Abstract
Bronchiolitis causes a remarkable number of hospitalizations; its epidemiology follows that of respiratory syncytial virus (RSV), its main pathogen. The aim of this study was to evaluate the presenting features, treatment approach, and impact of medical therapy in four pediatric hospitals in Italy. Data on infants < 24 months of age hospitalized with bronchiolitis in the 2021–2022 season were collected. Between October 2021 and February 2022, 214 children were admitted. Median hospital stay was 5 days; none of the patients died. The distribution of the presenting features is largely comparable in the 33 (15.8%) RSV-negative versus the 176 (84.2%) RSV-positive children; also, no difference was observed in medical therapy provided: duration of oxygen therapy, administration of steroid, and duration of hospital stay. Systemic steroids, inhalation, or antibiotic therapy were given to 34.6%, 79.4%, and 49.1% of children respectively. Of the 214 patients with bronchiolitis, only 19 (8.8%) were admitted to ICU.
Conclusion: Our data suggest that, irrespective of treatments provided, RSV-positive and RSV-negative children had a similar clinical course. The results of our retrospective study further underline the need to improve adherence to existing guidelines on bronchiolitis treatment.
What is Known: • Bronchiolitis is a common diseases with seasonal peak. The outcome is usually favorable but hospitalization and even ICU admission is not exceptional. | |
What is New: • Children with RSV associated bronchiolitis do not have a different course and outcome. The analysis of the 2021-2022 cohort, following COVID pandemic peaking, did not show a different course and outcome. • Adherence to literature recommendation, i.e. to focus on oxygen and hydration therapy while avoiding unnecessary systemic therapy with steroid and antibiotics, should be improved. |
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Bronchiolitis is a clinical syndrome of respiratory distress occurring in young children, with an accepted cut-off age of less than 2 years. It is characterized by upper respiratory symptoms, such as rhinitis, soon followed by signs of lower respiratory tract infection and inflammation. Involvement of bronchiole results in wheezing and/or rales.
A viral pathogen, and only occasionally other agents, such as Mycoplasma pneumoniae, usually trigger bronchiolitis. Most children with non-severe bronchiolitis can be managed at home, with supportive care based on adequate hydration, relief of nasal congestion/obstruction, and monitoring for disease progression. Yet, bronchiolitis is a major cause of illness and hospitalization in infants and children younger than 2 years [1].
In children with severe bronchiolitis, hospitalization for supportive care and monitoring is mandatory [2, 3]. Despite being a common disorder, agreement on several features of diagnosis and management of bronchiolitis has not yet been achieved. Pharmacologic interventions are usually considered potentially inappropriate, inasmuch they lack proven benefit, increase the cost of care, and may have adverse effects. Potential benefit of bronchodilators (inhaled or oral) [4,5,6,7,8,9], glucocorticoids (inhaled or systemic) [10,11,12,13,14], or nebulized hypertonic saline (with or without nebulized epinephrine) [15] is not supported by reported trials and robust metanalyses. Management of severe bronchiolitis is based on stabilization of respiratory and fluid status, until criteria for admission in intensive care unit (ICU) are achieved.
Bronchiolitis is a self-limited illness and often resolves without complications in most previously healthy infants. Children born prematurely or those with associated cardiopulmonary disease or immune defect may have a higher risk to develop complications, such as respiratory failure or bacterial infection. Nevertheless, about 30% of previously healthy infants hospitalized for bronchiolitis have an increased risk for recurrent wheezing [16]. Mortality rate in children hospitalized with respiratory syncytial virus (RSV) bronchiolitis in developed countries is very low (less than 1 in 1000) [17,18,19].
Bronchiolitis typically spreads during the fall and winter [17]. The 2020–2021 epidemic arrived very late, and its impact was lower than in previous seasons, with a reduction in hospitalization in the range of 30%. This was considered to be linked to the implementation of non-pharmaceutical COVID-19 prevention measures [20, 21]. For 2021–2022, a novel increase of the incidence of bronchiolitis may be linked to an “immunity debt” resulting from the lower impact of the previous season [22, 23].
