT2 inhibit the lung metastasis of HCCLM3 residual cancer after insufficient RFA. c Immunohistochemistry revealed that E-cadherin expression was decreased, while N-cadherin, Vimentin and β-catenin expression were increased in HCCLM3-shFLOT1 and HCCLM3-shFLOT2 tumors compared with HCCLM3-controls. Representative images are shown at 200× magnification. Data are presented as mean ± SD. Experiments were independently conducted three times; *P < 0.05, **P < 0.01 vs. the HCCLM3-WT group; #P < 0.05, ##P < 0.01 vs. heat-treated HCCLM3-shVector group
Discussion
In this study, we first found that FLOT1 and FLOT2 were overexpressed in HCC tissues after insufficient RFA and in HCC cell lines after heat treatment. Next, we explored the role of FLOT1 and FLOT2 in HCC growth and metastasis in vivo and in vitro. In accordance with studies in other cancers (Berger et al. 2013; Song et al. 2012; Wang et al. 2013), knocking down FLOT1 and/or FLOT2 inhibited the invasion, migration, and anchorage-independent growth of HCCLM3 cells in vitro and decreased their tumor growth and metastasis in vivo.
Flotillins encode caveolae-associated integral membrane proteins that belong to the lipid raft family, and are involved in vesicular trafficking and signal transduction(Bickel et al. 1997). The roles of flotillins in cancer progression have been studied in various cancers (Gao et al. 2015; Li et al. 2014; Zhang et al. 2014b). Flotillin overexpression promotes the proliferation, invasion, migration, and metastasis of cancer cells. Our previous studies had demonstrated that thermal intervention can enhance the metastasis of residual cancer by activating the FAK/PI3k signaling pathway (Zhang et al. 2017). A recent research pointed out that overexpression of flotillin can interact with α-actin and affect the activity of FAK, thereby enhancing the migration and metastasis of cancer(Banning et al. 2018). Whether thermal interference can affect the expression of flotillin is worth exploring. The present study confirmed for the first time that the expression of flotillin was up-regulated in residual cancer tissues after thermal interference.
In this study, we found that insufficient RFA increased both FLOT1 and FLOT2 in HCCLM3 cells but not in HepG2 cells, indicating these human HCC cell lines have different biological characteristics. Inversely, knocking down either FLOT1 or FLOT2 almost completely blocked the enhanced aggressiveness of heat-treated HCCLM3 cells in vitro and of residual cancer after insufficient RFA in vivo. These findings indicated a prominent role for flotillins in insufficient RFA-induced metastasis.
EMT is a biological process through which polarized epithelial cells undergo phenotypic changes into mesenchymal cells with enhanced invasiveness, survival, and extracellular matrix production(Kalluri and Weinberg 2009). Furthermore, EMT contributes to metastasis in a variety of human cancers (Hugo et al. 2007; Yeung and Yang 2017). Several studies have demonstrated that suboptimal RFA accelerated HCC growth and spread by transiently inducing an EMT-like and more aggressive cellular phenotype (Iwahashi et al. 2016; Yoshida et al. 2013). Meanwhile, the EMT-related transcript factors snail and twist were significantly higher in RFA group than those in the non-RFA group in clinical HCC cases (Iwahashi et al. 2016; Zeng et al. 2018). Herein, we showed that silencing FLOT1 and FLOT2 increased E-cadherin expression and decreased N-cadherin, Vimentin and Snail in heat-treated HCCLM3 cells. Finally, these changes were also found in xenografts in nude mice after insufficient RFA. Thus, this study provides new insights into the mechanisms through which EMT is regulated in residual HCC after insufficient RFA and suggests FLOT1 and FLOT2 as potential targets for anti-metastatic therapy.
