Abstract
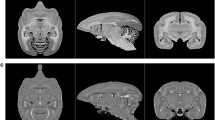
A modified and extended version, HCPex, is provided of the surface-based Human Connectome Project-MultiModal Parcellation atlas of human cortical areas (HCP-MMP v1.0, Glasser et al. 2016). The original atlas with 360 cortical areas has been modified in HCPex for ease of use with volumetric neuroimaging software, such as SPM, FSL, and MRIcroGL. HCPex is also an extended version of the original atlas in which 66 subcortical areas (33 in each hemisphere) have been added, including the amygdala, thalamus, putamen, caudate nucleus, nucleus accumbens, globus pallidus, mammillary bodies, septal nuclei and nucleus basalis. HCPex makes available the excellent parcellation of cortical areas in HCP-MMP v1.0 to users of volumetric software, such as SPM and FSL, as well as adding some subcortical regions, and providing labelled coronal views of the human brain.




Similar content being viewed by others
References
Avants BB, Tustison NJ, Song G (2009) Advanced normalization tools (ANTS). Insight j 2(365):1–35
Avants BB, Tustison NJ, Song G, Cook PA, Klein A, Gee JC (2011) A reproducible evaluation of ANTs similarity metric performance in brain image registration. Neuroimage 54(3):2033–2044. https://doi.org/10.1016/j.neuroimage.2010.09.025
Beauchamp MS (2021) Cortical Surface HCP. https://openwetware.org/wiki/Beauchamp:CorticalSurfaceHCP
Chakravarty MM, Steadman P, van Eede MC, Calcott RD, Gu V, Shaw P, Raznahan A, Collins DL, Lerch JP (2013) Performing label-fusion-based segmentation using multiple automatically generated templates. Hum Brain Mapp 34(10):2635–2654. https://doi.org/10.1002/hbm.22092
Coalson TS, Van Essen DC, Glasser MF (2018) The impact of traditional neuroimaging methods on the spatial localization of cortical areas. Proc Natl Acad Sci USA 115(27):E6356–E6365. https://doi.org/10.1073/pnas.1801582115
Dickie EW, Anticevic A, Smith DE, Coalson TS, Manogaran M, Calarco N, Viviano JD, Glasser MF, Van Essen DC, Voineskos AN (2019) Ciftify: a framework for surface-based analysis of legacy MR acquisitions. Neuroimage 197:818–826. https://doi.org/10.1016/j.neuroimage.2019.04.078
Eickhoff SB, Stephan KE, Mohlberg H, Grefkes C, Fink GR, Amunts K, Zilles K (2005) A new SPM toolbox for combining probabilistic cytoarchitectonic maps and functional imaging data. Neuroimage 25(4):1325–1335
Eickhoff SB, Heim S, Zilles K, Amunts K (2006) Testing anatomically specified hypotheses in functional imaging using cytoarchitectonic maps. Neuroimage 32(2):570–582. https://doi.org/10.1016/j.neuroimage.2006.04.204
Entis JJ, Doerga P, Barrett LF, Dickerson BC (2012) A reliable protocol for the manual segmentation of the human amygdala and its subregions using ultra-high resolution MRI. Neuroimage 60(2):1226–1235. https://doi.org/10.1016/j.neuroimage.2011.12.073
Fischl B, Sereno MI, Dale AM (1999a) Cortical surface-based analysis. II: inflation, flattening, and a surface-based coordinate system. Neuroimage 9(2):195–207. https://doi.org/10.1006/nimg.1998.0396
Fischl B, Sereno MI, Tootell RB, Dale AM (1999b) High-resolution intersubject averaging and a coordinate system for the cortical surface. Hum Brain Mapp 8(4):272–284. https://doi.org/10.1002/(sici)1097-0193(1999)8:4%3c272::aid-hbm10%3e3.0.co;2-4
Fischl B, Salat DH, Busa E, Albert M, Dieterich M, Haselgrove C, van der Kouwe A, Killiany R, Kennedy D, Klaveness S, Montillo A, Makris N, Rosen B, Dale AM (2002) Whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron 33(3):341–355. https://doi.org/10.1016/s0896-6273(02)00569-x
Fischl B, van der Kouwe A, Destrieux C, Halgren E, Segonne F, Salat DH, Busa E, Seidman LJ, Goldstein J, Kennedy D, Caviness V, Makris N, Rosen B, Dale AM (2004) Automatically parcellating the human cerebral cortex. Cereb Cortex 14(1):11–22. https://doi.org/10.1093/cercor/bhg087
Fonov VS, Evans AC, McKinstry RC, Almli CR, Collins DL (2009) Unbiased nonlinear average age-appropriate brain templates from birth to adulthood. Neuroimage 47:S102
Fonov V, Evans AC, Botteron K, Almli CR, McKinstry RC, Collins DL, Brain Development Cooperative G (2011) Unbiased average age-appropriate atlases for pediatric studies. Neuroimage 54(1):313–327. https://doi.org/10.1016/j.neuroimage.2010.07.033
Friston KJ, Ashburner JT, Kiebel SJ, Nichols TE, Penny WD (2006) Statistical parametric mapping: the analysis of functional brain images. Academic Press
