Abstract
Objectives
To measure anti-HAV antibodies 15 years after a single dose of live attenuated hepatitis A vaccine in Indian children.
Methods
Of the 143 children vaccinated in 2004, 109 were evaluated in 2019, clinically and for anti-HAV antibodies. These children have been assessed clinically every year, and for anti-HAV antibodies in 2004, 2007, 2010 and 2014.
Results
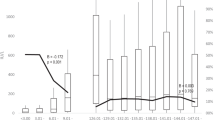
Of the 109 children who came for the present assessment, 11 had received additional doses of hepatitis A vaccine in 2004/2007 because of low anti-HAV titre (<20 mIU/mL). In the remaining 98 children, 94 (96%) had seroprotective levels with a geometric mean titre of 79.6 mIU/mL. Seroprotection rate in all 109 children was 86.2%.
Conclusions
Single dose of live attenuated hepatitis A vaccine in Indian children demonstrated robust immunogenicity at 15 years post vaccination.
Article PDF
Similar content being viewed by others
References
Mao JS, Dong DX, Zhang HY, et al. Primary study of attenuated live hepatitis A vaccine (H2 strain) in humans. J Infect Dis. 1989;159:621–24.
Zhuang FC, Qian W, et al. Persistent efficacy of live attenuated hepatitis A vaccine (H2-strain) after a mass vaccination program. Chin Med J (Engl). 2005;118:1851–856.
Zhuang FC, Mao ZA, Jiang LM, et al. Long term immunogenicity and effectiveness of live attenuated hepatitis A vaccine (H2-strain) — A study on the result of 15 years’ follow up. Zhonghua Liu Xing Bing Xue Za Zhi. 2010;31:1332–335.
Shah N, Faridi MMA, Mitra M, et al. Review of long term immunogenicity and tolerability of live hepatitis A vaccine. Hum Vaccin Immunother. 2020 Apr 3;1–6
WHO position paper on hepatitis A vaccines — June, 2012. Weekly Epidemiological Record. 2012;87:261–76.
Pemde H. Hepatitis A vaccines. In: Advisory Committee on Vaccines and Immunization Practices, Indian Academy of Pediatrics. IAP Guidebook on Immunization 2018–2019. 3rd edition. Jaypee Brothers Medical Publishers; 2020. p. 265–78.
Bhave S, Bavdekar A, Madan Z, et al. Evaluation of immunogenicity and tolerability of a live attenuated hepatitis A vaccine in Indian children. Indian Pediatr. 2006;43:983–7.
Bhave S, Sapru A, Bavdekar A, Bawangade S, Pandit A. Immunogenicity of single dose live attenuated hepatitis A vaccine. Indian Pediatr. 2011;48:135–37.
Bhave S, Sapru A, Bavdekar A, Kapatkar V, Mane A. Long-term immunogenicity of single dose of live attenuated hepatitis A vaccine in Indian children. Indian Pediatr. 2015;52:687–90.
Faridi MMA, Shah N, Ghosh TK, et al. Immunogenicity and safety of live attenuated hepatitis A vaccine: A multicentric study. Indian Pediatr. 2009;46:29–34.
Mitra M, Shah N, Faridi MMA, et al. Long term follow-up study to evaluate immunogenicity and safety of a single dose of live attenuated hepatitis A vaccine in children. Hum Vaccin Immunother. 2015;11:1147–152.
Wang XY, Xu ZY, Ma JC, et al. Long term immunogenicity after single and booster dose of a live attenuated hepatitis A vaccine: Results from 8-year follow-up. Vaccine. 2007;25:446–49.
Zhang QY, Chen H, Lin Z, Lin JM. Comparison of chemiluminescence enzyme immunoassay based on magnetic microparticles with traditional colorimetric ELISA for the detection of serum α-fetoprotein. J Pharm Analysis. 2012;2:130–35.
Arankalle V, Mitra M, Bhave S, et al. Changing epidemiology of hepatitis A virus in Indian children. Vaccine: Development and Therapy. 2014;4:7–13.
Chen Y, Zhou C-L, Zhang X-J, et al. Immune memory at 17-years of follow up of a single dose of live attenuated hepatitis A vaccine. Vaccine. 2018;36:114–21.
Acknowledgements
Dr Sonali Shah for data management and follow-up; Dr Deepak Langade (Clinsearch Healthcare Solutions Pvt Ltd) for statistical inputs; Dr Archana Karadkhele (Wockhardt Ltd) & Dr Pramit Sonone (Wockhardt Ltd) for valuable inputs.
Author information
Authors and Affiliations
Corresponding author
Additional information
Ethics clearance
KEM Hospital Research Centre Ethics Committee; No. KEMHRC/LFG/EC/778 dated June 6, 2016.
Contributors
SB, AS, AB: designed the study, recruited patients, analyzed results and wrote the manuscript; RJ, KD, VK: provided technical help needed for the study.
Funding
Wockhardt Ltd. Recipient of funds is KEM Hospital Research Centre, Pune.
Competing interests
SB, AS and AB: received investigator fee for conduct of the study; RJ, KD and VK are paid employees of Wockhardt Ltd.
Rights and permissions
About this article
Cite this article
Bhave, S., Sapru, A., Bavdekar, A. et al. Long term Immunogenicity of Single Dose of Live Attenuated Hepatitis A Vaccine in Indian Children — Results of 15-Year Follow-up. Indian Pediatr 58, 749–752 (2021). https://doi.org/10.1007/s13312-021-2285-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13312-021-2285-4