Abstract
Purpose
Better understanding of the molecular mechanisms of metastasis—the major cause of death in osteosarcoma (OS)—is a key for the development of more effective metastasis-suppressive therapy. Here, we investigated the biological relevance of the CXCL12/CXCR4 axis in OS.
Methods
We interfered with CXCL12/CXCR4 signaling in CXCR4-expressing human 143-B OS cells through stable expression of CXCL12, of its competitive antagonist P2G, or of CXCL12-KDEL, designed to retain CXCR4 within the cell. Intratibial OS xenograft mouse model metastasizing to the lung was used to assess tumorigenic and metastatic potential of the manipulated cell lines.
Results
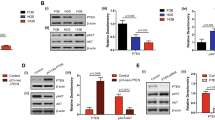
Constitutive expression of native CXCL12 promoted lung metastasis without affecting tumor growth. Stable expression of P2G or CXCL12-KDEL significantly accelerated tumor growth but diminished lung metastasis. Tumors grown from P2G- or CXCL12-KDEL-expressing cells contained higher levels of CXCR4-encoding mRNA going along with a higher percentage of CXCR4-expressing tumor cells. Lung metastases of all groups were predominantly enriched with CXCR4-expressing tumor cells.
Conclusion
Higher abundance of CXCR4 possibly contributed to increased local retention of tumor cells by bone marrow-derived CXCL12, reflected in the increased primary tumor growth and decreased number of lung metastases in P2G and CXCL12-KDEL groups. Higher percentage of CXCR4-expressing lung metastatic cells compared to the corresponding primary tumors point to important functions of the CXCL12/CXCR4 axis in late steps of metastasis. In conclusion, based on the here reported results, local treatment of lung metastases with novel CXCR4-targeting therapeutics might be considered and favored over anti-CXCR4 systemic therapy.




Similar content being viewed by others
References
Abi-Younes S, Sauty A et al (2000) The stromal cell-derived factor-1 chemokine is a potent platelet agonist highly expressed in atherosclerotic plaques. Circ Res 86(2):131–138
Arlt MJ, Banke IJ et al (2011) LacZ transgene expression in the subcutaneous Dunn/LM8 osteosarcoma mouse model allows for the identification of micrometastasis. J Orthop Res 29(6):938–946
Arlt MJ, Born W et al (2012) Improved visualization of lung metastases at single cell resolution in mice by combined in situ perfusion of lung tissue and X-Gal staining of lacZ-tagged tumor cells. Journal of visualized experiments JoVE(66):e4162
Bai S, Wang D et al (2011) Characterization of CXCR4 expression in chondrosarcoma of bone. Arch Pathol Lab Med 135(6):753–758
Baumhoer D, Smida J et al (2012) Strong expression of CXCL12 is associated with a favorable outcome in osteosarcoma. Modern Pathol 25(4):522–528
Berghuis D, Schilham MW et al (2012) The CXCR4–CXCL12 axis in Ewing sarcoma: promotion of tumor growth rather than metastatic disease. Clin Sarcoma Res 2(1):24
Berndt K, Campanile C et al (2013) Evaluation of quercetin as a potential drug in osteosarcoma treatment. Anticancer Res 33(4):1297–1306
Bertolini G, D’Amico L et al (2015) Microenvironment-modulated metastatic CD133+/CXCR4+/EpCAM-lung cancer-initiating cells sustain tumor dissemination and correlate with poor prognosis. Cancer Res 75(17):3636–3649
Brennecke P, Arlt MJ et al (2014) CXCR4 antibody treatment suppresses metastatic spread to the lung of intratibial human osteosarcoma xenografts in mice. Clin Exp Metastasis 31(3):339–349
Campanile C, Arlt MJ et al (2013) Characterization of different osteosarcoma phenotypes by PET imaging in preclinical animal models. J Nuclear Med 54(8):1362–1368
Chatterjee S, Behnam Azad B et al (2014) The intricate role of CXCR4 in cancer. Adv Cancer Res 124:31–82
de Nigris F, Rossiello R et al (2008) Deletion of Yin Yang 1 protein in osteosarcoma cells on cell invasion and CXCR4/angiogenesis and metastasis. Cancer Res 68(6):1797–1808
Kim SY, Lee CH et al (2008) Inhibition of the CXCR4/CXCL12 chemokine pathway reduces the development of murine pulmonary metastases. Clin Exp Metastasis 25(3):201–211
Klein MJ, Siegal GP (2006) Osteosarcoma: anatomic and histologic variants. Am J Clin Pathol 125(4):555–581
Li YJ, Dai YL et al (2015) Clinicopathological and prognostic significance of chemokine receptor CXCR4 in patients with bone and soft tissue sarcoma: a meta-analysis. Clin Exp Med. doi:10.1007/s10238-015-0405-y
Liao YX, Zhou CH et al (2013) The role of the CXCL12–CXCR4/CXCR7 axis in the progression and metastasis of bone sarcomas. Int J Mol Med 32(6):1239–1246
Lin F, Zheng SE et al (2011) Relationships between levels of CXCR4 and VEGF and blood-borne metastasis and survival in patients with osteosarcoma. Med Oncol 28(2):649–653
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) Method. Methods 25(4):402–408
