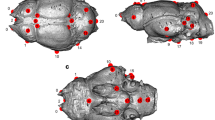
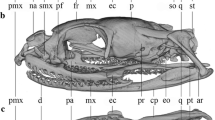
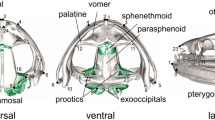
Abstract
Environmental properties, and the behavioral habits of species impact sensory cues available for foraging, predator avoidance and inter/intraspecific communication. Consequently, relationships have been discovered between the sensory ecology and brain morphology in many groups of vertebrates. However, these types of studies have remained scare on snake. Here, we investigate the link between endocranial shape and the sensory-related ecology of snakes by comparing 36 species of snakes for which we gathered six sensory-ecology characteristics. We use µCT scanning and 3D geometric morphometrics to compare their endocranium in a phylogenetically informed context. Our results demonstrate that size is a major driver of endocranial shape, with smaller species tending to maximize endocranial volume using a more bulbous shape, while larger species share an elongate endocranial morphology. Phylogeny plays a secondary role with more derived snakes diverging the most in endocranial shape, compared to other species. The activity period influences the shape of the olfactory and optic tract, while the foraging habitat impacts the shape of the cerebellum and cranial nerve regions: structures involved in orientation, equilibrium, and sensory information. However, we found that endocranial morphology alone is not sufficient to predict the activity period of a species without prior knowledge of its phylogenetic relationship. Our results thus demonstrate the value of utilizing endocranial shape as complementary information to size and volume in neurobiological studies.





Similar content being viewed by others
Data availability
The original landmarks coordinates for all specimens will be deposited in a Dryad Repository or added in the Supplementary Material and the 3D scans will be uploaded in MorphoSource.
Code availability
Available in supplementary material.
References
Adams DC (2014a) A generalized K statistic for estimating phylogenetic signal from shape and other high-dimensional multivariate data. Syst Biol 63:685–697. https://doi.org/10.1093/sysbio/syu030
Adams DC (2014b) A method for assessing phylogenetic least squares models for shape and other high-dimensional multivariate data. Evolution 68:2675–2688. https://doi.org/10.1111/evo.12463
Adams DC, Collyer ML, Kaliontzopoulou A (2020) Package “geomorph”, pp 1–140
Alfaro ME (2002) Forward attack modes of aquatic feeding garter snakes. Funct Ecol 16:204–215. https://doi.org/10.1046/j.1365-2435.2002.00620.x
Allemand R, Boistel R, Daghfous G et al (2017) Comparative morphology of snake (Squamata) endocasts: evidence of phylogenetic and ecological signals. J Anat 231:849–868. https://doi.org/10.1111/joa.12692
Balanoff AM, Bever GS (2017) The role of endocasts in the study of brain evolution. In: Kaas JH (ed) Evolution of nervous systems, Academic Press, pp 223–241
Balanoff AM, Bever GS, Colbert MW et al (2015) Best practices for digitally constructing endocranial casts: examples from birds and their dinosaurian relatives. J Anat 229:173–190. https://doi.org/10.1111/joa.12378
Balanoff AM, Smaers JB, Turner AH (2016) Brain modularity across the theropod–bird transition: testing the influence of flight on neuroanatomical variation. J Anat 229:204–214. https://doi.org/10.1111/joa.12403
Barton RA, Purvis A, Harvey PH (1995) Evolutionary radiation of visual and olfactory brain system in primates, bats and insectivores. Philos Trans R Soc B Biol Sci 348:381–392. https://doi.org/10.1098/rstb.1995.0076
Bertrand OC, San Martin-Flores G, Silcox MT (2019) Endocranial shape variation in the squirrel-related clade and their fossil relatives using 3D geometric morphometrics: contributions of locomotion and phylogeny to brain shape. J Zool 308:197–211. https://doi.org/10.1111/jzo.12665
