Abstract
Multiple criteria and growth references have been proposed for extrauterine growth restriction (EUGR). We hypothesized that these may impact the diagnosis of EUGR. The objective was to evaluate the prevalence of EUGR with its different definitions and the concordance according to Fenton, Olsen, and INTERGROWTH-21st in very-low-birthweight (VLBW) infants. This is an observational, retrospective, and multicenter study including VLBW infants from the Spanish SEN1500 Network from 2011 to 2020. Patients with major congenital anomalies, embryopathies, and gestational age less than 24 weeks were excluded. EUGR prevalence was calculated at discharge with cross-sectional, longitudinal, “true” cross-sectional, and “true” longitudinal definitions. Concordance was assessed with Fleiss’ kappa coefficient. 23582 VLBW infants from 77 NICUs were included. In total, 50.4% were men with a median of gestational age of 29 (4) weeks. The prevalence of EUGR (cross-sectional, longitudinal, and “true”) was variable for weight, length, and head circumference. Overall, the prevalence was higher with Fenton and lower with Olsen (cross-sectional and “true” cross-sectional) and INTERGROWTH-21st (longitudinal and “true” longitudinal). Agreement among the charts by weight was good only for cross-sectional EUGR and moderate for longitudinal, “true” cross-sectional, and “true” longitudinal. Concordance was good or very good for EUGR by length and head circumference.
Conclusions: The prevalence of EUGR with the most commonly used definitions was variable in the cohort. Agreement among growth charts was moderate for all the definitions of EUGR by weight except cross-sectional and good or very good for length and head circumference. The choice of reference chart can impact the establishment of the diagnosis of EUGR.
What is known: • EUGR has been defined in the literature and daily practice considering weight, length and head circumference with multiple criteria (cross-sectional, longitudinal, and “true” definition) • Different growth charts have been used for EUGR diagnosis |
What is new: • Prevalence of EUGR is variable depending on the definition and growth chart used in our cohort of VLBW infants • For the most frequently EUGR criteria used, traditionally considering weight, concordance among Fenton, Olsen and INTERGROWTH-21st growth charts is only moderate for all the definitions of EUGR by weight except cross-sectional definition. Concordance among the charts is good or very good for the different criteria of EUGR by head circumference and length |
Similar content being viewed by others

Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Postnatal growth of preterm infants continues to be a challenge in neonatology [1]. Consensus among neonatologists on the ideal extrauterine growth pattern for preterm infants and the optimal practices for monitoring growth in the neonatal intensive care unit (NICU) has not been reached [2].
Extrauterine growth restriction has been described in the literature as a concept to identify inadequate postnatal growth that does not meet the expectations [3]. Despite the frequency of use of the term extrauterine growth restriction both in the clinical practice and literature, there is significant variability on the definitions used and timing for diagnosis for EUGR [4]. Traditionally, EUGR has been described as weight at 36 weeks or discharge below the 10th percentile [5], < − 2 Z-score [6, 7], < − 1.5 z-score [8] or, less frequently, below the 3rd percentile [9] using different growth charts (referred to as cross-sectional definition). An alternate longitudinal definition has been applied to weight loss more than one [9] or two standard [6] deviations from weight at birth. Recently, a new definition has been proposed named as “true” EUGR that would include patients who meet the previously described criteria for EUGR (either cross-sectional definition or longitudinal definition) amongst those not being small for gestational age (SGA) at birth [10, 11]. Several studies have previously described the prevalence of EUGR with the cross-sectional and longitudinal definitions for weight in the neonatal population. However, the literature on prevalence of EUGR with the latest criteria is scarce.
Increasing growth references or standards have been developed and published in the last two decades, with varying methodology, populations, and objectives [12]. Olsen et al. [13] and Fenton et al. [14, 15] growth references are two of the most commonly used in the NICU for preterm infants. Recently, the International Fetal and Newborn Growth Consortium for the 21st Century (INTERGROWTH-21st) published their fetal [16], preterm [17], and newborn [18] growth standards. While Olsen and Fenton are based on transversal anthropometrical data of different populations, the latest was developed based on prospective follow-up of a cohort of newborns with the objective of developing a standard of growth. However, the establishment in preterms of the optimal conditions that would be required for the development of standards of growth can also be challenging. The particularities of the preterm population and the different comorbidities they experience can impact growth and difficult the achievement of a “healthy preterm” [12].
Despite the multiple criteria proposed for EUGR and the frequent use of this term in the literature [4], this term has also raised controversies. A group of experts has highlighted that EUGR could lead to overdiagnosing growth deviations with no clear relation to clinically significant outcomes such as neurodevelopmental [19]. A recent review has also raised the challenges to compare studies of EUGR and neurodevelopment due to the differences in EUGR criteria and neurodevelopmental assessments amongst different papers [20]. Moreover, the previously described group of experts have also suggested revision of the criteria and calculation of the diagnostic accuracy [19]. With this goal, a study from 2024 has evaluated infants born at less than 30 weeks or with a birthweight less than 1500 g from the Preterm Multi-center (PreM) Growth cohort study, describing that despite a significant proportion of preterms plotting low at 36 weeks of postmenstrual age, most of those with adequate nutritional support had experienced catch-up by 3 years of corrected gestational age with a limited ability of growth faltering to predict cognitive scores [21].