Loetscher P, Gong JH et al (1998) N-terminal peptides of stromal cell-derived factor-1 with CXC chemokine receptor 4 agonist and antagonist activities. J Biol Chem 273(35):22279–22283
Ma Q, Zhou Y et al (2012) The clinical value of CXCR4, HER2 and CD44 in human osteosarcoma: a pilot study. Oncol Lett 3(4):797–801
Mialou V, Philip T et al (2005) Metastatic osteosarcoma at diagnosis: prognostic factors and long-term outcome—the French pediatric experience. Cancer 104(5):1100–1109
Miettinen M, Wang ZF et al (2011) ERG transcription factor as an immunohistochemical marker for vascular endothelial tumors and prostatic carcinoma. Am J Surg Pathol 35(3):432–441
Miura K, Uniyal S et al (2005) Chemokine receptor CXCR4-beta1 integrin axis mediates tumorigenesis of osteosarcoma HOS cells. Biochem Cell Biol 83(1):36–48
Muller A, Homey B et al (2001) Involvement of chemokine receptors in breast cancer metastasis. Nature 410(6824):50–56
Murakami T, Maki W et al (2002) Expression of CXC chemokine receptor-4 enhances the pulmonary metastatic potential of murine B16 melanoma cells. Cancer Res 62(24):7328–7334
Nagasawa T (2014) CXC chemokine ligand 12 (CXCL12) and its receptor CXCR4. J Mol Med (Berl) 92(5):433–439
Namlos HM, Kresse SH et al (2012) Global gene expression profiling of human osteosarcomas reveals metastasis-associated chemokine pattern. Sarcoma 2012:639038
Norton L, Massague J (2006) Is cancer a disease of self-seeding? Nat Med 12(8):875–878
Oda Y, Yamamoto H et al (2006) CXCR4 and VEGF expression in the primary site and the metastatic site of human osteosarcoma: analysis within a group of patients, all of whom developed lung metastasis. Modern Pathol 19(5):738–745
Onai N, Zhang Y et al (2000) Impairment of lymphopoiesis and myelopoiesis in mice reconstituted with bone marrow-hematopoietic progenitor cells expressing SDF-1-intrakine. Blood 96(6):2074–2080
Portella L, Vitale R et al (2013) Preclinical development of a novel class of CXCR4 antagonist impairing solid tumors growth and metastases. PLoS One 8(9):e74548
Sabile AA, Arlt MJ et al (2012) Cyr61 expression in osteosarcoma indicates poor prognosis and promotes intratibial growth and lung metastasis in mice. J Bone Miner Res 27(1):58–67
Tan Y, Li Y et al (2009) A novel CXCR4 antagonist derived from human SDF-1beta enhances angiogenesis in ischaemic mice. Cardiovasc Res 82(3):513–521
Tsuruo T, Fujita N (2008) Platelet aggregation in the formation of tumor metastasis. Proc Jpn Acad Ser B Phys Biol Sci 84(6):189–198
Valastyan S, Weinberg RA (2011) Tumor metastasis: molecular insights and evolving paradigms. Cell 147(2):275–292
Williams SA, Harata-Lee Y et al (2010) Multiple functions of CXCL12 in a syngeneic model of breast cancer. Mol Cancer 9:250
Zeelenberg IS, Ruuls-Van Stalle L et al (2003) The chemokine receptor CXCR4 is required for outgrowth of colon carcinoma micrometastases. Cancer Res 63(13):3833–3839
Acknowledgments
We thank Christopher Bühler for the excellent technical assistance and Dr. Malgorzata Kisielow (ETH Zurich Flow Cytometry Facility, Zurich, Switzerland) and Dr. David Jarrossay (Università Della Svizzera Italiana, Bellinzona, Switzerland) for their help with flow cytometry analysis. We thank Prof. Kitamura and Dr. Ueda for providing us with the CXCL12-KDEL construct.
Authors’ roles
ON, MJEA, MT, and AG designed the study; ON and PB conducted the study; ON, MJEA, AK, BR, and SB were involved in the animal work; MT contributed the material; ON, MJEA, MT, and WB analyzed and interpreted the data; ON, MJEA, PB, WB, MT, and BF prepared the manuscript.
Financial support
Our work is supported by the University of Zurich, the Schweizerischer Verein Balgrist (Zurich, Switzerland), the Walter L. & Johanna Wolf Foundation (Zurich, Switzerland), the Highly Specialized Medicine for Musculoskeletal Oncology program of the Canton of Zurich, the Zurcher Krebsliga (Zurich, Switzerland), the “Kind und Krebs” fund (Zollikerberg, Switzerland), and the Swiss National Science Foundation SNF Nr. 310030_149649.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors state that they have no conflicts of interest.
Ethical approval
Research involving animals: All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. Research Involving humans participants: This article does not contain any studies with human participants performed by any of the authors.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Neklyudova, O., Arlt, M.J.E., Brennecke, P. et al. Altered CXCL12 expression reveals a dual role of CXCR4 in osteosarcoma primary tumor growth and metastasis. J Cancer Res Clin Oncol 142, 1739–1750 (2016). https://doi.org/10.1007/s00432-016-2185-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-016-2185-5