Abstract
Identifying cardiovascular-related measures that track from early childhood into later ages may help inform early prevention targets for cardiovascular disease. In this study, the tracking of triglycerides (TG), high-density cholesterol (HDL-c), atherogenic coefficient (AC), waist circumference to height ratio (WC/Height), mean arterial pressure (MAP), and homeostatic model assessment of insulin resistance (HOMA-IR) was examined in the INMA-Asturias cohort between 4 and 8 years of age. The analysis was conducted in 307 children who participated in the INMA-Asturias cohort (Spain) at 4 and at 8 years of age. Quantile regression models were used to evaluate tracking between measures at both ages, with each measure at 8 years as the dependent variable and the rank transformation of the same measure at 4 years as the independent variable. We found a positive association between HDL-c rank at 4 years and higher quantiles of the HDL-c distribution at 8 years, with an increase of 2.93 mg/dL (95% CI: 1.98, 3.87) per decile in the 0.9 quantile. A positive association was also found for WC/Height, with an increase of 0.008 (95% CI: 0.004, 0.012) per decile in the 0.9 quantile. We observed that tracking for AC increased in the higher quantiles of the distribution at 8 years, with an increase of 0.11 (95% CI: 0.09, 0.14) in the 0.6 quantile compared to an effect of 0.15 (95% CI: 0.09, 0.21) in the 0.9 quantile.
Conclusions: Adult markers of dyslipidemia and central obesity tracked between ages 4 and 8 years. For AC, tracking increased in the higher quantiles of the distribution.
What is Known: • Atherosclerosis begins in early life, so preventive efforts that start in childhood may delay progression to clinical disease. Determine what cardiovascular risk factors track into time since childhood bring the opportunity to identified those subjects at risk for later cardiovascular disease. • The study of risk factors in health populations and, particularly in children, copes with not clear and/or controversial thresholds definition. This makes it challenging to study tracking in pediatric ages. | |
What is New: • Quantile regression is a useful tool for assessing the tracking of risk factors for which there are no clinically meaningful thresholds. The increasing trend observed in the tracking of dyslipidemia suggests the possible difficulty that children with abnormal values at 4 years of age might have in normalizing them in future years. • The findings of this article may help to determine which cardiovascular-related measures could be screened and followed-up in children. |
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
Abnormal values of cardiovascular-related measures are frequently detected in adulthood [1] but may also be present in childhood [2]. This does not increase the risk of cardiovascular diseases (CVDs) in childhood itself; children rarely experience cardiovascular diseases and these occurrences are mainly caused by congenital heart problems or genetic syndromes [3]. Atherosclerosis, one of the main CVD triggers [4] in adults, is an accumulative process that can begin in childhood and youth [5,6,7]. Therefore, researchers have been trying to answer whether those subjects exposed to specific metabolic alterations in childhood will have higher risk of developing CVDs — or early CVDs — in adulthood [8].
The study of the association between underlying cardiovascular disease indicators in childhood and CVDs in adulthood has been challenging due to the difficulty of following a young sample the time required to observe in this population CVD events. Several studies have shown evidence that CVDs are associated with childhood metabolic alterations [9]. For instance, a study of 38,589 participants aged 3 to 19 years from the USA, Finland, and Australia found an association between body mass index (BMI), systolic blood pressure, triglycerides, and cholesterol with cardiovascular events in midlife [10], and strongest associations with these factors in aggregate. These findings can be explained by a risk accumulation model, in which risk factors present at each life stage further increase risk in adulthood; a risk chain model, in which risk in childhood is mediated by risk in adulthood; or a sensitivity period model, in which exposure at a particular time in life course confers more risk compared with other stages [9, 11]. Under either scenario, identifying metabolic alterations that are more likely to track from childhood into future years will help inform targets of early prevention.
One of the main difficulties in tracking metabolic disorders in children is the disorder definition itself. The lack of adequate studies linking cardiovascular risk factors in childhood to disease in adulthood leaves pediatric definitions of metabolic disorders based on the distribution of cardiovascular measures in generally healthy children [12]. Therefore, thresholds are controversial. One approach is to model measures continuously, and rely on categorization only for clinical diagnosis [13, 14]. Tracking studies — defined as the maintenance over time of a relative position in the distribution of a variable [15] — have been analyzed mainly using thresholds to categorize the variables of interest, and stratify subjects into risk groups [16]. To avoid that, we propose to study the tracking of the rank values instead of the values themselves. That is, we aim to study whether the subjects with higher values at 4 years still have higher values at 8 years in terms of the variable distribution. With this goal, we consider data from the Infancia y Medio Ambiente (INMA)-Asturias cohort [17] and use quantile regression models [18]. This methodology allows to estimate the effect of an explanatory variable on any quantile of the outcome distribution, permitting the analysis of extreme values of the outcome without setting arbitrary thresholds [19].
For this reason, we aimed to apply this approach to assess whether having extreme values in the cardiovascular-related measures at 4 years is associated with having extreme values in the same cardiovascular-related measures at 8 years. We consider the following measures: triglycerides (TG), high-density cholesterol (HDL-c), atherogenic coefficient (AC), waist circumference to height ratio (WC/Height ratio), mean arterial pressure (MAP), and the homeostatic model assessment of insulin resistance (HOMA-IR).
Materials and methods
Study design
Study subjects were children participating in the INMA (Infancia y Medio Ambiente [Environment and Childhood]) Asturias cohort (north of Spain). Details can be found in previous studies [20, 21]. Briefly, between May 2004 and June 2007, pregnant women in their first trimester of pregnancy were recruited at the San Agustín University Hospital (Avilés) following a common protocol [17]. This hospital is a public health center with 436 beds which provides primary care and central, medical, and surgical services to a population of 144,875 inhabitants according to 2021 census [22]. The inclusion criteria were maternal age ≥ 16 years, singleton pregnancy, delivery scheduled at the referenced hospital, no assisted conception, and no communication handicap. Data were collected by trained professionals in several phases of follow-up: at first and third trimester of pregnancy, at birth, and at children’s ages 18 months, 4, and 8 years. Information was collected by medical registries, interview-based questionnaires with mothers, blood sample collection, and physical examinations of the children conducted by trained staff.