Georges-François P, Rolls ET, Robertson RG (1999) Spatial view cells in the primate hippocampus: allocentric view not head direction or eye position or place. Cereb Cortex 9:197–212
Glasser MF, Coalson TS, Robinson EC, Hacker CD, Harwell J, Yacoub E, Ugurbil K, Andersson J, Beckmann CF, Jenkinson M, Smith SM, Van Essen DC (2016a) A multi-modal parcellation of human cerebral cortex. Nature 536(7615):171–178. https://doi.org/10.1038/nature18933
Glasser MF, Smith SM, Marcus DS, Andersson JL, Auerbach EJ, Behrens TE, Coalson TS, Harms MP, Jenkinson M, Moeller S, Robinson EC, Sotiropoulos SN, Xu J, Yacoub E, Ugurbil K, Van Essen DC (2016b) The Human Connectome Project’s neuroimaging approach. Nat Neurosci 19(9):1175–1187. https://doi.org/10.1038/nn.4361
Holmes CJ, Hoge R, Collins L, Woods R, Toga AW, Evans AC (1998) Enhancement of MR images using registration for signal averaging. J Comput Assist Tomogr 22(2):324–333. https://doi.org/10.1097/00004728-199803000-00032
Horn A (2016a) HCP-MMP1.0 projected on MNI2009a GM (volumetric) in NIfTI format.
Horn A (2016b) MMP 1.0 MNI projections. https://www.neurovaultorg/collections/1549/
Huang C-C, Rolls ET, Hsu C-CH, Feng J, Lin C-P (2021) Extensive cortical connectivity of the human hippocampal memory system: beyond the “what” and “where” dual-stream model. Cereb Cortex 31:4652–4669. https://doi.org/10.1093/cercor/bhab113
Iglesias JE, Insausti R, Lerma-Usabiaga G, Bocchetta M, Van Leemput K, Greve DN, van der Kouwe A, Alzheimer’s Disease Neuroimaging I, Fischl B, Caballero-Gaudes C, Paz-Alonso PM (2018) A probabilistic atlas of the human thalamic nuclei combining ex vivo MRI and histology. Neuroimage 183:314–326. https://doi.org/10.1016/j.neuroimage.2018.08.012
Jenkinson M, Beckmann CF, Behrens TE, Woolrich MW, Smith SM (2012) FSL Neuroimage 62(2):782–790. https://doi.org/10.1016/j.neuroimage.2011.09.015
Kesner RP, Rolls ET (2015) A computational theory of hippocampal function, and tests of the theory: new developments. Neurosci Biobehav Rev 48:92–147. https://doi.org/10.1016/j.neubiorev.2014.11.009
Krauth A, Blanc R, Poveda A, Jeanmonod D, Morel A, Szekely G (2010) A mean three-dimensional atlas of the human thalamus: generation from multiple histological data. Neuroimage 49(3):2053–2062. https://doi.org/10.1016/j.neuroimage.2009.10.042
Ma Q, Rolls ET, Huang C-C, Cheng W, Feng J (2021) Extensive cortical functional connectivity of the human hippocampal memory system.under review
Mills K (2016) HCP-MMP1.0 projected on fsaverage. figshare. Dataset. https://doi.org/10.6084/m9.figshare.3498446.v2
Pauli WM, Nili AN, Tyszka JM (2018) A high-resolution probabilistic in vivo atlas of human subcortical brain nuclei. Sci Data 5:180063. https://doi.org/10.1038/sdata.2018.63
Pipitone J, Park MT, Winterburn J, Lett TA, Lerch JP, Pruessner JC, Lepage M, Voineskos AN, Chakravarty MM, Alzheimer’s Disease Neuroimaging I (2014) Multi-atlas segmentation of the whole hippocampus and subfields using multiple automatically generated templates. Neuroimage 101:494–512. https://doi.org/10.1016/j.neuroimage.2014.04.054
Rolls ET (2018) The storage and recall of memories in the hippocampo-cortical system. Cell Tissue Res 373:577–604. https://doi.org/10.1007/s00441-017-2744-3
Rolls ET (2021) Neurons including hippocampal spatial view cells, and navigation in primates including humans. Hippocampus 31:593–611. https://doi.org/10.1002/hipo.23324
Rolls ET, Wirth S (2018) Spatial representations in the primate hippocampus, and their functions in memory and navigation. Prog Neurobiol 171:90–113. https://doi.org/10.1016/j.pneurobio.2018.09.004
Rolls ET, Robertson RG, Georges-François P (1997) Spatial view cells in the primate hippocampus. Eur J Neurosci 9:1789–1794
Rolls ET, Huang CC, Lin CP, Feng J, Joliot M (2020) Automated anatomical labelling atlas 3. Neuroimage 206:116189. https://doi.org/10.1016/j.neuroimage.2019.116189
Rolls ET, Deco G, Huang CC, Feng J (2021) The effective connectivity of the human hippocampal memory system. Cereb Cortex. https://doi.org/10.1093/cercor/bhab442
Smith SM, Jenkinson M, Woolrich MW, Beckmann CF, Behrens TE, Johansen-Berg H, Bannister PR, De Luca M, Drobnjak I, Flitney DE, Niazy RK, Saunders J, Vickers J, Zhang Y, De Stefano N, Brady JM, Matthews PM (2004) Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage 23(Suppl 1):S208-219. https://doi.org/10.1016/j.neuroimage.2004.07.051