In this study, we aimed at evaluating the characteristic of the patients and therapeutic approach used in four Italian pediatric wards during the bronchiolitis epidemic of the season 2021–2022.
Materials and methods
Retrospective data were collected in four pediatric wards. One was a Pediatric Infectious Diseases ward, embedded in a Children’s Hospital, part of a teaching, third level hospital; three were Pediatric inpatient wards, allocated within second-level, general hospitals.
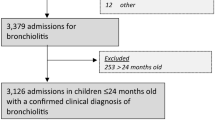
All children diagnosed with bronchiolitis, admitted to the participating centers between October 2021 and February 2022, were eligible for the study. Inclusion criteria were age ≤ 2 years, diagnosis of bronchiolitis, admission ending with ordinary discharge, and parental consent to treat the patient data for clinical research. Exclusion criteria were respiratory failure due to concurrent pulmonary, heart, or neuromuscular disorders, refusal of treatment, or voluntary discharge before treatment completion.
Samples for detection of RSV (and of other respiratory viruses or bacteria when obtained) were collected by disposable, nasal sterile swab or secretion aspirate and analyzed by PCR methods. Data collection included demographics, respiratory virus isolation, and details of treatment: oxygen administration, steroids, antibiotic drugs and need for ICU admission. The administration of systemic antibiotics or steroids was recorded.
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board (Comitato Etico per la Ricerca, Province di Chieti-Pescara; Verbale di seduta Comitato Etico, N. 07) on April 21, 2022. Patient consent was waived since data from patients were completely anonymized.
All the results were presented as mean and standard deviation (SD) or median and interquartile range (IQR) for continuous variables and as frequencies and percentages for categorical variables. Comparisons between groups were made through the t-test or the corresponding non-parametric Mann–Whitney test in case of non-normal distribution of continuous variables. In order to identify possible risk factors for admission to ICU, a multivariate analysis was run with the following variables: age, weight, and gender. The results were reported in terms of odds ratios with their relative 95% confidence intervals (95% CI). p-values < 0.05 were considered statistically significant. All the analyses were conducted with the R software (version 3.5.2).
Results
Between October 2021 and February 2022, the four participating centers diagnosed and treated 214 children with bronchiolitis, all eligible for the present study. Their main features are summarized in Table 1. The number of patients enrolled in each of the four centers ranged from 37 to 71. Of them, 57% were males and the median age was 3 months. Seventeen children had neonatal risk factors.
The median duration of the hospital stay was 5 days (IQR 4–8 days). None of the patients died. The main features of the 209/214 (97.7%) children in whom the RSV status was determined are summarized in Table 2.
The distribution of the presenting features was largely comparable in the 33 (15.8%) RSV-negative versus the 176 (84.2%) RSV-positive children. Duration of oxygen therapy and duration of hospital stay were comparable in the two groups.
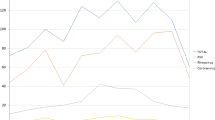
In 44 patients (20.6%), at least one pathogen other than RSV was reported. Of them, 16 were RSV-negative and were found positive for the following pathogens: SARS-CoV-2 (n = 3), metapneumovirus (n = 2), rhinovirus (n = 2), parainfluenza (n = 2), rhinovirus + bocavirus (n = 2), rhinovirus + coronavirus, bocavirus, hemophilus, not identified (n = 2); in the remaining 28 patients, who were positive for RSV, the following pathogens were also reported: rhinovirus (n = 7), bocavirus (n = 4), rhinovirus + pneumococcus (n = 1), SARS-CoV-2 (n = 2), adenovirus (n = 2), parainfluenza (n = 2), parainfluenza + pneumococcus (n = 1), pneumococcus (n = 9). Their distribution is summarized in Fig. 1.