Cardiovascular-related measurements
For this study we focused on cardiovascular-related measures that reflect well-established CVD risk factors in adulthood: central obesity, insulin resistance, dyslipidemia, and hypertension. These included WC/Height ratio for central obesity [23]; MAP for hypertension [24]; TG, HDL-c, and AC for dyslipidemia [25]; and HOMA-IR for insulin resistance [26].
Lipids
Lipids were measured at 4 and 8 years collecting non-fasting blood samples, obtained by antecubital venipuncture. Serum total cholesterol (T-c), TG, HDL-c, and low-density cholesterol (LDL-c) levels were determined using a Roche analyzer (Modular Analytics Serum Work Area, Mannheim, Germany). AC was calculated as the difference between T-c and HDL-c, divided by HDL-c. Lipids values are presented in milligrams per deciliter (mg/dL).
Anthropometry
At 4 and 8 years, trained staff measured children height and WC. Height was measured twice to the nearest 0.1 cm using a wall-mounted stadiometer after the participant removed their shoes. Waist circumference was measured to the nearest 0.1 cm at the children midpoint between the right lower rib and the iliac crest at the level of the umbilicus, using an inelastic nylon tape in a horizontal plane, and with children in a standing position. WC/Height ratio was calculated as waist circumference in cm divided by height in cm.
Blood pressure
Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured using an automated oscillometric system (OMRON®) at children 4 and 8 years. After a 5-min rest period, between two and three consecutive measurements were taken, with children in a seated position and their right arm at rest at the heart level. The SBP and DBP averaged paired values were used and MAP was calculated as DBP + 1/3(SBP − DBP) [27]. Values are presented in millimeter of mercury (mmHg).
Blood glucose and insulin
Blood glucose and insulin levels were determined using the same Roche analyzer at children 4 and 8 years through collecting non-fasting blood samples, obtained by antecubital venipuncture. Glucose values are presented in milligrams per deciliter (mg/dL) and insulin values in microunits per milliliter (µU/mL). HOMA-IR was calculated as glucose multiplied by insulin and divided by 405.
Potential confounding factors
The following parental characteristics were selected as potential confounders: maternal age at enrollment, maternal pre-pregnancy (BMI), paternal BMI, maternal educational level, maternal social class, maternal smoking during pregnancy, and parental CVD antecedents (neither parent has antecedents/one parent has at least one antecedent/both parents have at least one antecedent). Regarding pre-pregnancy BMI, the maternal height and pre-pregnancy weight were self-reported, both at the first-trimester visit. These values were used to calculate the pre-pregnancy BMI (in kg/m2). Paternal weight and height were reported by the mother at the first-trimester visit and were used to calculate paternal BMI. Questionnaires administered during the first and third trimester of pregnancy obtained information on maternal and paternal age and education, maternal country of birth, maternal and paternal occupation, and maternal smoking during pregnancy. Social class was defined according to the occupation during pregnancy of the mother or father, using a widely used Spanish adaptation of the International Standard Classification of Occupations coding system [28]. Parental CVD antecedent’s variable was reported by the mother in the first trimester of pregnancy. She was asked whether she or the father had been diagnosed with diabetes, heart disease, coagulation disorders, hypertension, or hypercholesterolemia and the responses were combined to create a categorical variable according to whether neither parent had any of them, whether one parent had at least one of them, or whether both parents had at least one of them. Children characteristics selected as potential confounders were age, height, weekly out-of-school physical activity time, and the mean of the daily energy intake. All of them were collected at the 4- and 8-year follow-ups. Week of gestation at delivery, birth weight, predominant breastfeeding duration, and sex were also considered. These information were collected from medical records, except for data on predominant breastfeeding duration, which were collected when the children were approximately 6 and 14 months old through questionnaires. Weekly out-of-school physical activity time was self-reported by mothers. The mean of the children daily energy intake was calculated based on validated food frequency questionnaires (FFQs) about children’s diet that were administered twice to the parents or care-givers of children over a 9-month period at 4 years, and over a 9–12-month period at 8 years. The FFQs were composed by 105 items at 4 years and by 46 items at 8 years. To explore the reproducibility of the FFQs, the nutrient and food group intake collected from the both FFQs at each age were compared, while validity was examined by contrasting the nutrient values from the FFQs and the average of three 24-h dietary recalls taken in this period, and also with the concentration of several vitamins in the blood (carotenoids, vitamin D, and α-tocopherol) [29, 30]. Nutrient values and total energy intake were calculated based on the US Department of Agriculture’s food composition tables and other published national sources. All questionnaires were conducted face-to-face by trained interviewers. The selection of these variables as potential confoundings was based on previous studies.
Study population
Initially, 494 eligible women agreed to participate and, at birth time, 485 children were part of the study. At 4 years, 453 children continued in the follow-up and 91.4% of them attended to this follow-up visit. At 8 years, 416 children continued in the follow-up and 87.0% of them assisted to the follow-up visit. We limited the study to those 416 children who continued in the study at the 8-year follow-up. Of these 416 children, there were 392 with some measure of anthropometry, blood pressure, lipids, or glucose/insulin at 4 years and 362 at 8 years. Only 154 children had measurements of all variables involved in the study at 4 and 8 years. To optimize the use of the available information, we only excluded from the final sample those children who, for at least one of the cardiovascular-related measures involved in the study, had no data at neither 4 nor 8 years. The final sample was composed by 307 children. Figure 1 shows the flowchart of the study sample and the resulting sample size.
Statistical analysis
Continuous variables were summarized by medians and interquartile ranges, and categorical variables by absolute and relative frequencies.