The primary objectives of this study are to describe the prevalence of extrauterine growth restriction with the most frequently used definitions (cross-sectional, longitudinal, and “true” EUGR) and to assess the concordance amongst Fenton, Olsen, and INTERGROWTH-21st for those definitions in a multicenter cohort of very-low-birthweight (VLBW) infants in Spain.
Materials and methods
An observational, retrospective, and multicenter cohort study was conducted including all VLBW newborns from SEN1500 Network. SEN1500 is a national database from the Spanish Society of Neonatology that collects data for preterms with gestational age less than 32 weeks or birthweight less than 1500 g admitted to all voluntarily integrated Spanish level three NICUs.
The study was conducted from January, 1st, 2011 to December, 31st, 2020. All VLBW infants admitted to each participating site in SEN1500 Network during the period of study were eligible for inclusion. Infants with major congenital anomalies, congenital embryopathies with growth impairment (as congenital infection by cytomegalovirus), and gestational age less than 24 weeks (as INTERGROWTH-21st growth chart is not available for those gestational ages) were excluded from the study.
Ethics approval was obtained from “Comité de Ética de la Investigación del Principado de Asturias” with reference number 2022.586. Parents of newborns included in the SEN1500 database provided written consent for the inclusion in the database and its use for clinical studies. Moreover, the authors have no conflict of interests to declare.
Data were collected in accordance to SEN1500 standardized operational definitions, with particular consideration of the criteria proposed by NICHD in 2019 for the diagnosis of bronchopulmonary dysplasia [22].
Percentiles and Z-score for weight (W), length (L), and head circumference (HC) of each participant were calculated using the University of Calgary Fenton calculator application [23], Peditool Olsen bulk calculator [24], and the INTERGROWTH-21st calculator application [25, 26], publicly available. For the latest, reference of newborn size for preterm infants was used to evaluate infants at birth while chart of postnatal growth of preterm infant was used to assess postnatal growth. Infants were categorized by weight at birth in small (less than the 10th centile), appropriate (between the 10th and 90th centile) and large for gestational age (more than the 90th centile).
Provided there is no standard definition of EUGR in neonates, we elected to follow the most commonly criteria previously published, defining EUGR as follows: (1) weight at discharge less than 10th centile (cross-sectional definition), (2) decrease of more than 1 SD since birth to the time of discharge home (longitudinal), and (3) “true” EUGR, either cross-sectional or longitudinal EUGR in non-SGA infants at birth. Considering the previously published potential implications of head circumference [27,28,29] and length [30, 31] in neurodevelopment, we elected to study EUGR with the same previously described definitions for length and head circumference.
All statistical analyses were carried out using R (v 3.4.4, Open-Source International Collaborative), R Studio (v 1.1.463, Open-Source International Collaborative) and SPSS (V27). Basic descriptive statistics were used to characterize data. As the majority of the quantitative variables were not normally distributed, median and interquartile range were used to describe quantitative variables, and percentages were used for the qualitative variables. The primary outcome of interest (concordance between Fenton, Olsen, and INTERGROWTH-21st growth references) was assessed with Fleiss’ kappa coefficient.
Results
Study population
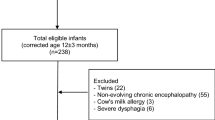
The initial data from SEN1500 included 26,146 very preterm infants or very low weight for gestational age. From this sample, a total of 25,382 very-low-weight gestational-age infants were eligible for the study. In total, 2565 patients met the exclusion criteria due to the following: major congenital anomalies (1222 patients), congenital embryopathies with growth impairment (147 patients), gestational age less than 24 weeks of gestation (431 patients). The final study sample was constituted by 23,582 patients as represented in the flowchart in Fig. 1.
Characteristics of the study population
Patients included were admitted from January 2011 to December 2020 in 77 participating sites. Median gestational age at birth was 29 (interquartile range [IQR] 4) weeks, and 50.4% of the sample were males (11,859 infants). Perinatal characteristics of the population included are summarized in Table 1.
Median weight, length, and head circumference at birth were 1119 (IQR 460) grams, 37.15 (IQR 5) centimeters, and 26.3 (IQR 3.5) centimeters, respectively. Classification of growth patterns at birth by weight and growth curve are presented in Table 2. Anthropometric measurements at the time of discharge were median weight 2200 (IQR 565) grams, length 44.5 (IQR 3.5) centimeters, and head circumference 32.5 (IQR 2) cm.
Global survival in the sample was 89.4% as 2499 patients died during the hospitalization in the NICU. From the patients in whom information was available, overall survival without morbidities was 49% (10,000 patients). Morbidities experienced during admission are summarized in Table 3. Prevalence of morbidities and mortality during admission of infants classified as SGA by weight with the different growth curves is summarized in Table 4.
