Abstract
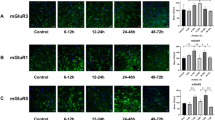
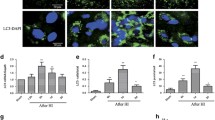
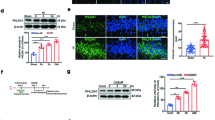
Following neonatal hypoxic–ischemia (HI) injury, it is crucial factor to reconstruct neural circuit and maintain neural network homeostasis for neurological recovery. A dynamic balance between the synthesis and degradation of synaptic protein is required for maintaining synaptic plasticity. Protein degradation is facilitated by autophagy. This study aimed to investigate the regulation of synaptic structural plasticity by the Notch pathway, by assessing changes in Notch pathway activation and their effects on synaptic proteins and autophagy after HI injury. The study involved 48 male newborn Yorkshire piglets, each weighing 1.0–1.5 kg and 3 days old. They were randomly assigned to two groups: the HI group and the Notch pathway inhibitor + HI group (n = 24 per group). Each group was further divided into six subgroups according to HI duration (n = 4 per group): a control subgroup, and 0–6, 6–12, 12–24, 24–48, and 48–72 h subgroups. The expression of Notch pathway-related proteins, including Notch1, Hes1, and Notch intracellular domains, increased following HI injury. The expression of autophagy proteins increased at 0–6 h and 6–12 h post-HI. The expression of synaptic proteins, such as postsynaptic density protein 95 (PSD95) and synaptophysin, increased 6–12 h and 12–24 h after HI, respectively. Notably, the increased expression of these proteins was reversed by a Notch pathway inhibitor. Transmission electron microscopy revealed the presence of autophagosome structures in synapses. These findings shed light on the underlying mechanisms of neurological recovery after HI injury and may provide insights into potential therapeutic targets for promoting neural circuit reconstruction and maintaining neural network homeostasis.








Similar content being viewed by others
Data availability
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Abbreviations
- Atg:
-
Autophagy-related gene
- DAPT:
-
N-[N-(3,5-difluorophenacetyl)-L-alanyl]-S-phenylglycine t-butyl ester
- DMSO:
-
Dimethyl sulfoxide
- Hes:
-
Hairy enhancer of split
- HI:
-
Hypoxic ischemia
- HIF-1:
-
Hypoxia-inducible factor-1
- LAMP-1:
-
Lysosome-associated membrane protein-1
- LC3-II:
-
Microtubule-associated protein 1 light chain 3-II
- NICD:
-
Notch intracellular domain
- PSD95:
-
Postsynaptic density protein 95
- SVs:
-
Synaptic vesicles
- SYP:
-
Synaptophysin
- TEM:
-
Transmission electron microscopy
References
Al HZA, Madeddu P (2009) Notch signaling in ischaemia-induced angiogenesis. Biochem Soc Trans 37:1221–1227. https://doi.org/10.1042/BST0371221
Alberi L, Hoey SE, Brai E, Scotti AL, Marathe S (2013) Notch signaling in the brain: in good and bad times. Ageing Res Rev 12:801–814. https://doi.org/10.1016/j.arr.2013.03.004
Androutsellis-Theotokis A, Leker RR, Soldner F, Hoeppner DJ, Ravin R, Poser SW, Rueger MA, Bae SK, Kittappa R, McKay RD (2006) Notch signalling regulates stem cell numbers in vitro and in vivo. Nature 442:823–826. https://doi.org/10.1038/nature04940
Arumugam TV, Baik SH, Balaganapathy P, Sobey CG, Mattson MP, Jo DG (2018) Notch signaling and neuronal death in stroke. Prog Neurobiol 165–167:103–116. https://doi.org/10.1016/j.pneurobio.2018.03.002