Crude and adjusted quantile regression models were performed to evaluate the association between the distribution of each cardiovascular-related measure at 8 years as the dependent variable, and the rank transformation — replacing the data by their corresponding ranks — of the same cardiovascular-related measure at 4 years as the independent variable. Quantile regression is a method used to evaluate the effects of exposures on the distribution of a continuous outcome [31]. It allows to assess whether the association between them differs for high-risk subjects (i.e., those at highest quantiles of the outcome) than for average subjects in the outcome distribution. To describe the effect of the independent variable on the cardiovascular-related measure distribution at 8 years, quantile sequence was estimated from 0.1 to 0.9. The models were fully adjusted with potentially confounding variables described in the “Potential confounding factors” section. Models were computed using the quantreg R package (version 5.94) [32] and standard errors were estimated using the “xy-pair” bootstrap method. To facilitate the interpretation of the regression parameters, the variables resulting from the rank transformation were expressed as percentiles. The reported parameters represent the effect on each quantile of the dependent variable of a 10-unit increase (1-decile) in the independent variable. The analysis was repeated with each cardiovascular-related measure at 8 years as the dependent variable, and the rank transformation of the same cardiovascular as the independent variable, but also including as independent variables the rank variables of other five remaining cardiovascular-related measures, adjusted by covariates (referred as the complete model in following sections).
All the analyses were performed after missing value multiple imputation in all the cardiovascular-related measures and the adjustment variables [33]. Under the missing at random (MAR) assumption, that our data suggest that this may be plausible, we applied multivariate imputation by chained equation (MICE) method with fully conditional specification using the mice R package (version 3.14.0) [34]. The results were pooled using Rubin’s combination rules [35].
The criteria used to select the final sample resulted in groups of children at 4 and 8 years with a complex structure between them, combining both independent and related measures. To test hypotheses about difference in means or proportions between these groups maintaining the original structure of the data and their relations, we use the general bootstrap algorithm (gBA) for hypothesis testing [36].
All the analyses were conducted using the R statistical software, version 4.2.1 (R Project for Statistical Computing). Statistical significance was considered at p-value < 0.05.
Results
Following the inclusion criteria mentioned in the “Study population” section, a total of 303 children with data on at least one of the cardiovascular-related measures at 4 years were included in the analysis, and a total of 292 children who meet the same criterion were included at 8 years. The merging of these two subsamples results in total sample of 307 children (Table 1). There were 288 children present in both subsamples at the same time, 15 children had only in the 4-year but not in the 8-year subsample, and 4 children had only 8-year data but not 4 years. Due to the small difference between the subjects in each sample, there are no relevant differences in not age-related characteristics. Maternal median age at delivery was 32.9 and 33.1 years at each subsample, respectively, and more than 96% of the mothers were from Spain. Overall, 33.0% of mothers and 66.8% of fathers in the 4-year subsamples, and 32.9% of mothers and 66.2% of fathers in the 8-year subsamples, were overweight or obese (BMI ≥ 25 kg/m2). The average daily energy consumed increased from age 4 to age 8 (a median of 1618 and 1753 cal, respectively; p-value = 0.001), and the number of weekly hours of physical activity outside school was considerably reduced from 4 to 8 years (a median of 11.5 and 3.00 h, respectively; p-value < 0.001). This decrement is explained because at age 4, parents reported an average of 8.3 h per week of playing at home or in playground, and this activity is no longer reported at 8 years. Table 2 contains the summary of anthropometric, serum lipids, blood pressure, and glucose and insulin variables in the 4- and 8-year subsamples.
Additional file 1 shows the percentage of imputed data for each variable over the overall sample. Lipids, glucose, and insulin measurements had the highest percentage of missing data (ranging from 19.2 to 25.1% at 4 years and from 19.9 to 23.1% at 8 years). Missing data in anthropometric and blood pressure measurements ranged from 2.0 to 6.2%.
Triglycerides
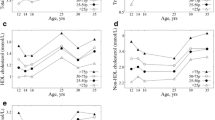
Figure 2 shows the estimated quantile regression parameters for each rank cardiovascular-related measure at 4 years, on the distribution of the same cardiovascular-related measure at 8 years, for all quantiles. We observe a positive association between TG rank at 4 years and TG distribution at 8 years above 0.5 quantile. The magnitude of the association was stronger in the upper part of the distribution: 1-decile increase in child’s rank at 4 years related to an increase of 2.28 mg/dL (95% CI: 0.13, 4.43) in the 0.6-TG quantile at 8 years compared to an increase of 5.82 mg/dL (95% CI: 1.00, 10.65) in the 0.9-TG quantile at 8 years (Additional file 2). In the complete model (Fig. 3), 1-decile increase at 4 years has an increase effect of 1.77 mg/dL (95% CI: − 0.68, 4.23) in the 0.6 quantile compared to a 2.47-mg/dL (95% CI: − 0.88, 5.83) increase effect in the 0.75 quantile (Additional file 3).
Quantile regression models with cardiovascular-related measure at 8 years as dependent variable and the rank variable of the corresponding cardiovascular-related measure at 4 years as the independent variable, for the quantiles between 0.1 to 0.9, with increments of 0.05, adjusted for maternal age at delivery, maternal level of education, maternal social class, maternal smoking during pregnancy, maternal pre-pregnancy body mass index, paternal body mass index, parental cardiovascular antecedents, child sex, child mean daily energy intake at 4 and 8 years, child weekly out-of-school physical activity time at 4 and 8 years, week of gestation at delivery, weeks of predominant breastfeeding, and child height at 4 and 8 years. Coefficient estimated are calculated with the independent variables in terms of percentiles and they represent the effect on the dependent variable for each 1-decile increase in the independent variable. They are expressed in terms of number of standard deviations of the dependent variable to homogenize the Y-axis scales
Quantile regression models with each cardiovascular-related measure at 8 years as dependent variable and the rank of all the cardiovascular-related measures at 4 years as the independent variables, for the quantiles 0.60 and 0.75, adjusted for maternal age at delivery, maternal level of education, maternal social class, maternal smoking during pregnancy, maternal prepregnancy body mass index, paternal body mass index, parental cardiovascular antecedents, child sex, child mean daily energy intake at 4 and 8 years, child weekly out-of-school physical activity time at 4 and 8 years, week of gestation at delivery, weeks of predominant breastfeeding, and child height at 4 and 8 years. Coefficient estimated are calculated with the independent variables in terms of percentiles and they represent the effect on the dependent variable for each 1-decile increase in the independent variable. They are expressed in terms of number of standard deviations of the dependent variable to homogenize the X-axis scales
High-density lipoprotein cholesterol
The association between HDL-c rank at 4 years and HDL-c distribution at 8 years was positive across all quantiles (Fig. 2). A gradual increase was observed in the upper part of the distribution with an increase of 2.68 mg/dL (95% CI: 1.75, 3.62) for the 0.6 HDL-c quantile at 8 years and of 2.93 mg/dL (95% CI: 1.98, 3.87) for the 0.9 HDL-c quantile at 8 years (Additional file 2). Associations in the complete model (Fig. 3) were lower than in the individual model but the overall trends were similar.