Oxygen therapy was administered to 160 children (74.8%), in 91 (42.5%) by LFNC only, in 18 by HFNC only (8%), and in 51 (24%) by both; in the remaining 54 cases, oxygen therapy was not given. Duration of oxygen therapy was 7.0 (5.0–8.0) days in the 18 patients who received HFNC only, vs. 4.0 (2.5–5.0) days in the 91 who received LFNC only (p < 0.0001) (data not shown). The weaning procedure depended on individual clinical response and on local practices.
Inhaled and antibiotic therapies were administered to 170 (79.4%) and to 105 children (49.1%) respectively.
Parenteral steroid therapy (usually i.v. methyl-prednisolone) was given to 74 (34.6%) children. The proportion of cases receiving steroids was different among the four participating centers (p = 0.0006). Interestingly, children receiving steroid therapy were significantly older (p < 0.0001). Administration of steroids was not associated with any other presenting feature, including detection of RSV, while patients receiving steroids more frequently received antibiotic therapy (p = 0.027) and inhaled therapy (p < 0.001). Otherwise, duration of respiratory support and of hospital stay was not different in children receiving or not steroid therapy.
Of the 214 patients with bronchiolitis, only 19 (8.8%) were admitted to ICU (Table 3). Their proportion was variable, ranging between 2 and 17% in the four centers. In a univariate analysis, they were significantly younger (p = 0.0167), had a lower weight (p = 0.0086), had received inhaled therapy, high-flow oxygen therapy, steroids and any antibiotic therapy more often than patients who were not admitted to ICU. In the attempt to identify possible independent risk factors for admission to ICU, we run a multivariate analysis including age, weight, and gender. None of the above features turned out to be protective against risk of admission to ICU (Table 4).
Discussion
Despite being a common disease in infancy, agreement on several features of diagnosis and management of bronchiolitis has not yet been achieved [24]. In this study, we describe our experience in diagnosis and treatment of over 200 patients with bronchiolitis, admitted during the season 2021–2022 in four Italian pediatric centers, scattered in the north, center, and south of Italy.
The median age of 3 months and mild prevalence of male gender is not unexpected. Bronchiolitis usually affects previously healthy babies, as remarked by the small minority (8%) of them in whom an associated neonatal risk factor was reported. In far most cases, RSV was documented as the only pathogen. In the 44 cases in which at least one non-RSV pathogen was identified, rhinovirus was more frequently detected; pneumococcal colonization, frequently observed in infants with bronchiolitis, was probably of no clinical significance [25]. Since this analysis was not uniformly performed in all cases, this information has no epidemiological value.
Oxygen therapy is definitely the basis of treatment in bronchiolitis. In recent years the use of noninvasive ventilation therapies, such as the nasal continuous positive air pressure (CPAP), and the high-flow nasal cannula (HFNC), has emerged as alternatives to oro-tracheal intubation and conventional invasive ventilation in patients with moderate to severe bronchiolitis [26]. Possible use of HFNC outside ICU was considered of interest, also in the hope to reduce ICU requirement and the hospital time. In a retrospective analysis of over 500 children in a single center over 9 years, Solana et al. tried to address the issue of the contribution of the introduction of HFNC. They observed an increase in HFNC and, paradoxically, an increase in ICU transfers. The risk of failure (ICU transfer and respiratory support therapy escalation) was higher for those who required HFNC or CPAP for clinical stabilization in the first 12 h after admission. Moreover, the risk of failure was also higher in children with standard oxygen therapy promptly escalated to HFNC, especially if they had atelectasis/viral pneumonia, coinfections, or a history of prematurity. No correlation was found between the usage of HFNC and a shorter length of hospital stay or less time spent on oxygen therapy [27]. In another recent study, a web-based survey of major tertiary care hospitals in Canada, approximately half of tertiary children’s hospitals currently used HFNC on the ward and utilize a range of practices and policies, while other centers considered its implementation [28]. Further research is needed to inform best practices for HFNC therapy, support stewardship of health care resources, and promote safe patient care. Our study reflects the current trend in clinical practice but no correlation could be demonstrated between the usage of HFNC and a shorter length of hospital stay or less time spent on oxygen therapy.
