Abstract
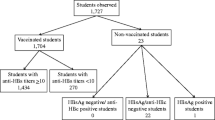
Neonatal vaccination against hepatitis B virus (HBV) infection was launched in 1998 in Turkey. The aim was to evaluate the persistence of seroprotection after HBV vaccination in order to determine the necessity of a single booster dose in 2- to 12-year-old children. This study was conducted retrospectively using hospital records of the children aged 2–12 years old who attended the pediatric outpatient clinics of Diskapi Training and Research Hospital, Ankara, Turkey between January 2010 and June 2011. Children who had received three doses of HBV vaccination in their infancy were included. A total of 530 children enrolled into the study, and 352 (66.4 %) of them had protective antibody to hepatitis surface antigens (anti-HBs) titer greater than 10 mIU/ml. The proportions of children with low, intermediate, and high anti-HB titers are different for those under 3 years of age. The majority were in the intermediate category. Those aged 4–10 years and 11 or older represented two-thirds of the children with high titers (p = 0.000). None of the children had chronic HBV infection. Unprotected children responded well after receiving the booster dose. The mean anti-HB concentration after the booster dose was more than 200 times higher than the mean antibody concentration before (p < 0.001). Conclusion: Our data suggest that HBV vaccination may confer long-term immunity. Use of routine booster doses of vaccine at these ages does not appear necessary to maintain long-term protection in successfully vaccinated immunocompetent children in the region.
Similar content being viewed by others
References
Alter MJ (2003) Epidemiology of hepatitis B in Europe and worldwide. J Hepatol 39:64–69
Bialek SR, Bower WA, Novak R, Helgenberger L, Auerbach SB, Williams IT, Bell BP (2008) Persistence of protection against hepatitis B virus infection among adolescents vaccinated with recombinant hepatitis B vaccine beginning at birth: a 15-year follow-up study. Pediatr Infect Dis J 27(10):881–885
Centers for Disease Control and Prevention (1991) Hepatitis B virus: a comprehensive strategy for eliminating transmission in the United States through universal childhood vaccination: recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 40(13):1–25
Chaves SS, Fischer G, Groeger J, Patel PR, Thompson ND, Teshale EH, Stevenson K, Yano VM, Armstrong GL, Samandari T, Kamili S, Drobeniuc J, Hu DJ (2012) Persistence of long-term immunity to hepatitis B among adolescents immunized at birth. Vaccine 21(30(9)):1644–1649
Chen DS (2009) Hepatitis B vaccination: the key towards elimination and eradication of hepatitis B. J Hepatol 50(4):805–816
Chinchai T, Chirathaworn C, Praianantathavorn K, Theamboonlers A, Hutagalung Y, Bock PH, Thantiworasit P, Poovorawan Y (2009) Long-term humoral and cellular immune response to hepatitis B vaccine in high-risk children 18–20 years after neonatal immunization. Viral Immunol 22(2):125–130
Dentinger CM, McMahon BJ, Butler JC, Dunaway CE, Zanis CL, Bulkow LR, Bruden DL, Nainan OV, Khristova ML, Hennessy TW, Parkinson AJ (2005) Persistence of antibody to hepatitis B and protection from disease among Alaska natives immunized at birth. Pediatr Infect Dis J 24:786–792
European Consensus Group on Hepatitis B Immunity (2000) Are booster immunisations needed for lifelong hepatitis B immunity? Lancet 355:561–565
Goldstein ST, Zhou F, Hadler SC, Bell BP, Mast EE, Margolis HS (2005) A mathematical model to estimate global hepatitis B disease burden and vaccination impact. Int J Epidemiol 34(6):1329–1339
Hammitt LL, Hennessy TW, Fiore AE, Zanis C, Hummel KB, Dunaway E, Bulkow L, McMahon BJ (2007) Hepatitis B immunity in children vaccinated with recombinant hepatitis B vaccine beginning at birth: a follow-up study at 15 years. Vaccine 25(39–40):6958–6964
Honorati MC, Palareti A, Dolzani P, Busachi CA, Rizzoli R, Facchini A (1999) A mathematical model predicting anti-hepatitis B virus surface antigen (HBs) decay after vaccination against hepatitis B. Clin Exp Immunol 116(1):121–126
James Koziel M, Siddiqui A (2005) Hepatitis B virus and hepatitis delta virus. In: Mandell GL, John E, Bennet RD (eds) Infectious diseases, principles and practice, 6th edn. Churchill Livingstone, Philadelphia, pp 1864–1884
Jan CF, Huang KC, Chien YC, Greydanus DE, Davies HD, Chiu TY, Huang LM, Chen CJ, Chen DS (2010) Determination of immune memory to hepatitis B vaccination through early booster response in college students. Hepatology 51(5):1547–1554
John TJ, Cooksley G (2005) Hepatitis B vaccine boosters: is there a clinical need in high endemicity populations? J Gastroenterol Hepatol 20(1):5–10