Atherogenic coefficient
The association between the AC rank at 4 years and AC distribution at 8 years also was positive (Fig. 2). The size of increase was greater at the highest part of the AC distribution (an increase of 0.11; 95% CI: 0.09, 0.14) in the 0.6 quantile vs. an effect of 0.15 (95% CI: 0.09, 0.21) in the 0.9 quantile (Additional file 2). Results were similar in the complete model (Fig. 3; Additional file 3).
Waist circumference to height ratio
A positive association was observed between WC/Height rank at 4 years and WC/Height distribution at 8 years (Fig. 2). The crude model shows a positive trend in the effect size as the quantile increases, evidenced by the positive slope of the plot in Fig. 2 (effect of 0.010 (95% CI: 0.006, 0.013) on the 0.6 WC/Height quantile at 8 years vs. an effect of 0.014 (95% CI: 0.010, 0.018) on the 0.9 WC/Height quantile at 8 years; Additional file 2). In the adjusted model, the effect sizes were smaller and generally constant in all quantiles (increase of 0.007 (95% CI: 0.005, 0.010) in the 0.6 WC/Height quantile vs. an increase of 0.008 (95% CI: 0.004, 0.012) in the 0.9 WC/Height quantile at 8 years; Additional file 2). This difference in the trend of the crude and adjusted model is mainly due to the adjustment for maternal BMI and educational level. The complete model (Fig. 3) produced similar results.
HOMA-IR
No clear association was found between HOMA-IR rank at 4 years and HOMA-IR distribution at 8 years (Fig. 2) (effect size of 0.037 (95% CI: − 0.058, 0.131) in the 0.6 HOMA-IR quantile vs. 0.067 (95% CI: − 0.312, 0.445) in the 0.9 HOMA-IR quantile at 8 years; Additional file 2). This also was seen in the complete model (Fig. 3).
Mean arterial pressure
The association of MAP rank at 4 years on MAP distribution at 8 years was positive, but only statistically significant between 0.3 and 0.6 quantiles (Fig. 2). Similar results were observed in the complete model (Fig. 3).
Discussion
This study found a positive association between the relative position of children at 4 years in the HDL-c, AC, and WC/Height distributions and all the quantiles of the same variable at 8 years. For TG distribution, it was found a positive association between the relative position at 4 years and the quantiles above 0.5 at 8 years, but which is not observed in the model adjusted for the rest of the cardiovascular-related measures. The stronger associations in the upper parts of the distribution in terms of standard deviations of each variable were found for HDL-c and AC outcomes. For AC, the more extreme the children’s values at 8 years, the greater the effect of the association. This trend is also observed for TG, although the effect is not statistically significant. No conclusive association was found for either HOMA-IR or MAP outcomes.
Our findings for HDL-c and AC suggest serum lipid track among children between the ages of 4 and 8 years. These results are in line with those found in children from two different Japan rural areas: one area reported tracking of serum lipids (specifically in T-c, HDL-c, and AC) between 8 and 12 years [37] and the other reported strong T-c tracking in children aged 6–7 after 9 years of follow-up [38]. Previously, The Muscatine Study observed T-c and TG tracking in children between 5 and 12 years after follow-ups of 2, 4, and 6 years [39]. The Bogalusa Heart Study showed tracking of serum lipids in 5-year-old children after a follow-up of 9 years [40]. These studies categorized serum lipids and evaluated which percentage of children remained in the highest category after the follow-up, which does not allow to observe differences in tracking within the highest-risk category itself. Using our quantile regression approach, we were able to observe that for AC the magnitude of tracking was stronger as the relative position at 8 years of age increases, showing the possible difficulty that children with abnormal values at 4 might have in normalizing them in future years. This is remarkable because AC (the ratio of non-HDL-c to HDL [25]) is clearly related to higher risk of CVDs in adulthood [41, 42]. Whereas higher HDL-c had similar tracking levels in all quantiles and higher HDL-c has unclear association with CVD risk [43].
Tracking of measures related to obesity have been widely studied in childhood and adolescence. The majority of studies use BMI as a marker of obesity [37, 44,45,46,47,48]. Increasingly studies are focusing on other anthropometric measures such as WC, WC/Height ratio, or skinfolds [49,50,51] and report the presence of tracking, consistent with our findings, in a variety range of ages but mostly between childhood and adolescence. Some of these studies find tracking among the same age ranges that were considered in this study [46, 48, 51], although the evaluation approach makes difficult to compare effect magnitudes. Hayes et al. [46] evaluated tracking of BMI in seven follow-up visits between 2–3 and 16–17 years and reported that the tracking magnitude was lower between 2 and 7 years than at later ages. A meta-regression analysis reported stronger BMI tracking after the age of 7 [52]. This suggests steeper tracking of central obesity than observed in our study for children of older ages. In the crude model, we observed higher tracking in children with a high-risk position in the distribution at 8 years. But when we adjusted for maternal pre-pregnancy BMI, and maternal educational level, the association became very similar across all the quantiles of the distribution: higher maternal BMI is associated with a tracking increasing effect in the highest quantiles, and higher maternal educational level is associated with a decreasing effect in the highest quantiles. Other studies reported similar findings with maternal pre-pregnancy BMI and measures related with obesity, and with blood pressure as well [53]. We only have observed this effect in measures related with obesity. Several studies have reported the influence of socioeconomical inequalities in BMI tracking, using parental educational level [54], parental socioeconomic position [46], or a combination of parental educational level, household income, and occupation [48]. We have observed this effect with maternal educational level but not with socioeconomic status.
