Abstract
Purpose
Bacteriochlorin derivatives are promising photosensitive agents for photodynamic therapy (PDT) of tumors. In the current study, the photodynamic activity of a novel bacteriochlorin derivative, cis-2, 3, 12, 13-tetracarboxymethyl-5, 10, 15, 20-tetraphenyl bacteriochlorin (TCTB), was evaluated both in vitro and in vivo.
Methods
Physicochemical characteristics of the novel photosensitizer were measured. The efficiency of TCTB-PDT in vitro was analyzed by MTT assay, clonogenic assay and in situ trypan blue exclusion test. The intracellular distribution of photosensitizer was detected with laser scanning confocal microscopy. The accumulation of TCTB in human malignant tumor cells was measured by fluorescence spectrometer, and the pathway of cell death was analyzed by flow cytometry. S180 tumor model was used to evaluate the anti-tumor effects of TCTB-PDT. And histopathological study was also used to confirm the anti-tumor effect.
Results
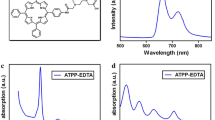
TCTB shows a singlet oxygen quantum yield of 0.56 and displays a characteristic long wavelength absorption peak at 732 nm. The accumulation of TCTB increased in time-dependent manner, and it was found in cytoplasm and nuclear membranes. In vitro PDT using TCTB and Nd:YAG laser showed drug concentration-, laser dose-dependent cytotoxicity to human esophageal cancer Eca-109 cells. In mice bearing osteosarcoma S180 tumors, the combined use of 10 mg/kg TCTB and 120 J/cm2 showed superior anti-tumor activity. Histology examination of tumor tissues revealed that PDT using TCTB and the Nd:YAG laser induced tumor cells shrunken and necrotic.
Conclusion
In in vitro and in vivo studies, we found that TCTB has excellent anti-tumor effect. It suggests that TCTB is a potential photosensitizer of PDT for cancer.







Similar content being viewed by others
References
Allison RR, Moghissi K (2013) Photodynamic Therapy (PDT): PDT Mechanisms. Clin endosc 46(1):24–29. doi:10.5946/ce.2013.46.1.24
Ashen-Garry D, Selke M (2014) Singlet oxygen generation by cyclometalated complexes and applications. Photochem Photobiol 90(2):257–274. doi:10.1111/php.12211
Brundish DE, Love WG (2000) Photodynamic therapy comes of age. IDrugs Investig Drugs J 3(12):1487–1508
Casas A, Fukuda H, Meiss R, Batlle AM (1999) Topical and intratumoral photodynamic therapy with 5-aminolevulinic acid in a subcutaneous murine mammary adenocarcinoma. Cancer Lett 141(1–2):29–38
Castano AP, Mroz P, Hamblin MR (2006) Photodynamic therapy and anti-tumour immunity. Nat Rev Cancer 6(7):535–545. doi:10.1038/nrc1894
Celli JP, Spring BQ, Rizvi I, Evans CL, Samkoe KS, Verma S, Pogue BW, Hasan T (2010) Imaging and photodynamic therapy: mechanisms, monitoring, and optimization. Chem Rev 110(5):2795–2838. doi:10.1021/cr900300p
Chen YH, Li GL, Pandey RK (2004) Synthesis of bacteriochlorins and their potential utility in photodynamic therapy (PDT). Curr Org Chem 8(12):1105–1134. doi:10.2174/1385272043370131
Cheng Y, Samia AC, Meyers JD, Panagopoulos I, Fei BW, Burda C (2008) Highly efficient drug delivery with gold nanoparticle vectors for in vivo photodynamic therapy of cancer. J Am Chem Soc 130(32):10643–10647. doi:10.1021/Ja801631c
Dabrowski JM, Urbanska K, Arnaut LG, Pereira MM, Abreu AR, Simoes S, Stochel G (2011) Biodistribution and photodynamic efficacy of a water-soluble, stable, halogenated bacteriochlorin against melanoma. ChemMedChem 6(3):465–475. doi:10.1002/cmdc.201000524