Kao JH, Chen DS (2005) Hepatitis B vaccination: to boost or not to boost? Lancet 366(9494):1337–1338
Kaya A, Erbey MF, Okur M, Sal E, Üstyol L, Bektaş SM (2011) Hepatitis B virus seropositivity and vaccination for children aged 0–18 in the Van Region. J Pediatr Inf 5:132–135
Lu CY, Chiang BL, Chi WK, Chang MH, Ni YH, Hsu HM, Twu SJ, Su IJ, Huang LM, Lee CY (2004) Waning immunity to plasma derived hepatitis B vaccine and the need for boosters 15 years after neonatal vaccination. Hepatology 40:1415–1420
Lu CY, Ni YH, Chiang BL, Chen PJ, Chang MH, Chang LY, Su IJ, Kuo HS, Huang LM, Chen DS, Lee CY (2008) Humoral and cellular immune responses to a hepatitis B vaccine booster 15–18 years after neonatal immunization. J Infect Dis 197(10):1419–1426
McMahon BJ, Bruden DL, Petersen KM, Bulkow LR, Parkinson AJ, Nainan O, Khristova M, Zanis C, Peters H, Margolis HS (2005) Antibody levels and protection after hepatitis B vaccine: results of a 15-year follow up. Ann Intern Med 142:333–341
McMahon BJ, Bulkow LR, Singleton RJ, Williams J, Snowball M, Homan C, Parkinson AJ (2011) Elimination of hepatocellular carcinoma and acute hepatitis B in children 25 years after a hepatitis B newborn and catch-up immunization program. Hepatology 54(3):801–807
Nalbantoğlu B, Nalbantoğlu A, Külcü NU, Say A (2010) Seroprevalence of hepatitis B and immunization status of children aged between 9 months–8 years old. J Child 10(3):116–121
Petersen KM, Bulkow LR, McMahon BJ, Zanis C, Getty M, Peters H, Parkinson AJ (2004) Duration of hepatitis B immunity in low risk children receiving hepatitis B vaccinations from birth. Pediatr Infect Dis J 23:650–655
Samandari T, Fiore AE, Negus S, Williams JL, Kuhnert W, McMahon BJ, Bell BP (2007) Differences in response to a hepatitis B vaccine booster dose among Alaskan children and adolescents vaccinated during infancy. Pediatrics 120(2):373–381
Seto D, West DJ, Ioli VA (2002) Persistence of antibody and immunologic memory in children immunized with hepatitis B vaccine at birth. Pediatr Infect Dis J 21(8):793–795
Su FH, Cheng SH, Li CY, Chen JD, Hsiao CY, Chien CC, Yang YC, Hung HH, Chu FY (2007) Hepatitis B seroprevalence and anamnestic response amongst Taiwanese young adults with full vaccination in infancy, 20 years subsequent to national hepatitis B vaccination. Vaccine 25(47):8085–8090
Süleyman A, Gökçay G, Badur S, Aykın S, Kılıç G, Tamay Z, Unüvar E, Güler N (2012) Evaluation of serological status of children following hepatitis B vaccination during infancy. Mikrobiyol Bul 46(1):47–56
Tan KL, Goh KT, Oon CJ, Chan SH (1994) Immunogenicity of recombinant yeast-derived hepatitis B vaccine in nonresponders to perinatal immunization. JAMA 271:859–861
Taşyaran MA (2003) HBV infeksiyonu epidemiyolojisi. In: Tekeli E, Balık İ (eds) Viral Hepatit. Karakter Col AŞ, Ankara, pp 121–128
Van Damme P, Kane M, Meheus A (1997) Integration of hepatitis B vaccination into national immunisation programmes. Br Med J 314:1033–1036
Van Damme P, Van Herck K (2007) A review of the long-term protection after hepatitis A and B vaccination. Travel Med Infect Dis 5(2):79–84
van der Sande MA, Waight P, Mendy M, Rayco-Solon P, Hutt P, Fulford T, Doherty C, McConkey SJ, Jeffries D, Hall AJ, Whittle HC (2006) Long-term protection against carriage of hepatitis B virus after infant vaccination. J Infect Dis 193(11):1528–1535
World Health Organization (1992) Expanded programme on immunization global advisory group. Wkly Epidemiol Rec 3:11–16
World Health Organization (2004) Hepatitis B vaccines WHO position paper. Wkly Epidemiol Rec 79:255–263
World Health Organization (2009) Hepatitis B vaccines WHO position paper. Wkly Epidemiol Rec 84:405–420
Yazdanpanah B, Safari M, Yazdanpanah S (2010) Persistence of HBV vaccine’s protection and response to hepatitis B booster immunization in 5- to 7-year-old children in the Kohgiloyeh and Boyerahmad Province, Iran. Hepat Mon 10(1):17–21
Zanetti AR, Mariano A, Romanò L, D’Amelio R, Chironna M, Coppola RC, Cuccia M, Mangione R, Marrone F, Negrone FS, Parlato A, Zamparo E, Zotti C, Stroffolini T, Mele A (2005) Long-term immunogenicity of hepatitis B vaccination and policy for booster: an Italian multicentre study. Lancet 366(9494):1379–1384
Zhu CL, Liu P, Chen T, Ni Z, Lu LL, Huang F, Lu J, Sun Z, Qu C (2011) Presence of immune memory and immunity to hepatitis B virus in adults after neonatal hepatitis B vaccination. Vaccine 13:7835–7841
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aypak, C., Yüce, A., Yıkılkan, H. et al. Persistence of protection of hepatitis B vaccine and response to booster immunization in 2- to 12-year-old children. Eur J Pediatr 171, 1761–1766 (2012). https://doi.org/10.1007/s00431-012-1815-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-012-1815-4