Ayuso-Dolado S, Esteban-Ortega GM, Vidaurre OG, Diaz-Guerra M (2021) A novel cell-penetrating peptide targeting calpain-cleavage of PSD-95 induced by excitotoxicity improves neurological outcome after stroke. Theranostics 11:6746–6765. https://doi.org/10.7150/thno.60701
Bento CF, Renna M, Ghislat G, Puri C, Ashkenazi A, Vicinanza M, Menzies FM, Rubinsztein DC (2016) Mammalian autophagy: how does it work? Annu Rev Biochem 85:685–713. https://doi.org/10.1146/annurev-biochem-060815-014556
Bingol B, Sheng M (2011) Deconstruction for reconstruction: the role of proteolysis in neural plasticity and disease. Neuron 69:22–32. https://doi.org/10.1016/j.neuron.2010.11.006
Bonnycastle K, Davenport EC, Cousin MA (2021) Presynaptic dysfunction in neurodevelopmental disorders: Insights from the synaptic vesicle life cycle. J Neurochem 157:179–207. https://doi.org/10.1111/jnc.15035
Buono KD, Goodus MT, Guardia CM, Jiang Y, Loporchio D, Levison SW (2015) Mechanisms of mouse neural precursor expansion after neonatal hypoxia-ischemia. J Neurosci 35:8855–8865. https://doi.org/10.1523/JNEUROSCI.2868-12.2015
Calabresi P, Centonze D, Pisani A, Cupini L, Bernardi G (2003) Synaptic plasticity in the ischaemic brain. Lancet Neurol 2:622–629. https://doi.org/10.1016/s1474-4422(03)00532-5
Cheng YL, Choi Y, Seow WL, Manzanero S, Sobey CG, Jo DG, Arumugam TV (2014a) Evidence that neuronal Notch-1 promotes JNK/c-Jun activation and cell death following ischemic stress. Brain Res 1586:193–202. https://doi.org/10.1016/j.brainres.2014.08.054
Cheng YL, Park JS, Manzanero S, Choi Y, Baik SH, Okun E, Gelderblom M, Fann DY, Magnus T, Launikonis BS, Mattson MP, Sobey CG, Jo DG, Arumugam TV (2014b) Evidence that collaboration between HIF-1alpha and Notch-1 promotes neuronal cell death in ischemic stroke. Neurobiol Dis 62:286–295. https://doi.org/10.1016/j.nbd.2013.10.009
Citri A, Malenka RC (2008) Synaptic plasticity: multiple forms, functions, and mechanisms. Neuropsychopharmacology 33:18–41. https://doi.org/10.1038/sj.npp.1301559
Cui D, Sun D, Wang X, Yi L, Kulikowicz E, Reyes M, Zhu J, Yang ZJ, Jiang W, Koehler RC (2017) Impaired autophagosome clearance contributes to neuronal death in a piglet model of neonatal hypoxic-ischemic encephalopathy. Cell Death Dis 8:e2919. https://doi.org/10.1038/cddis.2017.318
Dhuriya YK, Sharma D (2020) Neuronal plasticity: neuronal organization is associated with neurological disorders. J Mol Neurosci 70:1684–1701. https://doi.org/10.1007/s12031-020-01555-2
Fan YY, Hu WW, Nan F, Chen Z (2017) Postconditioning-induced neuroprotection, mechanisms and applications in cerebral ischemia. Neurochem Int 107:43–56. https://doi.org/10.1016/j.neuint.2017.01.006
Faust TE, Gunner G, Schafer DP (2021) Mechanisms governing activity-dependent synaptic pruning in the developing mammalian CNS. Nat Rev Neurosci 22:657–673. https://doi.org/10.1038/s41583-021-00507-y
Felling RJ, Covey MV, Wolujewicz P, Batish M, Levison SW (2016) Astrocyte-produced leukemia inhibitory factor expands the neural stem/progenitor pool following perinatal hypoxia-ischemia. J Neurosci Res 94:1531–1545. https://doi.org/10.1002/jnr.23929
Fleming A, Rubinsztein DC (2020) Autophagy in neuronal development and plasticity. Trends Neurosci 43:767–779. https://doi.org/10.1016/j.tins.2020.07.003
Ginet V, Pittet MP, Rummel C, Osterheld MC, Meuli R, Clarke PG, Puyal J, Truttmann AC (2014) Dying neurons in thalamus of asphyxiated term newborns and rats are autophagic. Ann Neurol 76:695–711. https://doi.org/10.1002/ana.24257