Neither the MAP nor HOMA-IR showed a relevant association between the relative position of children at age 4 years and the relative position at age 8 years in the upper part of the distribution. As with serum lipids, there are few studies that analyzed the tracking of these measures in childhood and adolescence, rather than in adulthood. In our study, MAP is used as a blood pressure index to try to capture the effect of systolic and diastolic pressure using a single measurement. However, we also examined SBP and DBP separately (data not shown), yielding identical findings and conclusions as those obtained using MAP. Existing studies predominantly employ SBP and DBP and most of them report weak or poor blood pressure tracking [37, 39, 55], in line with the results we obtained here. One exception is the study by Sánchez-Bayle et al. [56] that reported considerably higher level of blood pressure tracking in a school-aged population. For measures related to insulin resistance, two studies examined tracking between 8 and 21 years [57], and between 10 and 17 years [58], with disparate results. Joshi et al. [57] reported moderate tracking of the HOMA-IR but no tracking of fasting insulin or glucose measures, while Wang et al. [58] reported no tracking in the HOMA-IR but tracking of fasting glucose. It should be pointed that in longitudinal studies a change in the behavior of the subjects can arise based on the knowledge of the results and the recommendations given in this regard, influenciating the tracking effect. Despite the limitation this may imply, these recommendations make it possible to reverse trends that would be more difficult in adulthood.
Numerous studies have reported interrelations between cardiovascular risk measures including markers of obesity, blood pressure, insulin resistance, and lipids, although these relations and the pathways explaining them are not clear yet [59, 60]. Therefore, in our study, the analyses were repeated including the cardiovascular-related measures as independent variables altogether in our models to observe whether any were acting as confounding factors for each other. The results of the individual analysis were remained, although with a general attenuation of the magnitude of the effects.
To our knowledge, this is among the first studies to address the analysis of cardiovascular measure tracking in children using quantile regression. Only one prior study conducted the analysis of BMI tracking between childhood and adulthood using this statistical technique [54]. This approach allows the introduction of several adjustment variables and exploration of the effects of different variables at the same time, as well as avoids using thresholds, always controversial in pediatric ages. Among other strengths of this study is that by using quantile regression and the independent variables in terms of their ranges allows to control for age-dependent variation in the effects observed for measures as HDL-c or TG.
This study has also some limitations. The sample size is moderate/small, with the consequent loss of power in the analysis. This is an exploratory study, in which numerous hypotheses are tested (different quantiles and different results), so that multi-testing problems arise, making problematic to calculate the statistical power of the study. Some of the variables were self-reported by the children’s parents. Other variables that have shown to influence the associations evaluated here have not been included such as children diet quality [61], maternal diet quality during pregnancy and breastfeeding [62], or pre-eclampsia [63]. It should also be noted that blood samples were collected under non-fasting conditions. It is unclear to what extent glucose and insulin levels might be biased due to prior caloric intake [64]. However, in non-diabetic subjects, the HOMA-IR would not be expected to show large variations. If blood glucose is higher due to previous intake, insulin also raises its levels, and therefore, the ratio between them will be similar. No blood glucose levels suggestive of diabetes have been detected in our sample, so we expect the HOMA-IR to be similar to that under fasting conditions. Regarding to lipids, the use of non-fasting samples is already recommended, except in unusual cases that do not apply to our sample [65,66,67]. On the other hand, most studies evaluate tracking between longer periods, and extreme values of cardiovascular-related measurements in childhood have shown to have an age-dependent impact on adult cardiovascular health, being predictive of subclinical atherosclerosis from the age of 9 [68]. Yet, it is still relevant to know the tracking of these measures throughout early childhood and adolescence more as a continuum, as long-term effect of childhood exposures on adult health is likely cumulative [11].
Conclusions
Our study found tracking between 4 and 8 years of age at the highest quantiles of the distribution of cardiovascular-related measures established as adult markers of dyslipidemia and central obesity (specifically for HDL-c and AC for dyslipidemia and WC/Height ratio for central obesity). The results indicated that for AC distribution tracking appears to be stronger at higher quantiles, suggesting the difficulty of normalizing their extreme values. These findings can help determine what cardiovascular-related measures could be the targets of screening and monitoring in children.
Availability of data and materials
The data and computing code are available for replication from the corresponding author on reasonable request.