Prevalence of EUGR
The prevalence of EUGR with cross-sectional, longitudinal, and “true” criteria for weight in the study sample varied with the use of different references as depicted in Table 5. Overall, the prevalence of EUGR is higher in all the proposed definitions of the concept with the use of Fenton reference in our population as presented in Fig. 2. This tendency persisted by sex and evaluating length and head circumference, as described in Table 6.
The prevalence of EUGR by growth pattern at birth (weight), classified by SGA, AGA, and LGA, was calculated with the different criteria and growth curves by weight and is presented in Table 7.
Concordance among Fenton, Olsen, and INTERGROWTH-21st references for diagnosis of EUGR
Fleiss’ kappa coefficients and 95% confidence intervals for diagnosis of extrauterine growth restriction using the three assessed growth charts by each definition of EUGR are depicted in Table 8. Agreement amongst charts was good for cross-sectional EUGR criteria by weight and moderate for the rest of the criteria of EUGR by weight (longitudinal, “true” cross-sectional, and “true” longitudinal) in VLBW infants. Concordance for EUGR criteria by length and head circumference was good or very good with all the definitions studied.
Discussion
There is no consensus definition of extrauterine growth restriction nor ideal growth reference for the assessment. In consequence, there is significant variability in the prevalence of EUGR depending on the definition and growth chart used as reflected in this study. In our population, the prevalence ranged from 19.6 using the EUGR longitudinal definition to 56.6% using the cross-sectional definition.
According to the cross-sectional definition (weight at discharge-discharge below the 10th percentile for GA), EUGR prevalence was reported to be 56.8% in Shanghai [32], 50.3% by Vermont Oxford Network [33], 74% in newborns with GA less than 30 weeks in Italy [6], and 59.2% in a Spanish cohort [34].
For the longitudinal definition, studies also differ in the choice of cutoff for the change in z-score, which results in difficulties in interpreting and comparing findings. Peila et al. [6] reported a prevalence of 92% of longitudinal EUGR defined as the loss of 1 standard deviation (SD) in preterms < 30 weeks while Franz et al. [28] described 50% in preterms < 26 weeks of GA. For longitudinal EUGR described as a change in more than 2 z-score from birth to discharge, a study from Israel reported a prevalence ranging from 11.7 to 7.2% for severe EUGR [35].
The most recent criteria proposed is “true” EUGR. Prevalence of the newest EUGR concept, which would include cross-sectional and longitudinal criteria in non-SGA infants at birth, is also variable. “True” cross-sectional EUGR prevalence has previously been described as 60.4% [6], 12.3% [10], and 17% (in non-SGA babies with birthweight between 750 and 1250 g) [36]. “True” EUGR with longitudinal criteria has been estimated from 39.6 to 52.7% with 1 SD cutoff [37] and 13% using 2 SD criteria [38].
As proposed by Peila et al. [6], the different definitions measure different aspects of the growth. While the cross-sectional criteria only consider the evaluation at discharge, the longitudinal criteria evaluate changes from birth to discharge. Moreover, “true” EUGR would consider only those infants with postnatal growth impairment, excluding those with fetal growth restriction [10].
Moreover, variability in prevalence persists amongst the three growth charts studied when considering each definition alone. Overall, prevalence is higher in our study with the use of the Fenton growth chart and lower with Olsen (cross-sectional and “true” cross-sectional) and IW-21 (longitudinal and “true” longitudinal). El Rafei et al. [39], Reddy et al. [40], González-García et al. [37], and Tuzun et al. [41] reported similar results, with higher prevalence of EUGR with cross-sectional definition using Fenton charts than with intergrowth-21st charts. In contrast, Lan et al. [42] did not find significant differences in the incidences of cross-sectional EUGR by Fenton and Intergrowth-21st charts. Despite our choice of criteria for the cross-sectional EUGR definition, it is important to highlight the prevalence described in studies using other criteria for this definition. A study by Yazidi et al. [7] described a significantly higher prevalence of cross-sectional EUGR (using the criteria of weight at discharge less than − 2 z-score) with Fenton curves than INTERGROWTH-21st. Our study describes results consistent with the literature for the longitudinal EUGR, with higher prevalence when using Fenton charts compared to Intergrowth-21st [42, 43]. It is important to consider for the interpretation of the previously described results that the varying methodology followed for the construction of each growth curve could potentially also affect these results. While Fenton charts from 2013 are based on the study of the anthropometry of 3,986,456 infants and Olsen studied 391,681 preterms, INTERGROWTH-21st preterm growth standards include a smaller number of preterm patients and are based on the study of less than 500 patients. Consideration should also be given to the prospective study conducted in INTERGROWTH-21st compared to Fenton and Olsen (retrospective).