The contribution of medical therapy for treatment of bronchiolitis remains controversial. In particular, the role of antibiotics and, even more, of steroids, did not reach a consensus. In this series, about one third of patients received steroid therapy. Although no systematic analysis on treatment decisions could be made by study design, we can only speculate that this is likely to depend on local or individual attitude of the attending physician, as suggested also by the range of variability of antibiotic administration, between 16 and 50%, in the four centers. We noted that steroid therapy was administered significantly more often to older patients, while was independent from detection of RSV infection. Since this was not a prospective, controlled trial aimed at exploring the contribution of systemic steroid therapy, we cannot draw any conclusion on any role of steroid therapy in sparing from ICU admission, duration of respiratory support or duration of hospital stay.
In a very recent randomized trial of corticosteroids and inhaled epinephrine for bronchiolitis in children in intensive care, 107 children received positive pressure support for a geometric mean of 26 h compared with 40 h in controls [29]. Yet, the accompanying editorial remarks some limitations of this study, which thus cannot be taken as a final evidence [30]. In our series, 79% of patients received inhaled therapy, independently of association with RSV. Accumulating evidence suggests that bronchiolitis is not a single disease but can have different “phenotypes,” based on age of presentation, history of atopy, etiology, and clinical presentation [31,32,33]. New studies aimed at defining the relationship between therapeutic options and different phenotypes appear warranted. In this frame, a bronchodilator-trial (using short-acting beta-2 agonists with metered-dose inhalers and valved holding chambers) has been proposed in children with bronchiolitis aged > 6 months [34].
The lack of evidence in favor of a specific treatment of bronchiolitis is probably the main reason for the frequent non-uniformity of therapeutic approach, also mirrored in our analysis. In a retrospective chart review of 146 infants with bronchiolitis requiring respiratory support in winter season 2015/2016 treated in two regions in Central Europe (France or Germany), infants in the German group received more frequently infusion therapy, more antibiotics, and more inhalation therapy. [35]. In a very recent retrospective analysis of the application of an Italian inter-society consensus document in 2014, when comparing patients admitted in the prior four epidemic seasons with patients admitted in the latter six epidemic seasons, a significant reduction in the prescription of systemic corticosteroids (58.9% vs. 41.8%, p < 0.001), nebulized epinephrine (73.8% vs. 38.3%, p < 0.001), and antibiotics (59.5% vs. 42.3%, p < 0.001) was observed, together with a drastic decrease in the use of chest X-ray (92.2% vs. 54.4%, p < 0.001). On the contrary, the use of inhaled salbutamol remained substantially stable over time (39.4% vs. 37.6%, p = 0.505) [32]. Altogether, questions such as “do steroids protect from ICU treatment in bronchiolitis?” or “should we routinely give antibiotics to kids with bronchiolitis?” will probably receive uniformly negative answers from most of the pediatricians. Yet, when detailed analysis is performed based on data collection from pretty large patient numbers, we face the evidence that a uniform clinical practice is not yet in place. This is exactly what we found in our multicenter data collection from four Italian wards, during the fall-winter season 2021–2022.
This study has limitations. Since RSV was expected to be the main pathogen involved, extensive investigation of other viruses was not performed in all cases. Thus, our data cannot reflect local epidemiology of respiratory viruses in children with bronchiolitis. Furthermore, this was a retrospective observational study and not a prospective, therapeutic trial. Since the patients were not treated according to a common therapeutic protocol, differences in administration of antibiotics or steroids might, possibly, reflect also individual choice of the attending physician, not always in agreement with accepted international guidelines.