Abbreviations
- AC:
-
Atherogenic coefficient
- BMI:
-
Body mass index
- CVDs:
-
Cardiovascular diseases
- DBP:
-
Diastolic blood pressure
- gBA:
-
General bootstrap algorithm
- HDL-c:
-
High-density cholesterol
- HOMA-IR:
-
Homeostatic model assessment of insulin resistance
- LDL-c:
-
Low-density cholesterol
- MAP:
-
Mean arterial pressure
- MAR:
-
Missing at random
- MICE:
-
Multivariate imputation by chained equations
- SBP:
-
Systolic blood pressure
- T-c:
-
Total cholesterol
- TG:
-
Triglycerides
- WC:
-
Waist circumference
- WC/Height:
-
Waist circumference to height ratio
References
Abbafati C, Abbas KM, Abbasi-Kangevari M, et al (2020) Global burden of 87 risk factors in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease Study 2019. Lancet 396:1223–1249. https://doi.org/10.1016/S0140-6736(20)30752-2
Genovesi S, Giussani M, Orlando A, et al (2019) Prevention of cardiovascular diseases in children and adolescents. High Blood Press Cardiovasc Prev 26:191–197. https://doi.org/10.1007/s40292-019-00316-6
Vetter VL, Covington TM, Dugan NP, et al (2015) Cardiovascular deaths in children: General overview from the National Center for the Review and Prevention of Child Deaths. Am Heart J 169:426-437.e23. https://doi.org/10.1016/j.ahj.2014.11.014
Zanchetti A, Bond MG, Hennig M, et al (1998) Risk factors associated with alterations in carotid intima—media thickness in hypertension: baseline data from the European Lacidipine Study on Atherosclerosis. J Hypertens 16:949–61. https://doi.org/10.1097/00004872-199816070-00008
Milei J, Ottaviani G, Lavezzi AM, et al (2008) Perinatal and infant early atherosclerotic coronary lesions. Can J Cardiol 24:137–41. https://doi.org/10.1016/s0828-282x(08)70570-1
Mcgill HC, Mcmahan ; C Alex, Herderick EE, et al (2000) Origin of atherosclerosis in childhood and adolescence. Am J Clin Nutr 72:1307S-1315S. https://doi.org/10.1093/ajcn/72.5.1307s
Erald G, Erenson SB, Rinivasan ARS, et al (1998) Association between multiple cardiovascular risk factors and atherosclerosis in children and young adults. N Engl J Med 338:1650–1656. https://doi.org/10.1056/NEJM199806043382302
De Ferranti SD, Steinberger J, Ameduri R, et al (2019) Cardiovascular risk reduction in high-risk pediatric patients: A scientific statement from the American Heart Sssociation. Circulation 139:E603–E634. https://doi.org/10.1161/CIR.0000000000000618
Pool LR, Aguayo L, Brzezinski M, et al (2021) Childhood risk factors and adulthood cardiovascular disease: A systematic review. J Pediatr 232:118-126.e23. https://doi.org/10.1016/j.jpeds.2021.01.053
Jacobs DR, Woo JG, Sinaiko AR, et al (2022) Childhood cardiovascular risk factors and adult cardiovascular events. N Engl J Med 386:1877–1888. https://doi.org/10.1056/NEJMoa2109191
Ben-Shlomo Y, Mishra G, Kuh D (2014) Life course epidemiology. Handb Epidemiol Second Ed 1521–1549. https://doi.org/10.1007/978-0-387-09834-0_56
Lurbe E, Agabiti-Rosei E, Cruickshank JK, et al (2016) 2016 European Society of Hypertension guidelines for the management of high blood pressure in children and adolescents. J Hypertens 34:1887–1920. https://doi.org/10.1097/HJH.0000000000001039
Reisinger C, Nkeh-Chungag BN, Fredriksen PM, Goswami N (2020) The prevalence of pediatric metabolic syndrome—a critical look on the discrepancies between definitions and its clinical importance. Int J Obes 45:12–24. https://doi.org/10.1038/s41366-020-00713-1
Ahrens W, Moreno L, Mårild S, et al (2014) Metabolic syndrome in young children: Definitions and results of the IDEFICS study. Int J Obes 38:S4–S14. https://doi.org/10.1038/ijo.2014.130
Foulkes MA, Davis CE (1981) An index of rracking for longitudinal data. Biometrics 37:439-446. https://doi.org/10.2307/2530557
Twisk JWR (2003) The problem of evaluating the magnitude of tracking coefficients. Eur J Epidemiol 18:1025–1026. https://doi.org/10.1023/a:1026161919170
Guxens M, Ballester F, Espada M, et al (2012) Cohort profile: The INMA-INfancia y Medio Ambiente-(environment and childhood) project. Int J Epidemiol 41:930–940. https://doi.org/10.1093/ije/dyr054
Koenker R (2005) Quantile regression. Cambridge University Press, Quantile Regres. Cambridge
Waldmann E (2018) Quantile regression: A short story on how and why. Stat Modelling 18:203–218. https://doi.org/10.1177/1471082X18759142
Fernández-Somoano A, Estarlich M, Ballester F, et al (2011) Outdoor NO2 and benzene exposure in the INMA (Environment and Childhood) Asturias cohort (Spain). Atmos Environ 45:5240–5246. https://doi.org/10.1016/j.atmosenv.2011.02.010
Fernández-Somoano A, Tardon A (2014) Socioeconomic status and exposure to outdoor NO2 and benzene in the Asturias INMA birth cohort, Spain. J Epidemiol Community Health 68:29-36. https://doi.org/10.1136/JECH-2013-202722
Sociedad Asturiana de Estudios Económicos e Industriales (2021) Actualización demográfica del Mapa Sanitario de Asturias. Available from: https://www.astursalud.es/documents/35439/37169/Mapa_Padron_2021_Libro.pdf/0c9cf850-f5d4-9292-3fb1-9fa26d449276
Mokha JS, Srinivasan SR, DasMahapatra P, et al (2010) Utility of waist-to-height ratio in assessing the status of central obesity and related cardiometabolic risk profile among normal weight and overweight/obese children: The Bogalusa Heart Study. BMC Pediatr 10:1–7. https://doi.org/10.1186/1471-2431-10-73
Sesso HD, Stampfer MJ, Rosner B, et al (2000) Systolic and diastolic blood pressure, pulse pressure, and mean arterial pressure as predictors of cardiovascular disease risk in Men. Hypertens (Dallas, Tex 1979) 36:801–807. https://doi.org/10.1161/01.HYP.36.5.801
Çelik E, Çora AR, Karadem KB (2021) The effect of untraditional lipid parameters in the development of coronary artery disease: atherogenic index of plasma, atherogenic coefficient and lipoprotein combined index. J Saudi Hear Assoc 33:244–250. https://doi.org/10.37616/2212-5043.1266
Yajnik CS, Katre PA, Joshi SM, et al (2015) Higher glucose, insulin and insulin resistance (HOMA-IR) in childhood predict adverse cardiovascular risk in early adulthood: the Pune Children’s Study. Diabetologia 58:1626–1636. https://doi.org/10.1007/s00125-015-3602-z
DeMers D, Wachs D (2022) Physiology, mean arterial pressure. StatPearls Publishing, StatPearls
Domingo-Salvany A, Regidor E, Alonso J, Alvarez-Dardet C (2000) Una propuesta de medida de la clase social. Atención Primaria 25:350–363. https://doi.org/10.1016/S0212-6567(00)78518-0
Vioque J, Gimenez-Monzo D, Navarrete-Muñoz EM, et al (2016) Reproducibility and validity of a food frequency questionnaire designed to assess diet in children aged 4-5 years. PLoS One 11:1–17. https://doi.org/10.1371/JOURNAL.PONE.0167338
Vioque J, Garcia-De-La-Hera M, Gonzalez-Palacios S, et al (2019) Reproducibility and validity of a short food frequency questionnaire for dietary assessment in children aged 7-9 years in Spain. Nutrients 11:1–15. https://doi.org/10.3390/NU11040933
Hao L, Naiman DQ (2007) Quantile regression. Quantile Regres. Thousand Oaks, Calif: Sage Publications
Koenker R (2022) Package ‘quantreg.’ Quantile Regres 1–349
Sterne JAC, White IR, Carlin JB, et al (2009) Multiple imputation for missing data in epidemiological and clinical research: potential and pitfalls. BMJ 338:b2393. https://doi.org/10.1136/bmj.b2393
van Buuren S, Groothuis-Oudshoorn K (2011) MICE: multivariate imputation by chained equations. J Stat Softw 45:1–67. https://doi.org/10.18637//jss.v045.i03
Heymans MW, Eekhout I (2019) Applied Missing Data Analysis with SPSS and (R)Studio. https://bookdown.org/mwheymans/bookmi/. Accessed 6 Nov 2022
Martínez-Camblor P, Corral N (2012) A general bootstrap algorithm for hypothesis testing. J Stat Plan Inference 142:589–600. https://doi.org/10.1016/j.jspi.2011.09.003
Tan F, Okamoto M, Suyama A, Miyamoto T (2010) Tracking of cardiovascular risk factors and a cohort study on hyperlipidemia in rural schoolchildren in Japan. J Epidemiol 10:255–261. https://doi.org/10.2188/jea.10.255
Osawa E, Asakura K, Okamura T, et al (2022) Tracking pattern of total cholesterol levels from childhood to adolescence in Japan. J Atheroscler Thromb 29:38–49. https://doi.org/10.5551/jat.59790
Clarke WR, Schrott HG, Leaverton PE, et al (1978) Tracking of blood lipids and blood pressures in school age children: The Muscatine study. Circulation 58:626–634. https://doi.org/10.1161/01.CIR.58.4.626
Freedman DS, Shear CL, Srinivasan SR, et al (1985) Tracking of serum lipids and lipoproteins in children over an 8-year period: the Bogalusa heart study. Prev Med (Baltim) 14:203–216. https://doi.org/10.1016/0091-7435(85)90036-2
Brunner FJ, Waldeyer C, Ojeda F, et al (2019) Application of non-HDL cholesterol for population-based cardiovascular risk stratification: results from the Multinational Cardiovascular Risk Consortium. Lancet (London, England) 394:2173–2183. https://doi.org/10.1016/S0140-6736(19)32519-X
McBride P (2008) Triglycerides and risk for coronary artery disease. Curr Atheroscler Rep 10:386–390. https://doi.org/10.1007/s11883-008-0060-9
Casula M, Colpani O, Xie S, et al (2021) HDL in atherosclerotic cardiovascular disease: in search of a role. Cells 10:1–17. https://doi.org/10.3390/cells10081869
Freedman DS, Lawman HG, Galuska DA, et al (2018) Tracking and variability in childhood levels of BMI: the Bogalusa Heart Study. Obesity 26:1197–1202. https://doi.org/10.1002/OBY.22199
De Wilde JA, Middelkoop BJC, Verkerk PH (2018) Tracking of thinness and overweight in children of Dutch, Turkish, Moroccan and South Asian descent from 3 through 15 years of age: a historical cohort study. Int J Obes 42:1230–1238. https://doi.org/10.1038/s41366-018-0135-9
Hayes AJ, Carrello JP, Kelly PJ, et al (2021) Looking backwards and forwards: tracking and persistence of weight status between early childhood and adolescence. Int J Obes 45:870–878. https://doi.org/10.1038/s41366-021-00751-3
Toselli S, Brasili P, Di Michele R (2013) Tracking of weight status and body fatness in Italian children. Eat Weight Disord 18:383–388. https://doi.org/10.1007/S40519-013-0074-3
Wheaton N, Millar L, Allender S, Nichols M (2015) The stability of weight status through the early to middle childhood years in Australia: a longitudinal study. BMJ Open 5:1–9. https://doi.org/10.1136/BMJOPEN-2014-006963
Zvonar M, Štefan L, Kasović M, Piler P (2022) Tracking of anthropometric characteristics from childhood to adolescence: an 8-year follow-up findings from the Czech ELSPAC study. BMC Public Health 22:1–8. https://doi.org/10.1186/s12889-022-13178-w
Ochiai H, Shirasawa T, Nishimura R, et al (2020) Changes in overweight/obesity and central obesity status from preadolescence to adolescence: a longitudinal study among schoolchildren in Japan. BMC Public Health 20:1–7. https://doi.org/10.1186/s12889-020-8343-3
Ortiz-Marrón H, Ortiz-Pinto MA, Cabañas Pujadas G, et al (2022) Tracking and risk of abdominal and general obesity in children between 4 and 9 years of age. The Longitudinal Childhood Obesity Study (ELOIN). BMC Pediatr 22:1–11. https://doi.org/10.1186/s12887-022-03266-6
Bayer O, Krüger H, Von Kries R, Toschke AM (2011) Factors associated with tracking of BMI: a meta-regression analysis on BMI tracking. Obesity 19:1069–1076. https://doi.org/10.1038/oby.2010.250
Cox B, Luyten LJ, Dockx Y, et al (2020) Association Between Maternal Prepregnancy Body Mass Index and Anthropometric Parameters, Blood Pressure, and Retinal Microvasculature in Children Age 4 to 6 Years. JAMA Netw open 3:e204662–e204662. https://doi.org/10.1001/jamanetworkopen.2020.4662
Norris T, Bann D, Hardy R, Johnson W (2020) Socioeconomic inequalities in childhood-to-adulthood BMI tracking in three British birth cohorts. Int J Obes 44:388–398. https://doi.org/10.1038/s41366-019-0387-z
Sarganas G, Schaffrath Rosario A, Niessner C, et al (2018) Tracking of blood pressure in children and adolescents in Germany in the context of risk factors for hypertension. Int J Hypertens 2018:1–10. https://doi.org/10.1155/2018/8429891