Agreement among the three assessed growth charts for each definition of EUGR was good or very good for EUGR criteria by length and head circumference. EUGR by weight, which has been traditionally used for the definition, has good concordance only for the cross-sectional EUGR criteria and is moderate for the rest of the criteria of EUGR by weight (longitudinal, “true” cross-sectional, and “true” longitudinal) in VLBW infants. Results are consistent with those from González-García et al. [37]. In contrast, a study from Lan et al. [42] reported low concordances for cross-sectional EUGR for weight, length, and head circumference with Fenton and Intergrowth-21st in infants < 32 weeks of GA, with good concordance between those previously described for longitudinal EUGR. Previous studies have also reported low agreement between the cross-sectional and longitudinal definitions of EUGR with Cohen’s kappa coefficient from 0.07 to 0.38 [6]. Moreover, a recent study from 2024 has evaluated the same growth curves used in our study (Fenton, Olsen, and INTERGROWTH-21st) and studied the discrimination power of the curves for the risk of mortality in different populations, finding that INTERGROWTH-21st fetal growth curves had the strongest discrimination power for very-low-birthweight infants, the population assessed in our study [44].
It is important to consider that INTERGROWTH-21st preterm postnatal growth standards present some particularities for the anthropometric evaluation of preterm infants. While length and head circumference can be monitored with this standard from birth, as physiological weight loss was not considered for their development, weight centiles may be assigned and evaluated when preterms start regaining weight.
Considering all the above presented, it is crucial to be mindful of the significant variability of diagnosis depending on the choice of growth chart and definition of EUGR as establishing the diagnosis of EUGR will likely have a direct impact on the clinical evaluation and follow-up of the patients.
Limitations of the study include that our evaluation of EUGR in very-low-birthweight infants was conducted with the exclusion of infants born at less than 24 weeks of gestational age. This exclusion was performed due to the absence of percentiles and z-score for anthropometry at this gestational age with INTERGROWTH-21st, to allow comparisons among the three studied growth curves, but it may have impacted the results obtained. Moreover, the retrospective nature of the study conducted with limited data on nutrition practices did not allow us to study any potential association between changes in nutritional practices and EUGR. Another limitation of our study is that no follow-up after discharge from the hospital was included, so morbidities and mortality studied are limited to in-hospital. Finally, despite the study being multicenter and the size of the sample included, our study includes only infants cared for at hospitals participating in SEN1500, which can also limit the external validity of the study.
Conclusions
Prevalence of EUGR varied in our cohort of VLBW infants with the most commonly used definitions of the term (cross-sectional, longitudinal, “true” cross-sectional, and “true” longitudinal) using Fenton, Olsen, and Intergrowth-21st. Moreover, agreement among the three growth charts was moderate for all the definitions of EUGR by weight except cross-sectional and good or very good for length and head circumference. The choice of reference chart can impact the establishment of the diagnosis of EUGR.
Data availability
All data, code, and material will be available on request following publication.
Abbreviations
- cm:
-
Centimeters
- EUGR:
-
Extrauterine growth restriction
- g:
-
Grams
- HC:
-
Head circumference
- IQR:
-
Interquartilic range
- L:
-
Length
- NICU:
-
Neonatal intensive care unit
- SD:
-
Standard deviation
- SGA:
-
Small for gestational age
- VLBW:
-
Very low birthweight
- W:
-
Weight
References
Barros FC, Papageorghiou AT, Victora CG, Noble JA, Pang R, Iams J, Cheikh Ismail L, Goldenberg RL, Lambert A, Kramer MS et al (2015) The distribution of clinical phenotypes of preterm birth syndrome: implications for prevention. JAMA Pediatr 169:220. https://doi.org/10.1001/jamapediatrics.2014.3040