Guruharsha KG, Kankel MW, Artavanis-Tsakonas S (2012) The Notch signalling system: recent insights into the complexity of a conserved pathway. Nat Rev Genet 13:654–666. https://doi.org/10.1038/nrg3272
He C, Klionsky DJ (2009) Regulation mechanisms and signaling pathways of autophagy. Annu Rev Genet 43:67–93. https://doi.org/10.1146/annurev-genet-102808-114910
Hefter D, Draguhn A (2017) APP as a protective factor in acute neuronal insults. Front Mol Neurosci 10:22. https://doi.org/10.3389/fnmol.2017.00022
Hirata H, Yoshiura S, Ohtsuka T, Bessho Y, Harada T, Yoshikawa K, Kageyama R (2002) Oscillatory expression of the bHLH factor Hes1 regulated by a negative feedback loop. Science 298:840–843. https://doi.org/10.1126/science.1074560
Ho DM, Artavanis-Tsakonas S, Louvi A (2020) The Notch pathway in CNS homeostasis and neurodegeneration. Wiley Interdiscip Rev Dev Biol. https://doi.org/10.1002/wdev.358
Hogan MK, Hamilton GF, Horner PJ (2020) Neural Stimulation and molecular mechanisms of plasticity and regeneration: a review. Front Cell Neurosci 14:271. https://doi.org/10.3389/fncel.2020.00271
Johnston MV, Ishida A, Ishida WN, Matsushita HB, Nishimura A, Tsuji M (2009) Plasticity and injury in the developing brain. Brain Dev 31:1–10. https://doi.org/10.1016/j.braindev.2008.03.014
Jung WB, Im GH, Chung JJ, Ahn SY, Jeon TY, Chang YS, Park WS, Kim JH, Kim KS, Lee JH (2016) Neuroplasticity for spontaneous functional recovery after neonatal hypoxic ischemic brain injury in rats observed by functional MRI and diffusion tensor imaging. Neuroimage 126:140–150. https://doi.org/10.1016/j.neuroimage.2015.11.032
Kageyama R, Shimojo H, Isomura A (2018) Oscillatory control of notch signaling in development. Adv Exp Med Biol 1066:265–277. https://doi.org/10.1007/978-3-319-89512-3_13
Li Z, Wang J, Zhao C, Ren K, Xia Z, Yu H, Jiang K (2016) Acute blockage of notch Signaling by DAPT induces neuroprotection and neurogenesis in the neonatal rat brain after stroke. Transl Stroke Res 7:132–140. https://doi.org/10.1007/s12975-015-0441-7
Li K, Zheng Y, Wang X (2021a) Self-Regulation of cerebral metabolism and its neuroprotective effect after hypoxic-ischemic injury: evidence from 1H-MRS. Front Neuroanat 15:672412. https://doi.org/10.3389/fnana.2021.672412
Li K, Zheng Y, Wang X (2021b) The potential relationship between HIF-1α and amino acid metabolism after hypoxic ischemia and dual effects on neurons. Front Neurosci 15:676553. https://doi.org/10.3389/fnins.2021.676553
Lieberman OJ, Sulzer D (2020) The synaptic autophagy cycle. J Mol Biol 432:2589–2604. https://doi.org/10.1016/j.jmb.2019.12.028
Liu D, Bai X, Ma W, Xin D, Chu X, Yuan H, Qiu J, Ke H, Yin S, Chen W, Wang Z (2020) Purmorphamine attenuates neuro-inflammation and synaptic impairments after hypoxic-ischemic injury in neonatal mice via shh signaling. Front Pharmacol 11:204. https://doi.org/10.3389/fphar.2020.00204
Liu J, Gu Y, Guo M, Ji X (2021) Neuroprotective effects and mechanisms of ischemic/hypoxic preconditioning on neurological diseases. CNS Neurosci Ther 27:869–882. https://doi.org/10.1111/cns.13642
Moriyama M, Moriyama H, Uda J, Matsuyama A, Osawa M, Hayakawa T (2014) BNIP3 plays crucial roles in the differentiation and maintenance of epidermal keratinocytes. J Invest Dermatol 134:1627–1635. https://doi.org/10.1038/jid.2014.11
Natsumeda M, Maitani K, Liu Y, Miyahara H, Kaur H, Chu Q, Zhang H, Kahlert UD, Eberhart CG (2016) Targeting notch signaling and autophagy increases cytotoxicity in glioblastoma neurospheres. Brain Pathol 26:713–723. https://doi.org/10.1111/bpa.12343