Multiple signaling pathways regulate EMT, but considering the central role of Wnt/β-catenin in HCC biology and the well-characterized association between Wnt/β-catenin signaling and EMT, we focused on the Wnt/β-catenin pathway (Dahmani et al. 2011; Monga 2015; Waisberg and Saba 2015). In this study, the effects of flotillins on β-catenin expression were examined, and elevated total and intra-nuclear β-catenin levels were verified in HCCLM3 cells in vitro. The key downstream gene of Wnt/β-catenin signaling, Cyclin-D1, was also upregulated. Moreover, we also detected pAKT levels because AKT phosphorylation can enhance β-catenin nuclear accumulation (Fang et al. 2007). Moreover, AKT signaling pathway is closely related to EMT in HCC, colorectal cancer, gastric cancer and breast cancer (Duan et al. 2018; Huang et al. 2018; Jiang et al. 2019; Zhang et al. 2018). Li et al. found that up- and downregulation of FLOT1 remarkably affected cervical cancer cell motility and invasion, respectively, through the EMT (via Wnt/β-catenin) and NF-κB pathways (Li et al. 2016). Liu et al. showed that FLOT2 promoted metastasis in nasopharyngeal carcinoma by activating the NF-κB and PI3K/Akt3 pathways (Liu et al. 2015). In bladder cancer, up-regulation of FLOT1 could also reverse the suppressed cell proliferation caused by miR-608 via activating AKT signaling (Liang et al. 2017). Additionally, as a direct target of miR-133, FLOT2 was regulated via Akt signaling and played pro-metastatic role in lung adenocarcinoma cell (Wei et al. 2018). Our results showed that pAKT was increased by heat treatment and decreased by silencing FLOT1 and/or FLOT2. Conversely, overexpressing β-catenin rescued the increase in motility and invasion of HCCLM3 cells following heat treatment that was lost when FLOT1 or FLOT2 were knocked out. Our findings demonstrate that insufficient RFA enhances the invasion and metastasis of residual cancer cells via Akt/Wnt/β-catenin-regulated EMT.
In summary, this study demonstrated that insufficient RFA enhanced the metastatic ability of HCC cells in vivo and in vitro, which was mediated by Akt/Wnt/β-catenin signaling. Furthermore, insufficient RFA increased FLOT1 and FLOT2 expression, suggesting these lipid raft protein promote these signaling pathways. Therefore, future studies will attempt to decrease the invasion and metastasis of residual cancer cells to improve the curative effects of RFA in HCC patients.
References
Au JS, Frenette CT (2015) Management of hepatocellular carcinoma: current status and future directions. Gut Liver 9:437–448. https://doi.org/10.5009/gnl15022
Babuke T, Tikkanen R (2007) Dissecting the molecular function of reggie/flotillin proteins. Eur J Cell Biol 86:525–532. https://doi.org/10.1016/j.ejcb.2007.03.003
Banning A, Babuke T, Kurrle N, Meister M, Ruonala MO, Tikkanen R (2018) Flotillins regulate focal adhesions by interacting with alpha-actinin and by influencing the activation of focal. Adhes Kinase Cells. https://doi.org/10.3390/cells7040028
Berger T et al (2013) Flotillin-2 deficiency leads to reduced lung metastases in a mouse breast cancer model. Oncogene 32:4989–4994. https://doi.org/10.1038/onc.2012.499
Bickel PE, Scherer PE, Schnitzer JE, Oh P, Lisanti MP, Lodish HF (1997) Flotillin and epidermal surface antigen define a new family of caveolae-associated integral membrane proteins. J Biol Chem 272:13793–13802
Bodin S, Planchon D, Rios Morris E, Comunale F, Gauthier-Rouviere C (2014) Flotillins in intercellular adhesion—from cellular physiology to human diseases. J Cell Sci 127:5139–5147. https://doi.org/10.1242/jcs.159764
Dahmani R, Just PA, Perret C (2011) The Wnt/beta-catenin pathway as a therapeutic target in human hepatocellular carcinoma. Clin Res Hepatol Gastroenterol 35:709–713. https://doi.org/10.1016/j.clinre.2011.05.010
Deng Y et al (2018) Prognostic value of flotillins (flotillin-1 and flotillin-2) in human cancers: a meta-analysis. Clin Chim Acta 481:90–98. https://doi.org/10.1016/j.cca.2018.02.036