Villar J, Giuliani F, Barros F, Roggero P, Coronado Zarco IA, Rego MAS, Ochieng R, Gianni ML, Rao S, Lambert A et al (2018) Monitoring the postnatal growth of preterm infants: a paradigm change. Pediatrics 141:e20172467. https://doi.org/10.1542/peds.2017-2467
Gounaris AK, Sokou R, Gounari EA, Panagiotounakou P, Grivea IN (2023) Extrauterine growth restriction and optimal growth of very preterm neonates: state of the art. Nutrients 15:3231. https://doi.org/10.3390/nu15143231
Fenton TR, Chan HT, Madhu A, Griffin IJ, Hoyos A, Ziegler EE, Groh-Wargo S, Carlson SJ, Senterre T, Anderson D et al (2017) Preterm infant growth velocity calculations: a systematic review. Pediatrics 139:e20162045. https://doi.org/10.1542/peds.2016-2045
Clark RH, Thomas P, Peabody J (2003) Extrauterine growth restriction remains a serious problem in prematurely born neonates. Pediatrics 111:986–990. https://doi.org/10.1542/peds.111.5.986
Peila C, Spada E, Giuliani F, Maiocco G, Raia M, Cresi F, Bertino E, Coscia A (2020) Extrauterine growth restriction: definitions and predictability of outcomes in a cohort of very low birth weight infants or preterm neonates. Nutrients 12:1224. https://doi.org/10.3390/nu12051224
Yazici A, Buyuktiryaki M, Sari FN et al (2023) Comparison of different growth curves in the assessment of extrauterine growth restriction in very low birth weight preterm infants. Arch Pediatr 30:31–35. https://doi.org/10.1016/j.arcped.2022.11.008
Zozaya C, Díaz C, Saenz de Pipaón (2018) M. How should we define postnatal growth restriction in preterm infants? Neonatology 114:177–180. https://doi.org/10.1159/000489388
Shah PS, Wong KY, Merko S, Bishara R, Dunn M, Asztalos E (2006) Darling, P.B. Postnatal growth failure in preterm infants: ascertainment and relation to long-term outcome. Journal of Perinatal Medicine 34. https://doi.org/10.1515/JPM.2006.094
Figueras-Aloy J, Palet-Trujols C, Matas-Barceló I, Botet-Mussons F, Carbonell-Estrany X (2020) Extrauterine growth restriction in very preterm infant: etiology, diagnosis, and 2-year follow-up. Eur J Pediatr 179:1469–1479. https://doi.org/10.1007/s00431-020-03628-1
Zhao T, Feng H-M, Caicike B, Zhu Y-P (2021) Investigation into the current situation and analysis of the factors influencing extrauterine growth retardation in preterm infants. Front Pediatr 9:643387. https://doi.org/10.3389/fped.2021.643387
Cormack BE, Embleton ND, Van Goudoever JB, Hay WW, Bloomfield FH (2016) Comparing apples with apples: it is time for standardized reporting of neonatal nutrition and growth studies. Pediatr Res 79:810–820. https://doi.org/10.1038/pr.2016.26
Olsen IE, Groveman SA, Lawson ML, Clark RH, Zemel BS (2010) New intrauterine growth curves based on United States data. Pediatrics 125:e214–e224. https://doi.org/10.1542/peds.2009-0913
Fenton TR (2003) A new growth chart for preterm babies: Babson and Benda’s chart updated with recent data and a new format. BMC Pediatr 3:13. https://doi.org/10.1186/1471-2431-3-13
Fenton TR, Kim JH (2013) A systematic review and meta-analysis to revise the fenton growth chart for preterm infants. BMC Pediatr 13:59. https://doi.org/10.1186/1471-2431-13-59
Papageorghiou AT, Ohuma EO, Altman DG, Todros T, Ismail LC, Lambert A, Jaffer YA, Bertino E, Gravett MG, Purwar M et al (2014) International standards for fetal growth based on serial ultrasound measurements: the fetal growth longitudinal study of the INTERGROWTH-21st project. The Lancet 384:869–879. https://doi.org/10.1016/S0140-6736(14)61490-2
Villar J, Giuliani F, Bhutta ZA, Bertino E, Ohuma EO, Ismail LC, Barros FC, Altman DG, Victora C, Noble JA et al (2015) Postnatal growth standards for preterm infants: the preterm postnatal follow-up study of the INTERGROWTH-21 St project. Lancet Glob Health 3:e681–e691. https://doi.org/10.1016/S2214-109X(15)00163-1
Villar J, Cheikh Ismail L, Victora CG, Ohuma EO, Bertino E, Altman DG, Lambert A, Papageorghiou AT, Carvalho M, Jaffer YA et al (2014) International standards for newborn weight, length, and head circumference by gestational age and sex: the newborn cross-sectional study of the INTERGROWTH-21st project. Lancet 384:857–868. https://doi.org/10.1016/S0140-6736(14)60932-6