Dabrowski JM, Arnaut LG, Pereira MM, Urbanska K, Simoes S, Stochel G, Cortes L (2012) Combined effects of singlet oxygen and hydroxyl radical in photodynamic therapy with photostable bacteriochlorins: evidence from intracellular fluorescence and increased photodynamic efficacy in vitro. Free Radic Biol Med 52(7):1188–1200. doi:10.1016/j.freeradbiomed.2011.12.027
Detty MR, Gibson SL, Wagner SJ (2004) Current clinical and preclinical photosensitizers for use in photodynamic therapy. J Med Chem 47(16):3897–3915. doi:10.1021/Jm040074b
Ding X, Xu Q, Liu F, Zhou P, Gu Y, Zeng J, An J, Dai W, Li X (2004) Hematoporphyrin monomethyl ether photodynamic damage on HeLa cells by means of reactive oxygen species production and cytosolic free calcium concentration elevation. Cancer Lett 216(1):43–54. doi:10.1016/j.canlet.2004.07.005
Dougherty TJ (1995) Photodynamic therapy: part II. Semin Surg Oncol 11(5):333–334
Finlay JC, Mitra S, Patterson MS, Foster TH (2004) Photobleaching kinetics of Photofrin in vivo and in multicell tumour spheroids indicate two simultaneous bleaching mechanisms. Phys Med Biol 49(21):4837–4860
Gollnick SO, Owczarczak B, Maier P (2006) Photodynamic therapy and anti-tumor immunity. Lasers Surg Med 38(5):509–515. doi:10.1002/lsm.20362
Hampton JA, Selman SH (1992) Mechanisms of cell killing in photodynamic therapy using a novel in vivo drug/in vitro light culture system. Photochem Photobiol 56(2):235–243
Huang Z (2005) A review of progress in clinical photodynamic therapy. Technol Cancer Res Treat 4(3):283–293
Huang YY, Mroz P, Zhiyentayev T, Sharma SK, Balasubramanian T, Ruzie C, Krayer M, Fan D, Borbas KE, Yang E, Kee HL, Kirmaier C, Diers JR, Bocian DF, Holten D, Lindsey JS, Hamblin MR (2010) In vitro photodynamic therapy and quantitative structure-activity relationship studies with stable synthetic near-infrared-absorbing bacteriochlorin photosensitizers. J Med Chem 53(10):4018–4027. doi:10.1021/jm901908s
Ji Z, Yang G, Shahzidi S, Tkacz-Stachowska K, Suo Z, Nesland JM, Peng Q (2006) Induction of hypoxia-inducible factor-1alpha overexpression by cobalt chloride enhances cellular resistance to photodynamic therapy. Cancer Lett 244(2):182–189. doi:10.1016/j.canlet.2005.12.010
Juarranz A, Jaen P, Sanz-Rodriguez F, Cuevas J, Gonzalez S (2008) Photodynamic therapy of cancer. Basic principles and applications. Clin Transl Oncol 10(3):148–154
Juzeniene A, Peng Q, Moan J (2007) Milestones in the development of photodynamic therapy and fluorescence diagnosis. Photochem Photobiol Sci 6(12):1234–1245. doi:10.1039/b705461k
Lovell JF, Liu TW, Chen J, Zheng G (2010) Activatable photosensitizers for imaging and therapy. Chem Rev 110(5):2839–2857. doi:10.1021/cr900236h
Miskovsky P (2002) Hypericin–a new antiviral and antitumor photosensitizer: mechanism of action and interaction with biological macromolecules. Curr Drug Targets 3(1):55–84
O’Connor AE, Gallagher WM, Byrne AT (2009) Porphyrin and nonporphyrin photosensitizers in oncology: preclinical and clinical advances in photodynamic therapy. Photochem Photobiol 85(5):1053–1074. doi:10.1111/j.1751-1097.2009.00585.x
Osaki T, Takagi S, Hoshino Y, Okumura M, Fujinaga T (2006) Intracellular localization and concentration as well as photodynamic effects of benzoporphyrin derivative monoacid ring A in four types of rodent tumor cells. Cancer Lett 243(2):281–292. doi:10.1016/j.canlet.2005.11.044