Niture S, Gyamfi MA, Kedir H, Arthur E, Ressom H, Deep G, Kumar D (2018) Serotonin induced hepatic steatosis is associated with modulation of autophagy and notch signaling pathway. Cell Commun Signal 16:78. https://doi.org/10.1186/s12964-018-0282-6
Oya S, Yoshikawa G, Takai K, Tanaka JI, Higashiyama S, Saito N, Kirino T, Kawahara N (2009) Attenuation of Notch signaling promotes the differentiation of neural progenitors into neurons in the hippocampal CA1 region after ischemic injury. Neuroscience 158:683–692. https://doi.org/10.1016/j.neuroscience.2008.10.043
Perrone-Capano C, Volpicelli F, Penna E, Chun JT, Crispino M (2021) Presynaptic protein synthesis and brain plasticity: from physiology to neuropathology. Prog Neurobiol. https://doi.org/10.1016/j.pneurobio.2021.102051
Qi ZF, Luo YM, Liu XR, Wang RL, Zhao HP, Yan F, Song ZJ, Luo M, Ji XM (2012) AKT/GSK3beta-dependent autophagy contributes to the neuroprotection of limb remote ischemic postconditioning in the transient cerebral ischemic rat model. CNS Neurosci Ther 18:965–973. https://doi.org/10.1111/cns.12016
Rao P, Lou F, Luo D, Huang C, Huang K, Yao Z, Xiao J (2021) Decreased autophagy impairs osteogenic differentiation of adipose-derived stem cells via notch signaling in diabetic osteoporosis mice. Cell Signal. https://doi.org/10.1016/j.cellsig.2021.110138
Ren C, Yao Y, Han R, Huang Q, Li H, Wang B, Li S, Li M, Mao Y, Mao X, Xie L, Zhou L, Hu J, Ji X, Jin K (2018) Cerebral ischemia induces angiogenesis in the peri-infarct regions via Notch1 signaling activation. Exp Neurol 304:30–40. https://doi.org/10.1016/j.expneurol.2018.02.013
Rocha-Ferreira E, Hristova M (2016) Plasticity in the Neonatal brain following hypoxic-ischaemic injury. Neural Plast 2016:4901014. https://doi.org/10.1155/2016/4901014
Shao G, Wang Y, Guan S, Burlingame AL, Lu F, Knox R, Ferriero DM, Jiang X (2017) Proteomic analysis of mouse cortex postsynaptic density following neonatal brain hypoxia-ischemia. Dev Neurosci 39:66–81. https://doi.org/10.1159/000456030
Stavoe A, Holzbaur E (2019) Autophagy in neurons. Annu Rev Cell Dev Biol 35:477–500. https://doi.org/10.1146/annurev-cellbio-100818-125242
Tang G, Gudsnuk K, Kuo SH, Cotrina ML, Rosoklija G, Sosunov A, Sonders MS, Kanter E, Castagna C, Yamamoto A, Yue Z, Arancio O, Peterson BS, Champagne F, Dwork AJ, Goldman J, Sulzer D (2014) Loss of mTOR-dependent macroautophagy causes autistic-like synaptic pruning deficits. Neuron 83:1131–1143. https://doi.org/10.1016/j.neuron.2014.07.040
Wu X, Fleming A, Ricketts T, Pavel M, Virgin H, Menzies FM, Rubinsztein DC (2016) Autophagy regulates Notch degradation and modulates stem cell development and neurogenesis. Nat Commun 7:10533. https://doi.org/10.1038/ncomms10533
Xu Y, Tian Y, Tian Y, Li X, Zhao P (2016) Autophagy activation involved in hypoxic-ischemic brain injury induces cognitive and memory impairment in neonatal rats. J Neurochem 139:795–805. https://doi.org/10.1111/jnc.13851
Yang Z, Levison SW (2007) Perinatal hypoxic/ischemic brain injury induces persistent production of striatal neurons from subventricular zone progenitors. Dev Neurosci 29:331–340. https://doi.org/10.1159/000105474
Ye L, Feng Z, Doycheva D, Malaguit J, Dixon B, Xu N, Zhang JH, Tang J (2018) CpG-ODN exerts a neuroprotective effect via the TLR9/pAMPK signaling pathway by activation of autophagy in a neonatal HIE rat model. Exp Neurol 301:70–80. https://doi.org/10.1016/j.expneurol.2017.12.008