Duan S et al (2018) IMPDH2 promotes colorectal cancer progression through activation of the PI3K/AKT/mTOR and PI3K/AKT/FOXO1 signaling pathways. J Exp Clin Cancer Res 37:304. https://doi.org/10.1186/s13046-018-0980-3
Fang D et al (2007) Phosphorylation of beta-catenin by AKT promotes beta-catenin transcriptional activity. J Biol Chem 282:11221–11229. https://doi.org/10.1074/jbc.M611871200
Forner A, Llovet JM, Bruix J (2012) Hepatocellular carcinoma. Lancet 379:1245–1255. https://doi.org/10.1016/S0140-6736(11)61347-0
Gao W et al (2015) Plasma membrane proteomic analysis of human gastric cancer tissues: revealing flotillin 1 as a marker for gastric cancer. BMC Cancer 15:367. https://doi.org/10.1186/s12885-015-1343-5
Huang Y, Du J, Mi Y, Li T, Gong Y, Ouyang H, Hou Y (2018) Long non-coding RNAs contribute to the inhibition of proliferation and EMT by pterostilbene in human breast. Cancer Front Oncol 8:629. https://doi.org/10.3389/fonc.2018.00629
Hugo H, Ackland ML, Blick T, Lawrence MG, Clements JA, Williams ED, Thompson EW (2007) Epithelial–mesenchymal and mesenchymal–epithelial transitions in carcinoma progression. J Cell Physiol 213:374–383. https://doi.org/10.1002/jcp.21223
Iwahashi S et al (2016) Epithelial–mesenchymal transition-related genes are linked to aggressive local recurrence of hepatocellular carcinoma after radiofrequency ablation. Cancer Lett 375:47–50. https://doi.org/10.1016/j.canlet.2016.02.041
Jiang M et al (2019) miR-1254 inhibits cell proliferation, migration, and invasion by down-regulating Smurf1 in gastric cancer. Cell Death Dis 10:32. https://doi.org/10.1038/s41419-018-1262-x
Kalluri R, Weinberg RA (2009) The basics of epithelial-mesenchymal transition. J Clin Invest 119:1420–1428. https://doi.org/10.1172/JCI39104
Lam VW et al (2008) Risk factors and prognostic factors of local recurrence after radiofrequency ablation of hepatocellular carcinoma. J Am Coll Surg 207:20–29. https://doi.org/10.1016/j.jamcollsurg.2008.01.020
Lee MW et al (2017) Radiofrequency ablation of hepatocellular carcinoma as bridge therapy to liver transplantation: a 10-year intention-to-treat analysis. Hepatology 65:1979–1990. https://doi.org/10.1002/hep.29098
Li Y et al (2004) Stepwise metastatic human hepatocellular carcinoma cell model system with multiple metastatic potentials established through consecutive in vivo selection and studies on metastatic characteristics. J Cancer Res Clin Oncol 130:460–468. https://doi.org/10.1007/s00432-004-0564-9
Li H et al (2014) Prognostic significance of Flotillin1 expression in clinically N0 tongue squamous cell cancer. Int J Clin Exp Pathol 7:996–1003
Li Z et al (2016) Elevated expression of flotillin-1 is associated with lymph node metastasis and poor prognosis in early-stage cervical cancer. Am J Cancer Res 6:38–50
Liang Z et al (2017) MicroRNA-608 inhibits proliferation of bladder cancer via AKT/FOXO3a signaling pathway. Mol Cancer 16:96. https://doi.org/10.1186/s12943-017-0664-1
Liu J et al (2015) Flotillin-2 promotes metastasis of nasopharyngeal carcinoma by activating NF-kappaB and PI3K/Akt3 signaling pathways. Sci Rep 5:11614. https://doi.org/10.1038/srep11614
Liu XX et al (2018) Roles of flotillins in tumors. J Zhejiang Univ Sci B 19:171–182. https://doi.org/10.1631/jzus.B1700102
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)). Method Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Monga SP (2015) beta-Catenin signaling and roles in liver homeostasis injury tumorigenesis. Gastroenterology 148:1294–1310. https://doi.org/10.1053/j.gastro.2015.02.056
Schlachterman A, Craft WW Jr, Hilgenfeldt E, Mitra A, Cabrera R (2015) Current and future treatments for hepatocellular carcinoma. World J Gastroenterol 21:8478–8491. https://doi.org/10.3748/wjg.v21.i28.8478
Shiina S et al (2012) Radiofrequency ablation for hepatocellular carcinoma: 10-year outcome and prognostic factors. Am J Gastroenterol 107:569–577. https://doi.org/10.1038/ajg.2011.425 (quiz 578)
Siegel RL, Miller KD, Jemal A (2015) Cancer statistics, 2015. CA Cancer J Clin 65:5–29. https://doi.org/10.3322/caac.21254