Blomberg SP, Garland TJ, Ives AR (2003) Testing for phylogenetic signal in comparative data: behavioral traits are more labile. Evolution 57:717–745. https://doi.org/10.1111/j.0014-3820.2003.tb00285.x
Burghardt GM, Ford NB (1993) Perceptual mechanisms and the behavioral ecology of snakes. Snakes: ecology and behavior. The Blackburn Press, New Jersey, pp 117–164
Camilleri C, Shine R (1990a) Sexual dimorphism and dietary divergence : differences in trophic morphology between male and female snakes. Copeia 1990:649–658
Carril J, Tambussi CP, Degrange FJ et al (2016) Comparative brain morphology of Neotropical parrots (Aves, Psittaciformes) inferred from virtual 3D endocasts. J Anat 229:239–251. https://doi.org/10.1111/joa.12325
Catania KC, Leitch DB, Gauthier D (2010) Function of the appendages in tentacled snakes (Erpeton tentaculatus). J Exp Biol 213:359–367. https://doi.org/10.1242/jeb.039685
Cooper WEJ (2008) Tandem evolution of diet and chemosensory responses in snakes. Amphibia-Reptilia 29:393–398
Crowe-Riddell JM, Snelling EP, Watson AP et al (2016) The evolution of scale sensilla in the transition from land to sea in elapid snakes. Open Biol 6:160054. https://doi.org/10.1098/rsob.160054
Crowe-Riddell JM, Simões BF, Partridge JC et al (2019) Phototactic tails: evolution and molecular basis of a novel sensory trait in sea snakes. Mol Ecol 28:2013–2018. https://doi.org/10.1111/mec.15022
Czaplicki JA, Porter RH (1974) Visual cues mediating the selection of goldfish (Carassius auratus) by two species of Natrix. J Herpetol 8:129–134
Da Silva MAO, Heegaard S, Wang T et al (2017) Morphology of the snake spectacle reflects its evolutionary adaptation and development. BMC Vet Res 13:1–8. https://doi.org/10.1186/s12917-017-1193-2
Daghfous G, Smargiassi M, Libourel PA et al (2012) The function of oscillatory tongue-flicks in snakes: insights from kinematics of tongue-flicking in the banded water snake (Nerodia fasciata). Chem Senses 37:883–896. https://doi.org/10.1093/chemse/bjs072
De Cock BT (1983) Thermal sensitivity as a specialization for prey capture and feeding in snakes. Am Zool 23:363–375
Drummond HM (1985) The role of vision in the predatory behaviour of natricine snakes. Anim Behav 33:206–215. https://doi.org/10.1016/S0003-3472(85)80134-2
Ebert J, Westhoff G (2006) Behavioural examination of the infrared sensitivity of rattlesnakes (Crotalus atrox). J Comp Physiol A 192:941–947. https://doi.org/10.1007/s00359-006-0131-8
Ebert J, Müller S, Westhoff G (2007) Behavioural examination of the infrared sensitivity of ball pythons. J Zool 272:340–347. https://doi.org/10.1111/j.1469-7998.2006.00275.x
Franz R (1977) Observations on the food, feeding behavior, and parasites of the striped swamp snake, Regina alleni. Herpetologica 33:91–94
Friedel P, Young BA, Van Hemmen JL (2008) Auditory localization of ground-borne vibrations in snakes. Phys Rev Lett 100:048701. https://doi.org/10.1103/PhysRevLett.100.048701
Gunz P, Mitteroecker P (2013) Semilandmarks: a method for quantifying curves and surfaces. Hystrix 24:103–109. https://doi.org/10.4404/hystrix-24.1-6292
Gutiérrez-Ibáñez C, Iwaniuk AN, Wylie DR (2010) The independent evolution of the enlargement of the principal sensory nucleus of the trigeminal nerve in three different groups of birds. Brain Behav Evol 74:280–294. https://doi.org/10.1159/000270904
Halpern M, Kubie JL (1984) The role of the ophidian vomeronasal system in species-typical behavior. Trends Neurosci 7:472–477. https://doi.org/10.1016/S0166-2236(84)80258-1
Hart NS, Coimbra JP, Collin SP, Westhoff G (2012) Photoreceptor types, visual pigments, and topographic specializations in the retinas of Hydrophiid sea snakes. J Comp Neurol 520:1246–1261. https://doi.org/10.1002/cne.22784
Heatwole H (1999) Sea snakes. University of New South Wales Press Ltd, Kensington