Yu L, Chen Y, Tooze SA (2018) Autophagy pathway: cellular and molecular mechanisms. Autophagy 14:207–215. https://doi.org/10.1080/15548627.2017.1378838
Yuan P, Han W, Xie L, Cheng L, Chen H, Chen J, Jiang L (2020) The implications of hippocampal neurogenesis in adolescent rats after status epilepticus: a novel role of notch signaling pathway in regulating epileptogenesis. Cell Tissue Res. https://doi.org/10.1007/s00441-019-03146-z
Zhang C, Li W, Wen J, Yang Z (2017) Autophagy is involved in mouse kidney development and podocyte differentiation regulated by Notch signalling. J Cell Mol Med 21:1315–1328. https://doi.org/10.1111/jcmm.13061
Zhang M, Wu P, Li M, Guo Y, Tian T, Liao X, Tan S (2021) Inhibition of Notch1 signaling reduces hepatocyte injury in nonalcoholic fatty liver disease via autophagy. Biochem Biophys Res Commun 547:131–138. https://doi.org/10.1016/j.bbrc.2021.02.039
Zhang R, Engler A, Taylor V (2018) Notch: an interactive player in neurogenesis and disease. Cell Tissue Res 371:73–89. https://doi.org/10.1007/s00441-017-2641-9
Zhang Y, Xiang Z, Jia Y, He X, Wang L, Cui W (2019) The Notch signaling pathway inhibitor Dapt alleviates autism-like behavior, autophagy and dendritic spine density abnormalities in a valproic acid-induced animal model of autism. Prog Neuropsychopharmacol Biol Psychiatry. https://doi.org/10.1016/j.pnpbp.2019.109644
Ziemka-Nalecz M, Janowska J, Strojek L, Jaworska J, Zalewska T, Frontczak-Baniewicz M, Sypecka J (2018) Impact of neonatal hypoxia-ischaemia on oligodendrocyte survival, maturation and myelinating potential. J Cell Mol Med 22:207–222. https://doi.org/10.1111/jcmm.13309
Acknowledgements
We acknowledge the financial support from the National Natural Science Foundation of China (No.81871408, 81271631), National Science Foundation for Young Scientists of China (No.81801658), Outstanding Scientific Fund of Shengjing Hospital (No. 201402), and the 345 Talent Support Project at Shengjing Hospital (No 30 B). We acknowledge Figdraw (www.figdraw.com, accessed data: 20 March 2023) for providing the schematic figure support.
Funding
This study was supported by the National Natural Science Foundation of China (No.81871408, 81271631), Young Scientists Fund (CN) (No.81801658), Outstanding Scientific Fund of Shengjing Hospital (No. 201402), and the 345 Talent Support Project of Shengjing Hospital (No 30 B).
Author information
Authors and Affiliations
Contributions
Conceptualization: XW. Data curation: KL, ML, and MC. Investigation: KL. Methodology: KL. Project administration: XW. Resources: YZ. Supervision: YZ. Validation: YZ. Writing—original draft: KL. Writing—review and editing: XW.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval and consent to participate
All experimental procedures performed on the animals were approved by the Institutional Committee for Animal Care and Use of our hospital (No. 2018PS43K).
Consent for publication
All authors approved the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.





Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, K., Lu, M., Cui, M. et al. The Notch pathway regulates autophagy after hypoxic–ischemic injury and affects synaptic plasticity. Brain Struct Funct 228, 985–996 (2023). https://doi.org/10.1007/s00429-023-02639-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-023-02639-6