In conclusion, in our study of 214 children with bronchiolitis diagnosed and treated in four pediatric Italian centers, we observed a favorable prognosis in all cases, with requirement of ICU limited to a minority of patients. These findings are in keeping with those reported in a contemporary, comparable, multicenter setting in Italy [36, 37]. The vast majority of children was treated in the pediatric wards, equipped with low-flow and high-flow nasal cannula for oxygen therapy. The mean hospital stay of these children is usually expected to last less than 1 week. Our data suggest that, irrespective of treatments provided, RSV-positive and RSV-negative children had a similar clinical course. The results of our retrospective study further underline the need to improve adherence to existing guidelines on bronchiolitis treatment. Local healthcare professionals are asked to work together to effectively implement in their daily work bronchiolitis guidelines aiming to stop unnecessary tests and treatments.
Data Availability
Anonimyzed data analyzed for this study are available on request.
References
Meissner HC (2016) Viral bronchiolitis in children. N Engl J Med 374:62–72
Shaw KN, Bell LM, Sherman NH (1991) Outpatient assessment of infants with bronchiolitis. Am J Dis Child 145:151
Roback MG, Baskin MN (1997) Failure of oxygen saturation and clinical assessment to predict which patients with bronchiolitis discharged from the emergency department will return requiring admission. Pediatr Emerg Care 13:9
Hartling L, Bialy LM, Vandermeer B et al (2011) Epinephrine for bronchiolitis. Cochrane Database Syst Rev CD003123
Skjerven HO, Hunderi JO, Brügmann-Pieper SK et al (2013) Racemic adrenaline and inhalation strategies in acute bronchiolitis. N Engl J Med 368:2286
Gadomski AM, Scribani MB (2014) Bronchodilators for bronchiolitis. Cochrane Database Syst Rev CD001266
Patel H, Gouin S, Platt RW (2003) Randomized, double-blind, placebo-controlled trial of oral albuterol in infants with mild-to-moderate acute viral bronchiolitis. J Pediatr 142:509
Gadomski AM, Lichenstein R, Horton L et al (1994) Efficacy of albuterol in the management of bronchiolitis. Pediatrics 93:907
Cengizlier R, Saraçlar Y, Adalioğlu G, Tuncer A (1997) Effect of oral and inhaled salbutamol in infants with bronchiolitis. Acta Paediatr Jpn 39:61
Quinonez RA, Garber MD, Schroeder AR et al (2013) Choosing wisely in pediatric hospital medicine: five opportunities for improved healthcare value. J Hosp Med 8:479
King VJ, Viswanathan M, Bordley WC et al (2004) Pharmacologic treatment of bronchiolitis in infants and children: a systematic review. Arch Pediatr Adolesc Med 158:127
Cade A, Brownlee KG, Conway SP et al (2000) Randomised placebo controlled trial of nebulised corticosteroids in acute respiratory syncytial viral bronchiolitis. Arch Dis Child 82:126
Blom D, Ermers M, Bont L et al (2007) Inhaled corticosteroids during acute bronchiolitis in the prevention of post-bronchiolitic wheezing. Cochrane Database Syst Rev CD004881
Fernandes RM, Bialy LM, Vandermeer B et al (2013) Glucocorticoids for acute viral bronchiolitis in infants and young children. Cochrane Database Syst Rev CD004878
Elliott SA, Gaudet LA, Fernandes RM et al (2021) Comparative efficacy of bronchiolitis interventions in acute care: a network meta-analysis. Pediatrics 147