Hibbard E, Lavergne J (1972) Morphology of the retina of the sea-snake, Pelamis platurus. J Anat 112:125–136
Holloway WL, Claeson KM, O’keefe FR (2013) A virtual phytosaur endocast and its implications for sensory system evolution in archosaurs. J Vertebr Paleontol 33:848–857. https://doi.org/10.1080/02724634.2013.747532
Iwaniuk AN (2017) Functional correlates of brain and brain region sizes in nonmammalian vertebrates. In: Kaas JH (ed) Evolution of nervous systems, Academic Press, pp 335–348
Iwaniuk AN, Mardon K, Leseberg NP, Weisbecker V (2020) The endocast of the Night Parrot (Pezoporus occidentalis) reveals insights into its sensory ecology and the evolution of nocturnality in birds. Sci Rep 10(9258):1–9. https://doi.org/10.1038/s41598-020-65156-0
Jayne BC, Voris HK, Ng PKL (2002) Snake circumvents constraints on prey size. Nature 418:143
Jayne BC, Voris HK, Ng PKL (2018) How big is too big? Using crustacean-eating snakes (Homalopsidae) to test how anatomy and behaviour affect prey size and feeding performance. Biol J Linn Soc 123:636–650. https://doi.org/10.1093/biolinnean/bly007
Kaas JH (2017) Evolution of nervous systems, 2nd edn. Academic Press
Kawabe S, Shimokawa T, Miki H et al (2013) Variation in avian brain shape: relationship with size and orbital shape. J Anat 223:495–508. https://doi.org/10.1111/joa.12109
Kotrschal K, van Staaden MJ, Huber R (1998) Fish brains: evolution and environmental relationships. Rev Fish Biol Fish 8:373–408. https://doi.org/10.1023/A:1008839605380
Krochmal AR, Bakken GS, LaDuc TJ (2004) Heat in evolution’s kitchen: evolutionary perspectives on the functions and origin of the facial pit of pitvipers (Viperidae: Crotalinae). J Exp Biol 207:4231–4238. https://doi.org/10.1242/jeb.01278
Kropach CN (1975) The yellow-bellied sea snake, Pelamis, in the eastern Pacific. In: Dunson WA (ed) The biology of sea snakes. University Park Press, Baltimore, pp 185–213
Kutsuma R, Sasai T, Kishida T (2018) How snakes find prey underwater: sea snakes use visual and chemical cues for foraging. Zoolog Sci 35:483–486. https://doi.org/10.2108/zs180059
Lautenschlager S, Rayfield EJ, Altangerel P et al (2012) The endocranial anatomy of Therizinosauria and its implications for sensory and cognitive function. PLoS ONE 7:e52289. https://doi.org/10.1371/journal.pone.0052289
Lebrun R (2017) ISE-MeshTools v1.3.4. pp 1–101
Liu Y, Ding L, Lei J et al (2012) Eye size variation reflects habitat and daily activity patterns in colubrid snakes. J Morphol 273:883–893. https://doi.org/10.1002/jmor.20028
Macrì S, Savriama Y, Khan I, Di-Poï N (2019) Comparative analysis of squamate brains unveils multi-level variation in cerebellar architecture associated with locomotor specialization. Nat Commun 10:5560. https://doi.org/10.1038/s41467-019-13405-w
Mark Waters R, Burghardt GM (2005) The interaction of food motivation and experience in the ontogeny of chemoreception in crayfish snakes. Anim Behav 69:363–374. https://doi.org/10.1016/j.anbehav.2004.03.014
Marugán-Lobón J, Watanabe A, Kawabe S (2016) Studying avian encephalization with geometric morphometrics. J Anat 229:191–203. https://doi.org/10.1111/joa.12476
Moon BR, Penning DA, Segall M, Herrel A (2019) Feeding in snakes: form, function, and evolution of the feeding system. In: Bels V, Whishaw IQ (eds) Feeding in vertebrates: evolution, morphology, behavior, biomechanics. Springer Nature, Switzerland, pp 528–574
Naumann RK, Ondracek JM, Reiter S et al (2015) The reptilian brain. Curr Biol 25:R317–R321. https://doi.org/10.1016/j.cub.2015.02.049
Newman EA, Hartline PH (1982) The infrared “vision” of snakes. Sci Am 246:116–127. https://doi.org/10.1038/scientificamerican0382-116
Nieuwenhuys R, ten Donkelaar HJ, Nicholson C (1998) The central nervous system of vertebrates. Springer, Berlin