Sánchez-Bayle M, Muñoz-Fernández MT, Gonzzález-Requejo A (1999) A longitudinal study of blood pressure in Spanish schoolchildren. Res Rep U S Nav Sch Aviat Med 81:169–171. https://doi.org/10.1161/01.cir.26.4.530
Joshi SM, Katre PA, Kumaran K, et al (2014) Tracking of cardiovascular risk factors from childhood to young adulthood - The Pune Children’s Study. Int J Cardiol 175:176–178. https://doi.org/10.1016/j.ijcard.2014.04.105
Wang G, Arguelles L, Liu R, et al (2011) Tracking blood glucose and predicting prediabetes in chinese children and adolescents: a prospective twin study. PLoS One 6:e28573. https://doi.org/10.1371/journal.pone.0028573
Alaqil AI, Petushek EJ, Gautam YR, et al (2022) Determining independence and associations among various cardiovascular disease risk factors in 9-12 years old school-children: a cross sectional study. BMC Public Health 22:1–9. https://doi.org/10.1186/s12889-022-14035-6
Al-Hamad D, Raman V (2017) Metabolic syndrome in children and adolescents. Transl Pediatr 6:397–407. https://doi.org/10.21037/TP.2017.10.02
Siddiqui NZ, Nguyen AN, Santos S, Voortman T (2022) Diet quality and cardiometabolic health in childhood: the Generation R Study. Eur J Nutr 61:729–736. https://doi.org/10.1007/s00394-021-02673-2
Castillo P, Kuda O, Kopecky J, et al (2022) Reverting to a healthy diet during lactation normalizes maternal milk lipid content of diet-induced obese rats and prevents early alterations in the plasma lipidome of the offspring. Mol Nutr Food Res 66:1–12. https://doi.org/10.1002/mnfr.202200204
Wojczakowski W, Kimber-Trojnar Ż, Dziwisz F, et al (2021) Preeclampsia and cardiovascular risk for offspring. J Clin Med 10:1–17. https://doi.org/10.3390/jcm10143154
Moebus S, Göres L, Lösch C, Jöckel KH (2011) Impact of time since last caloric intake on blood glucose levels. Eur J Epidemiol 26:719–728. https://doi.org/10.1007/s10654-011-9608-z
Nordestgaard BG, Langsted A, Mora S, et al (2016) Fasting is not routinely required for determination of a lipid profile: clinical and laboratory implications including flagging at desirable concentration cut-points—a joint consensus statement from the European Atherosclerosis Society and European Federa. Eur Heart J 37:1944–1958. https://doi.org/10.1093/EURHEARTJ/EHW152
Pearson GJ, Thanassoulis G, Anderson TJ, et al (2021) 2021 Canadian cardiovascular society guidelines for the management of dyslipidemia for the prevention of cardiovascular disease in adults. Can J Cardiol 37:1129–1150. https://doi.org/10.1016/j.cjca.2021.03.016
Grundy SM, Stone NJ, Bailey AL, et al (2019) 2018 AHA/ACC/AACVPR/AAPA/ABC/ACPM/ADA/AGS/APhA/ASPC/NLA/PCNA guideline on the management of blood cholesterol: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 73:3168–3209. https://doi.org/10.1016/j.jacc.2018.11.002
Juonala M, Magnussen CG, Venn A, et al (2010) Influence of age on associations between childhood risk factors and carotid intima-media thickness in adulthood. Circulation 122:2514–2520. https://doi.org/10.1161/CIRCULATIONAHA.110.966465
Mansournia MA, Collins GS, Nielsen RO, et al (2021) A Checklist for statistical Assessment of Medical Papers (the CHAMP statement): explanation and elaboration. Br J Sports Med 55:1009–1017. https://doi.org/10.1136/BJSPORTS-2020-103652
Vandenbroucke JP, Von Elm E, Altman DG, et al (2007) Strengthening the reporting of observational studies in epidemiology (STROBE): explanation and elaboration. PLoS Med 4:1628–1654. https://doi.org/10.1371/journal.pmed.0040297
Acknowledgements
The authors would particularly like to thank all the participants and the families for their valuable collaboration. They also like to thank the medical board, the gynecology and pediatrics services of Hospital San Agustín (Avilés, Asturias), and the health center of Las Vegas (Corvera, Asturias) for their generous implication in the project.
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. This study was supported by grants from CIBERESP (PhD-employment-contract and fellowship for short stays abroad-2022), ISCIII: PI04/2018, PI09/02311, PI13/02429, and PI18/00909 co-funded by the European Regional Development Fund (FEDER), “A way to make Europe”/ “Investing in your future”; Fundación Cajastur; and Universidad de Oviedo.
Author information
Authors and Affiliations
Contributions
Rocío Fernández-Iglesias: formal analysis, data curation, investigation, methodology, visualization, writing—original draft, writing—review and editing. Pablo Martínez-Camblor: formal analysis, conceptualization, investigation, methodology, supervision, visualization, writing—review and editing. Ana Fernández-Somoano: conceptualization, investigation, methodology, supervision, writing—review and editing. Cristina Rodríguez-Dehli: data collection, writing—review and editing. Rafael Venta-Obaya: resources, writing—review and editing. Margaret R. Karagas: conceptualization, writing—review and editing. Ana Fernández-Somoano: conceptualization, investigation, methodology, supervision, writing—review and editing. Adonina Tardón: conceptualization, supervision, project administration, funding acquisition, writing—review and editing. Isolina Riaño-Galán: conceptualization, data collection, writing—review and editing. All authors have read an agreed to the publisher version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study was conducted to conform to the principles of the Declaration of Helsinki and its protocol was approved by the Asturias Regional Ethics Committee. Informed consent was obtained from every participant woman and, in such case, her partner.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Peter de Winter
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Fernández-Iglesias, R., Martinez-Camblor, P., Fernández-Somoano, A. et al. Tracking between cardiovascular-related measures at 4 and 8 years of age in the INMA-Asturias cohort. Eur J Pediatr 182, 3893–3906 (2023). https://doi.org/10.1007/s00431-023-05051-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-023-05051-8