Mansbach JM, Hasegawa K, Geller RJ, Espinola JA, Sullivan AF, Camargo CA Jr (2020) MARC-35 Investigators. Bronchiolitis severity is related to recurrent wheezing by age 3 years in a prospective, multicenter cohort. Pediatr Res 87:428–430. https://doi.org/10.1038/s41390-019-0589-z
Fujiogi M, Goto T, Yasunaga H, Fujishiro J, Mansbach JM, Camargo CA Jr, Hasegawa K (2019) Trends in bronchiolitis hospitalizations in the United States: 2000–2016. Pediatrics 144:e20192614. https://doi.org/10.1542/peds.2019-2614
Shay DK, Holman RC, Roosevelt GE et al (2001) Bronchiolitis-associated mortality and estimates of respiratory syncytial virus-associated deaths among US children, 1979–1997. J Infect Dis 183:16
Holman RC, Shay DK, Curns AT et al (2003) Risk factors for bronchiolitis-associated deaths among infants in the United States. Pediatr Infect Dis J 22:483
Vaux S, Viriot D, Forgeot C, Pontais I, Savitch Y, Barondeau-Leuret A, Smadja S, Valette M, Enouf V, Parent du Chatelet I (2022) Bronchiolitis epidemics in France during the SARS-CoV-2 pandemic: the 2020–2021 and 2021–2022 seasons. Infect Dis Now S2666- 9919(22)00119–1. https://doi.org/10.1016/j.idnow.2022.06.003
Saravanos GL, Hu N, Homaira N, Muscatello DJ, Jaffe A, Bartlett AW, Wood NJ, Rawlinson W, Kesson A, Lingam R, Britton PN (2022) RSV epidemiology in Australia before and during COVID-19. Pediatrics 149:e2021053537. https://doi.org/10.1542/peds.2021-053537
Montejo M, Sánchez A, Paniagua N, Saiz-Hernando C, Benito J (2022) Reduction in the incidence of acute bronchiolitis and related hospital admissions during the COVID-19 pandemic. An Pediatr (Engl Ed) 96:537–539. https://doi.org/10.1016/j.anpede.2022.03.003
Camporesi A, Morello R, Ferro V, Pierantoni L, Rocca A, Lanari M, Trobia GL, Sciacca T, Bellinvia AG, De Ferrari A, Valentini P, Roland D, Buonsenso D (2022) Epidemiology, microbiology and severity of bronchiolitis in the first post-lockdown cold season in three different geographical areas in Italy: a prospective, observational study. Children (Basel) 9(4):491. https://doi.org/10.3390/children9040491
Korppi M (2022) Local healthcare professionals need to work together to implement bronchiolitis guidelines that stop unnecessary tests and treatments. Acta Paediatr 111(7):1319–1323. https://doi.org/10.1111/apa.16322
Pernica JM, Inch K, Alfaraidi H, Van Meer A, Carciumaru R, Luinstra K, Smieja M (2021) Assessment of nasopharyngeal Streptococcus pneumoniae colonization does not permit discrimination between Canadian children with viral and bacterial respiratory infection: a matched-cohort cross-sectional study. BMC Infect Dis 21(1):509. https://doi.org/10.1186/s12879-021-06235-z
Tang G, Lin J, Zhang Y, Shi Q (2021) The effects and safety of continuous positive airway pressure in children with bronchiolitis: a systematic review and meta-analysis. J Trop Pediatr 67(2):fmaa128. https://doi.org/10.1093/tropej/fmaa128
Solana-Gracia R, Modesto I Alapont V, Bueso-Inchausti L, Luna-Arana M, Möller-Díez A, Medina A, Pérez-Moneo B (2022) Changes in ventilation practices for bronchiolitis in the hospital ward and need for ICU transfer over the last decade. J Clin Med 11(6):1622. https://doi.org/10.3390/jcm11061622
Novak C, Vomiero G, de Caen A, Cooke S (2021) Current practices and policies regarding the use of high-flow nasal cannula on general pediatric inpatient wards in Canada. Paediatr Child Health 26(7):414–420. https://doi.org/10.1093/pch/pxab013