Fenton TR, Cormack B, Goldberg D et al (2020) “Extrauterine growth restriction” and “postnatal growth failure” are misnomers for preterm infants. J Perinatol 40:704–714. https://doi.org/10.1038/s41372-020-0658-5
González-López C, Solís-Sánchez G, Lareu-Vidal S et al (2024) Variability in definitions and criteria of extrauterine growth restriction and its association with neurodevelopmental outcomes in preterm infants: a narrative review. Nutrients 16:968. https://doi.org/10.3390/nu16070968
Fenton TR, Samycia L, Elmrayed S et al (2024) Growth patterns by birth size of preterm children born at 24–29 gestational weeks for the first 3 years. Paediatr Perinat Epidemiol. https://doi.org/10.1111/ppe.13081
Jensen EA, Dysart K, Gantz MG et al (2019) The diagnosis of bronchopulmonary dysplasia in very preterm infants. An evidence-based approach. Am J Respir Crit Care Med 200:751–759. https://doi.org/10.1164/rccm.201812-2348OC
Calculators & Apps Available online: https://ucalgary.ca/resource/preterm-growth-chart/calculators-apps (accessed on 19 March 2023)
PediTools Universal Calculator Available online: https://peditools.org/peditools_universal/ (accessed on 20 September 2023)
Newborn size for very preterm infants. INTERGROWTH-21st available online: https://intergrowth21.tghn.org/very-preterm-size-birth/ (accessed on 20 March 2024)
Postnatal growth of preterm infants. INTERGROWTH-21st available online: http://intergrowth21.ndog.ox.ac.uk/preterm (accessed on 21 March 2024)
Ehrenkranz RA, Dusick AM, Vohr BR, Wright LL, Wrage LA, Poole WK (2006) For the national institutes of child health and human development neonatal research network growth in the neonatal intensive care unit influences neurodevelopmental and growth outcomes of extremely low birth weight infants. Pediatrics 117:1253–1261. https://doi.org/10.1542/peds.2005-1368
Franz AR, Pohlandt F, Bode H, Mihatsch WA, Sander S, Kron M, Steinmacher J (2009) Intrauterine, Early neonatal, and postdischarge growth and neurodevelopmental outcome at 54 years in extremely preterm infants after intensive neonatal nutritional support. Pediatrics 123:e101-109. https://doi.org/10.1542/peds.2008-1352
Belfort MB, Rifas-Shiman SL, Sullivan T, Collins CT, McPhee AJ, Ryan P, Kleinman KP, Gillman MW, Gibson RA, Makrides M (2011) Infant growth before and after term: effects on neurodevelopment in preterm infants. Pediatrics 128:e899–e906. https://doi.org/10.1542/peds.2011-0282
Meyers JM, Tan S, Bell EF, Duncan AF, Guillet R, Stoll BJ, D’Angio CT (2019) Eunice Kennedy Shriver National Institute of Child Health and Human Development neonatal research network neurodevelopmental outcomes among extremely premature infants with linear growth restriction. J Perinatol 39:193–202. https://doi.org/10.1038/s41372-018-0259-8
Sammallahti S, Pyhälä R, Lahti M, Lahti J, Pesonen A-K, Heinonen K, Hovi P, Eriksson JG, Strang-Karlsson S, Andersson S et al (2014) Infant growth after preterm birth and neurocognitive abilities in young adulthood. J Pediatr 165:1109-1115.e3. https://doi.org/10.1016/j.jpeds.2014.08.028
Shan HM, Cai W, Cao Y, Fang BH, Feng Y (2009) Extrauterine growth retardation in premature infants in Shanghai: a multicenter retrospective review. Eur J Pediatr 168:1055–1059. https://doi.org/10.1007/s00431-008-0885-9
Horbar JD, Ehrenkranz RA, Badger GJ, Edwards EM, Morrow KA, Soll RF, Buzas JS, Bertino E, Gagliardi L, Bellù R (2015) Weight growth velocity and postnatal growth failure in infants 501 to 1500 grams: 2000–2013. Pediatrics 136:e84-92. https://doi.org/10.1542/peds.2015-0129
Avila-Alvarez A, Solar Boga A, Bermúdez-Hormigo C, Fuentes Carballal J (2018) Extrauterine growth restriction among neonates with a birthweight less than 1,500grams. An Pediatr (Engl Ed) 89:325–332. https://doi.org/10.1016/j.anpedi.2018.02.004
Ofek Shlomai N, Reichman B, Lerner-Geva L, Boyko V, Bar-Oz B (2014) Population-based study shows improved postnatal growth in preterm very-low-birthweight infants between 1995 and 2010. Acta Paediatr 103:498–503. https://doi.org/10.1111/apa.12569