Su JH, Thomas FT, Kasoff WS, Tourdias T, Choi EY, Rutt BK, Saranathan M (2019) Thalamus Optimized Multi Atlas Segmentation (THOMAS): fast, fully automated segmentation of thalamic nuclei from structural MRI. Neuroimage 194:272–282. https://doi.org/10.1016/j.neuroimage.2019.03.021
Tian Y, Margulies DS, Breakspear M, Zalesky A (2020) Topographic organization of the human subcortex unveiled with functional connectivity gradients. Nat Neurosci 23(11):1421–1432. https://doi.org/10.1038/s41593-020-00711-6
Treadway MT, Waskom ML, Dillon DG, Holmes AJ, Park MTM, Chakravarty MM, Dutra SJ, Polli FE, Iosifescu DV, Fava M, Gabrieli JDE, Pizzagalli DA (2015) Illness progression, recent stress, and morphometry of hippocampal subfields and medial prefrontal cortex in major depression. Biol Psychiatry 77(3):285–294. https://doi.org/10.1016/j.biopsych.2014.06.018
Van Essen DC, Smith J, Glasser MF, Elam J, Donahue CJ, Dierker DL, Reid EK, Coalson T, Harwell J (2017) The Brain Analysis Library of Spatial maps and Atlases (BALSA) database. Neuroimage 144(Pt B):270–274. https://doi.org/10.1016/j.neuroimage.2016.04.002
Zaborszky L, Hoemke L, Mohlberg H, Schleicher A, Amunts K, Zilles K (2008) Stereotaxic probabilistic maps of the magnocellular cell groups in human basal forebrain. Neuroimage 42(3):1127–1141. https://doi.org/10.1016/j.neuroimage.2008.05.055
Acknowledgements
The use of the HCP-MMP v1.0 atlas (Glasser et al. 2016a, b) in the construction of HCPex is acknowledged, and reference should be made to that paper if use is made of HCPex. The volumetric version of the Glasser et al. atlas (2016a) produced by Coalson et al. (2018) was downloaded with grateful acknowledgement from the publicly released version of the parcellation (https://balsa.wustl.edu/file/show/nvrZ). The neuroimaging data were provided by the Human Connectome Project, WU-Minn Consortium (Principal Investigators: David Van Essen and Kamil Ugurbil; 1U54MH091657) funded by the 16 NIH Institutes and Centers that support the NIH Blueprint for Neuroscience Research; and by the McDonnell Center for Systems Neuroscience at Washington University. Professor Laszlo Zaborszky is warmly thanked for providing a parcellation of the Ch1-Ch4 nuclei based on Zaborszky et al. (2008).
Funding
This research was supported by a grant to Professor C-P. Lin that included research with Professor E.T. Rolls (Ministry of Science and Technology (MOST) of Taiwan, MOST 110-2321-B-010-010-004 and MOST 110-2634-F-010-001). The research was also supported by the following grants to Professor J. Feng: National Key R&D Program of China (No. 2019YFA0709502); 111 Project (No. B18015); Shanghai Municipal Science and Technology Major Project (No. 2018SHZDZX01), ZJLab, and Shanghai Center for Brain Science and Brain-Inspired Technology; and National Key R&D Program of China (No. 2018YFC1312904). The funding agencies took no part in the design of this research.
Author information
Authors and Affiliations
Contributions
C–C Huang and E T Rolls prepared the atlas and wrote the paper. C-P Lin and J.Feng read and approved the paper, and provided funding.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Ethical Permissions
No data were collected as part of the research described here. The data were from the Human Connectome Project, and the WU-Minn HCP Consortium obtained full informed consent from all participants, and research procedures and ethical guidelines were followed in accordance with the Institutional Review Boards (IRB), with details at the HCP website (http://www.humanconnectome.org/).
Software and code for the extended HCP atlas
The HCPex atlas, including its different versions, labels, code, and the User guide, is available in association with this paper at the authors’ websites https://www.oxcns.org as HCPex_v1.0.zip and https://github.com/wayalan/HCPex.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huang, CC., Rolls, E.T., Feng, J. et al. An extended Human Connectome Project multimodal parcellation atlas of the human cortex and subcortical areas. Brain Struct Funct 227, 763–778 (2022). https://doi.org/10.1007/s00429-021-02421-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-021-02421-6