Osaki T, Takagi S, Hoshino Y, Okumura M, Fujinaga T (2007) Antitumor effects and blood flow dynamics after photodynamic therapy using benzoporphyrin derivative monoacid ring A in KLN205 and LM8 mouse tumor models. Cancer Lett 248(1):47–57. doi:10.1016/j.canlet.2006.05.021
Phillips D (2010) Light relief: photochemistry and medicine. Photochem Photobiol Sci 9(12):1589–1596. doi:10.1039/c0pp00237b
Pineiro M, Gonsalves AMDR, Pereira MM, Formosinho SJ, Arnaut LG (2002) New halogenated phenylbacteriochlorins and their efficiency in singlet-oxygen sensitization. J Phys Chem A 106(15):3787–3795. doi:10.1021/Jp013678p
Redmond RW, Gamlin JN (1999) A compilation of singlet oxygen yields from biologically relevant molecules. Photochem Photobiol 70(4):391–475
Silva EF, Serpa C, Dabrowski JM, Monteiro CJ, Formosinho SJ, Stochel G, Urbanska K, Simoes S, Pereira MM, Arnaut LG (2010) Mechanisms of singlet-oxygen and superoxide-ion generation by porphyrins and bacteriochlorins and their implications in photodynamic therapy. Chemistry 16(30):9273–9286. doi:10.1002/chem.201000111
Spiller W, Kliesch H, Wohrle D, Hackbarth S, Roder B, Schnurpfeil G (1998) Singlet oxygen quantum yields of different photosensitizers in polar solvents and micellar solutions. J Porphyr Phthalocyanines 2(2):145–158. doi:10.1002/(Sici)1099-1409(199803/04)2:2<145:Aid-Jpp60>3.0.Co;2-2
Stylli SS, Kaye AH (2006) Photodynamic therapy of cerebral glioma—a review. Part II—clinical studies. J clin neurosci 13(7):709–717. doi:10.1016/j.jocn.2005.11.012
Triesscheijn M, Baas P, Schellens JH, Stewart FA (2006) Photodynamic therapy in oncology. Oncologist 11(9):1034–1044. doi:10.1634/theoncologist.11-9-1034
Tsai JC, Wu CL, Chien HF, Chen CT (2005) Reorganization of cytoskeleton induced by 5-aminolevulinic acid-mediated photodynamic therapy and its correlation with mitochondrial dysfunction. Lasers Surg Med 36(5):398–408. doi:10.1002/lsm.20179
van Duijnhoven FH, Rovers JP, Engelmann K, Krajina Z, Purkiss SF, Zoetmulder FA, Vogl TJ, Terpstra OT (2005) Photodynamic therapy with 5,10,15,20-tetrakis (m-hydroxyphenyl) bacteriochlorin for colorectal liver metastases is safe and feasible: results from a phase I study. Ann Surg Oncol 12(10):808–816. doi:10.1245/ASO.2005.09.005
Wang H, Li J, Lv T, Tu Q, Huang Z, Wang X (2013) Therapeutic and immune effects of 5-aminolevulinic acid photodynamic therapy on UVB-induced squamous cell carcinomas in hairless mice. Exp Dermatol 22(5):362–363. doi:10.1111/exd.12132
Yan YJ, Zheng MZ, Chen ZL, Yu XH, Yang XX, Ding ZL, Xu L (2010) Studies on preparation and photodynamic mechanism of chlorin P6-13,15-N-(cyclohexyl) cycloimide (Chlorin-H) and its antitumor effect for photodynamic therapy in vitro and in vivo. Bioorg Med Chem 18(17):6282–6291. doi:10.1016/j.bmc.2010.07.027
Acknowledgments
This work was supported by Chinese National Natural Science Foundation (21372042, 81101298, 81301878), Foundation of Shanghai government (13431900700, 14431906200, 13430722300, 14140903500, 13ZR1441000, 13ZR1440900, 14ZR1439800, 14ZR1439900, 201370) and Foundation of Yiwu Science and Technology Bureau (2011-G1-15, 2013-G3-03, 2012-G3-02).
Conflict of interest
We declare that we have no conflict of interest in relation to this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, LJ., O’Shea, D., Zhang, CY. et al. Evaluation of a bacteriochlorin-based photosensitizer’s anti-tumor effect in vitro and in vivo. J Cancer Res Clin Oncol 141, 1921–1930 (2015). https://doi.org/10.1007/s00432-015-1960-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-015-1960-z