Karagol BS, Zenciroglu A, Okumus N, Polin RA (2013) Randomized controlled trial of slow vs rapid enteral feeding advancements on the clinical outcomes of preterm infants with birth weight 750–1250 g. JPEN J Parenter Enteral Nutr 37:223–228. https://doi.org/10.1177/0148607112449482
González-García L, García-López E, Fernández-Colomer B, Mantecón-Fernández L, Lareu-Vidal S, Suárez-Rodríguez M, Arias-Llorente RP, Solís-Sánchez G (2021) Extrauterine growth restriction in very low birth weight infants: concordance between Fenton 2013 and INTERGROWTH-21st growth charts. Front Pediatr 9:690788. https://doi.org/10.3389/fped.2021.690788
Roggero P, Giannì ML, Orsi A, Amato O, Piemontese P, Liotto N, Morlacchi L, Taroni F, Garavaglia E, Bracco B et al (2012) Implementation of nutritional strategies decreases postnatal growth restriction in preterm infants. PLoS ONE 7:e51166. https://doi.org/10.1371/journal.pone.0051166
El Rafei R, Jarreau P-H, Norman M, Maier RF, Barros H, Reempts PV, Pedersen P, Cuttini M, Zeitlin J (2021) EPICE research group variation in very preterm extrauterine growth in a European multicountry cohort. Arch Dis Child Fetal Neonatal Ed 106:316–323. https://doi.org/10.1136/archdischild-2020-319946
Reddy KV, Sharma D, Vardhelli V, Bashir T, Deshbotla SK, Murki S (2021) Comparison of Fenton 2013 growth curves and intergrowth-21 growth standards to assess the incidence of intrauterine growth restriction and extrauterine growth restriction in preterm neonates ≤32 weeks. J Matern Fetal Neonatal Med 34:2634–2641. https://doi.org/10.1080/14767058.2019.1670795
Tuzun F, Yucesoy E, Baysal B, Kumral A, Duman N, Ozkan H (2018) Comparison of INTERGROWTH-21 and Fenton growth standards to assess size at birth and extrauterine growth in very preterm infants. J Matern Fetal Neonatal Med 31:2252–2257. https://doi.org/10.1080/14767058.2017.1339270
Lan S, Fu H, Zhang C, Chen Y, Pan L, Song S, Wang Y, Hong L (2023) Comparison of intergrowth-21st and Fenton growth standards to evaluate and predict the postnatal growth in eastern Chinese preterm infants. Front Pediatr 11:1259744. https://doi.org/10.3389/fped.2023.1259744
Kim Y-J, Shin SH, Cho H, Shin SH, Kim SH, Song IG, Kim E-K, Kim H-S (2021) Extrauterine growth restriction in extremely preterm infants based on the intergrowth-21st project preterm postnatal follow-up study growth charts and the Fenton growth charts. Eur J Pediatr 180:817–824. https://doi.org/10.1007/s00431-020-03796-0
Figueras-Aloy J, Izquierdo Renau M, Herranz Barbero A et al (2024) Comparative analysis of foetal and neonatal growth curves. An Pediatr (Engl Ed) 100:333–341. https://doi.org/10.1016/j.anpede.2024.04.002
Acknowledgements
We are indebted to all the investigators, coordinators, and members of the Spanish Neonatal Network SEN1500:
H. GERMANS TRIAS I PUJOL (Gemma Ginovart); H. CLÍNIC DE BARCELONA (Josep Figueras Aloy); HOSPITAL VALL D’HEBRON (Cèsar Wenceslao Ruiz Campillo);H. U. BASURTO (Alberto Pérez Legorburu); H.G. DE CASTELLÓN (Flavia Pronzato Cuello); H.U. DE SAN CECILIO (Ana María Campos Martínez); H. DE LEON (Sandra Terroba); H. CARLOS HAYA (Tomás Sanchez-Tamayo); H.I. LA PAZ (Ma Dolores Elorza Fernandez); H. CLÍNICO SAN CARLOS (Araceli Corredera Sánchez); H. CENTRAL DE ASTURIAS (Belén Fernández Colomer); COMPLEXO HOSPITALARIO PONTEVEDRA (Ma Angeles Martinez Fernandez); H. UNIVERSITARIO MARQUES DE VALDECILLA (Ana Belén Pérez Santos); H. DONOSTIA (Miguel Ángel Cortajarena Altuna); H.U.I. VIRGEN DEL ROCÍO (Carmen Macias Díaz); H.U. DE CANARIAS (Pedro Fuster Jorge); H. MIGUEL SERVET (Segundo Rite Gracia); H.C.U. LOZANO BLESA (Ma Purificación Ventura Faci); H.U. LA FE (Ma Isabel Izquierdo Macián); H. VIRGEN DE LA SALUD (Víctor Manuel Marugán Isabel); H.G. DE SEGOVIA (Alfonso Urbón); H.C.U. DE SANTIAGO (Ma Luz Couce Pico); H.U. SALAMANCA (Beatriz Vacas del Arco); H.G.U. GREGORIO MARAÑÓN (S. Zeballos Sarrato); H. SAN PEDRO DE LOGROÑO (Ma Yolanda Ruiz del Prado); H.U.M.I. LAS PALMAS (Lourdes Urquía Martí); H. DE CABUEÑES (Rafael García Mozo); H.U. REINA SOFÍA (Ma Pilar Jaraba Caballero); H.U. DE BURGOS (Cristina de Frutos Martínez); SCIAS-H. BARCELONA (Sílvia Martínez-Nadal); H. SAN JOAN DE DEU (Martin Iriondo); H. DE CRUCES (Amaya Rodríguez Serna); H.G.U. DE ALICANTE (Miriam Salvador Iglesias); H. VIRGEN DE LAS NIEVES (Maria Fernanda Moreno Galdo); CORPORACIO PARC TAULÍ (Joan Badia Barnusell); H.U. RIO HORTEGA (Ma Mar Montejo Vicente); H. TXAGORRITXU (Aintzane Euba); H. JUAN XXIII (Mar Albújar); H. DE GETAFE (Irene Cuadrado Perez); INSTI- TUT DEXEUS (Paula Serrano Acebes); COMPLEJO HOSPITALARIO ALBACETE (Andres Martinez Gutierrez); H. DEL BIERZO (Teresa Prada); H. DE LA SANTA CREU I SANT PAU (Elisenda Moliner Calderón); H. INFANTA MARGARITA (Jose Mª Barcia Ruiz); H. SAN PEDRO DE ALCÁNTARA CÁCERES (Elena María Marquez Isidro); H. SEVERO OCHOA (María Arroyas Sánchez); HOSPITAL ALVARO CUNQUEIRO (María Suárez Albo); H. VIRGEN DE LA CONCHA—COMPLEJO ASISTENCIAL DE ZAMORA (Víctor Manuel Marugán Isabel); H. DE JEREZ (María Victoria Ramos Ramos); H. MONTEPRINCIPE (Gerardo Romera Modamio); H. DE ELCHE (Carolina Vizcaíno); H. JUAN RAMÓN JIMÉNEZ (David Mora Navarro); H. VALME DE SEVILLA (Laura Acosta Gordillo); H.U. ARNAU DE VILANOVA DE LLEIDA (Eduard Soler Mir); H. VIRGEN DE LA MACARENA DE SEVILLA (Mercedes Granero Asensio); H.C.U. DE VALENCIA (Dr. Javier Estañ Capell); H.G.U. DE CIUDAD REAL (Miguel Angel García Cabezas); H. DE LA ZARZUELA (D. López Gómez); H. U. DE GIRONA DR. JOSEP TRUETA (Josep Perapoch Lopez); H. DE GRANOLLERS (Israel Anquela Sanz); H. PUERTA DEL MAR (Almudena Alonso Ojembarrena); H. 12 DE OCTUBRE (Carmen Rosa Pallás Alonso); H.U. NUESTRA SEÑORA DE CANDELARIA (Sabina Romero); H. MADRID- TORRELODONES (Isabel Llana Martín); H. PUERTA DE HIERRO (Carmen González Armengod); H.U. SANTA LUCIA DE CARTAGENA (Jose María Lloreda García); H. FUENLABRADA (Laura Domingo Comeche); H.G. DE CATALUÑA (Laura Castells Vilella); H. GUADALAJARA (María Pangua); H. MATERNO-INFANTIL TORRE CÁRDENAS (Javier Díez-Delgado Rubio); H. UNIVERSITARIO DE NAVARRA (Concepción Goñi Orayen); H.U. QUIRÓN; CLÍNICA CORACHÁN (Ma Dolores Muro Sebastian); COMPLEXO HOSPITALARIO UNIVERSITARIO DE A CORUÑA (Alejandro Avila-Alvarez); H. QUIRÓN MÁLAGA (Manuel Baca Cots); H. PUNTA DE EUROPA DE ALGECIRAS (Paula Martín- Mora Bermúdez); COMPLEJO HOSPITALARIO UNIVERSITARIO DE OURENSE (Alicia Sardina Ríos); H. DE BADAJOZ (Emilia María Martínez Tallo); H. GENERAL LA MANCHA CENTRO (David Lozano Díaz); H. COSTA DE LA LUZ; H. HM PUERTA DEL SUR (Isabel Llana); H.U. INFANTA ELENA (Lorena Patricia Peña González); H. U. SANT JOAN DE REUS; H. QUIRÓN SAGRADO CORAZÓN (Marisela Guido Ferrera); H. U. FUNDACIÓN JIMENEZ DÍAZ (Tamara Carrizosa Molina); H.U. PRÍNCIPE DE ASTURIAS DE ALCALÁ DE HENARES; H.C.U. DE VALLADOLID; CLÍNICA UNIVERSIDAD DE NAVARRA; H.U. SON LLÀTZER (Ana Filgueira Posse); H.C.U. VIRGEN DE LA ARRIXACA (Ana Martínez García-Cervantes); COMPLEJO HOSPITALARIO UNIVERSITARIO DE FERROL (Jose Luaces Gonzalez); COMPLEJO HOSPITALARIO DE JAÉN (Elisenda Hernández García).
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature.
Author information
Authors and Affiliations
Contributions
G.S.S. and L.G.G.G. developed the original research idea. G.S.S., M.S.R., and C.G.L. designed the protocol. G.S.S., M.S.R., B.F.C., and L.M.F. requested the data. C.G.L., S.L.V., R.P.A.L., and A.I.F. analyzed and interpreted the data. C.G.L. wrote the initial draft of the manuscript, which was critically revised by G.S.S., B.F.C., L.M.F., S.L.V., R.P.A.L., A.I.F., L.G.G.G., and M.S.R. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by “Comité de Ética de la Investigación del Principado de Asturias” with reference number 2022.586. Informed consent was obtained from all individual participants’ parents included in the SEN1500 database. Consent was obtained for the inclusion in the database and the use of the information collected for clinical studies. Permission for data analysis was obtained from the executive committee of the Spanish Neonatal Network SEN1500.
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Communicated by Daniele De Luca
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
González López, C., Solís Sánchez, G., Fernández Colomer, B. et al. Extrauterine growth restriction in very-low-birthweight infants: prevalence and concordance according to Fenton, Olsen, and INTERGROWTH-21st growth charts in a multicenter Spanish cohort. Eur J Pediatr 183, 4073–4083 (2024). https://doi.org/10.1007/s00431-024-05673-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-024-05673-6