Gelbart B, McSharry B, Delzoppo C, Erickson S, Lee K, Butt W, Rea M, Wang X, Beca J, Kazemi A, Shann F (2022) Pragmatic randomized trial of corticosteroids and inhaled epinephrine for bronchiolitis in children in intensive care. J Pediatr 244:1723.e1. https://doi.org/10.1016/j.jpeds.2022.01.031
Shein SL, Yehya N (2022) Trials and tribulations in bronchiolitis. J Pediatr 244:8–10. https://doi.org/10.1016/j.jpeds.2022.02.053
Rodríguez-Martínez CE, Castro-Rodriguez JA, Nino G, Midulla F (2020) The impact of viral bronchiolitis phenotyping: Is it time to consider phenotype-specific responses to individualize pharmacological management? Paediatr Respir Rev 34:53–58. https://doi.org/10.1016/j.prrv.2019.04.003
Polack FP, Stein RT, Custovic A (2019) The syndrome we agreed to call bronchiolitis. J Infect Dis 220:184–186. https://doi.org/10.1093/infdis/jiz082
Petrarca L, Nenna R, Di Mattia G, Frassanito A, Castro-Rodriguez JA, Rodriguez Martinez CE et al (2022) Bronchiolitis phenotypes identified by latent class analysis may influence the occurrence of respiratory sequelae. Pediatr Pulmonol 57:616–622. https://doi.org/10.1002/ppul.25799
Bottau P, Liotti L, Laderchi E, Palpacelli A, Calamelli E, Colombo C, Serra L, Cazzato S (2022) Something is changing in viral infant bronchiolitis approach. Front Pediatr 10:865977. https://doi.org/10.3389/fped.2022.865977
Dohna-Schwake C, Mücher K, Stehling F, Rothoeft T, Roll C, Brevis Nuńez F, Seiffert P, Heister P, Hofmann M, Stein A, Jacobs A, Zuzak T, Durand P, Felderhoff-Müser U, Tissières P (2018) Differences of medical care for acute severe viral bronchiolitis in two urban areas in Europe. Klin Padiatr 230:245–250. https://doi.org/10.1055/a-0611-6109
Biagi C, Scarpini S, Paleari C, Fabi M, Dondi A, Gabrielli L, Gennari M, Lanari M, Pierantoni L (2021) Impact of guidelines publication on acute bronchiolitis management: 10-year experience from a tertiary care center in Italy. Microorganisms 9:2221. https://doi.org/10.3390/microorganisms9112221
Nenna R, Matera L, Licari A, Manti S, Di Bella G, Pierangeli A, Palamara AT, Nosetti L, Leonardi S, Marseglia GL, Midulla F, ICHRIS Group (2022) An Italian multicenter study on the epidemiology of respiratory syncytial virus during SARS-CoV-2 pandemic in hospitalized children. Front Pediatr 10:930281. https://doi.org/10.3389/fped.2022.930281
Author information
Authors and Affiliations
Contributions
Conceptualization, M.A., D.C., E.V. and M.F.; methodology, G.G.; formal analysis, G.G..; data curation, G.C., C.C., M.O.A., G.C., M.L., C.M.; writing — original draft, preparation, M.A., D.C., E.V.; writing — review and editing, D.T., M.O.A., G.C., M.F.
Corresponding author
Ethics declarations
Ethical approval
The study was conducted according to the guidelines of the Declaration of Helsinki, and approved by the Institutional Review Board (Comitato Etico delle Province di Chieti e Pescara—Verbale di seduta Del Comitato Etico N. 07 del giorno 21.04.2022).
Competing interests
The authors declare no competing interests.
Additional information
Communicated by Tobias Tenenbaum.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Carlone, G., Graziano, G., Trotta, D. et al. Bronchiolitis 2021–2022 epidemic: multicentric analysis of the characteristics and treatment approach in 214 children from different areas in Italy. Eur J Pediatr 182, 1921–1927 (2023). https://doi.org/10.1007/s00431-023-04853-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-023-04853-0