Song L et al (2012) Flotillin-1 promotes tumor necrosis factor-alpha receptor signaling and activation of NF-kappaB in esophageal squamous cell carcinoma cells. Gastroenterology 143:995–1005 e1012. https://doi.org/10.1053/j.gastro.2012.06.033
Sun FX, Tang ZY, Lui KD, Ye SL, Xue Q, Gao DM, Ma ZC (1996) Establishment of a metastatic model of human hepatocellular carcinoma in nude mice via orthotopic implantation of histologically intact tissues. Int J Cancer 66:239–243 https://doi.org/10.1002/(SICI)1097-0215(19960410)66:2%3C239::AID-IJC17%3E3.0.CO;2-7
Tsukamoto M et al (2018) Long-term favorable outcomes of radiofrequency ablation for hepatocellular carcinoma as an initial treatment: a single-center experience over a 10-year. Period Anticancer Res 38:1047–1052. https://doi.org/10.21873/anticanres.12321
Waisberg J, Saba GT (2015) Wnt-/-beta-catenin pathway signaling in human hepatocellular carcinoma. World J Hepatol 7:2631–2635. https://doi.org/10.4254/wjh.v7.i26.2631
Wang X, Yang Q, Guo L, Li XH, Zhao XH, Song LB, Lin HX (2013) Flotillin-2 is associated with breast cancer progression and poor survival outcomes. J Transl Med 11:190. https://doi.org/10.1186/1479-5876-11-190
Wang CH et al (2017) Flot2 promotes tumor growth and metastasis through modulating cell cycle and inducing epithelial-mesenchymal transition of hepatocellular carcinoma. Am J Cancer Res 7:1068–1083
Wei G, Xu Y, Peng T, Yan J (2018) miR-133 involves in lung adenocarcinoma cell metastasis by targeting FLOT2 Artif. Cells Nanomed Biotechnol 46:224–230. https://doi.org/10.1080/21691401.2017.1324467
Yeung KT, Yang J (2017) Epithelial–mesenchymal transition in tumor metastasis. Mol Oncol 11:28–39. https://doi.org/10.1002/1878-0261.12017
Yoshida S et al (2013) Sublethal heat treatment promotes epithelial-mesenchymal transition and enhances the malignant potential of hepatocellular carcinoma. Hepatology 58:1667–1680. https://doi.org/10.1002/hep.26526
Zeng J et al (2018) LncRNA FUNDC2P4 down-regulation promotes epithelial–mesenchymal transition by reducing E-cadherin expression in residual hepatocellular carcinoma after insufficient radiofrequency ablation. Int J Hyperthermia 34:802–811. https://doi.org/10.1080/02656736.2017.1422030
Zhang SH, Wang CJ, Shi L, Li XH, Zhou J, Song LB, Liao WT (2013) High expression of FLOT1 is associated with progression and poor prognosis in hepatocellular carcinoma. PLoS One 8:e64709. https://doi.org/10.1371/journal.pone.0064709
Zhang N et al (2014a) Incomplete radiofrequency ablation enhances invasiveness and metastasis of residual cancer of hepatocellular carcinoma cell HCCLM3 via activating beta-catenin signaling. PLoS One 9:e115949. https://doi.org/10.1371/journal.pone.0115949
Zhang Y, Li J, Song Y, Chen F, Pei Y, Yao F (2014b) Flotillin-1 expression in human clear-cell renal cell carcinoma is associated with cancer progression and poor patient survival. Mol Med Rep 10:860–866. https://doi.org/10.3892/mmr.2014.2310
Zhang N et al (2017) Insufficient radiofrequency ablation treated hepatocellular carcinoma cells promote metastasis by up-regulation ITGB3. J Cancer 8:3742–3754. https://doi.org/10.7150/jca.20816
Zhang ZQ et al (2018) FAM134B induces tumorigenesis and epithelial-to-mesenchymal transition via Akt signaling in hepatocellular carcinoma. Mol Oncol. https://doi.org/10.1002/1878-0261.12429
Acknowledgements
This study was supported by the National Natural Science Foundation of China (No. 81702371).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We have read and understood the Journal of Cancer Research and Clinical Oncology’s policy on disclosing conflicts of interest and declare that we have none.
Ethical statements
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. The article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Zhang, N., Li, H., Qin, C. et al. Insufficient radiofrequency ablation promotes the metastasis of residual hepatocellular carcinoma cells via upregulating flotillin proteins. J Cancer Res Clin Oncol 145, 895–907 (2019). https://doi.org/10.1007/s00432-019-02852-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-019-02852-z