Noonloy T, Kunya K, Chanhome L et al (2018) Crab-ripping: an unusual feeding behavior newly recorded in freshwater snakes. Bull Chicago Herpetol Soc 53:53–56
Olori JC (2010) Digital endocasts of the cranial cavity and osseous labyrinth of the burrowing snake Uropeltis woodmasoni (Alethinophidia: Uropeltidae). Copeia 2010:14–26. https://doi.org/10.1643/CH-09-082
Pawar S, Dell AI, Savage VM (2012) Dimensionality of consumer search space drives trophic interaction strengths. Nature 486:485–489. https://doi.org/10.1038/nature11131
Pedersen TL, Crameri F (2020) Package “scico”, pp 1–7
Povel DE, Van Der Kooij J (1997) Scale sensillae of the file snake (Serpentes: Acrochordidae) and some other aquatic and burrowing snakes. Netherlands J Zool 47:443–456. https://doi.org/10.1017/CBO9781107415324.004
Pyron RA, Burbrink FT (2014) Early origin of viviparity and multiple reversions to oviparity in squamate reptiles. Ecol Lett 17:13–21. https://doi.org/10.1111/ele.12168
Randall JA, Matocq MD (1997) Why do kangaroo rats (Dipodomys spectabilis) footdrum at snakes? Behav Ecol 8:404–413. https://doi.org/10.1093/beheco/8.4.404
Revell LJ (2012) Phytools: an R package for phylogenetic comparative biology (and other things), pp 217–223
Ripley B, Venables W (2020) Package ‘class’, pp 1–19
Schaeffel F, de Queiroz A (1990) Alternative mechanisms of enhanced underwater vision in the garter snakes Thamnophis melanogaster and T. couchii. Copeia 1990:50–58
Schaeffel F, Mathis U (1991) Underwater vision in semi-aquatic European snakes. Naturwissenschaften 78:373–375. https://doi.org/10.1007/BF01131614
Schlager S (2017) Morpho and Rvcg–Shape analysis in R: R-Packages for geometric morphometrics, shape analysis and surface manipulations. In: Zheng G, Li S, Szekely G (eds) Statistical shape and deformation analysis: methods implementation and applications. Academic Press, pp 217–256
Schott RK, Van Nynatten A, Card DC et al (2018) Shifts in selective pressures on snake phototransduction genes associated with photoreceptor transmutation and dim-light ancestry. Mol Biol Evol 35:1376–1389. https://doi.org/10.1093/molbev/msy025/4904158
Schwenk K (2008) Comparative anatomy and physiology of chemical senses in nonavian aquatic reptiles. In: Thewissen JGM, Nummela S (eds) Sensory evolution on the threshold adaptations in secondarily aquatic vertebrates. University of California Press, pp 65–81
Sepulcre J, Liu H, Talukdar T et al (2010) The organization of local and distant functional connectivity in the human brain. PLoS Comput Biol 6:1–15. https://doi.org/10.1371/journal.pcbi.1000808
Shine R (2005) All at sea: Aquatic life modifies mate-recognition modalities in sea snakes (Emydocephalus annulatus, Hydrophiidae). Behav Ecol Sociobiol 57:591–598. https://doi.org/10.1007/s00265-004-0897-z
Shine R, Bonnet X, Elphick MJ, Barrott EG (2004a) A novel foraging mode in snakes: browsing by the sea snake Emydocephalus annulatus (Serpentes, Hydrophiidae). Funct Ecol 18:16–24. https://doi.org/10.1046/j.0269-8463.2004.00803.x
Shine R, Brown GP, Elphick MJ (2004b) Field experiments on foraging in free-ranging water snakes Enhydris polylepis (Homalopsinae). Anim Behav 68:1313–1324. https://doi.org/10.1016/j.anbehav.2004.03.004
Simões BF, Sampaio FL, Douglas RH et al (2016) Visual pigments, ocular filters and the evolution of snake vision. Mol Biol Evol 33:2483–2495. https://doi.org/10.1093/molbev/msw148
Simões BF, Gower DJ, Rasmussen AR et al (2020) Spectral diversification and trans-species allelic polymorphism during the land-to-sea transition in snakes. Curr Biol 30:2608–2615. https://doi.org/10.1016/j.cub.2020.04.061
Skinner M, Miller N (2020) Aggregation and social interaction in garter snakes (Thamnophis sirtalis sirtalis). Behav Ecol Sociobiol 74:51. https://doi.org/10.1007/s00265-020-2827-0
Smargiassi M, Daghfous G, Leroy B et al (2012) Chemical basis of prey recognition in Thamnophiine snakes: the unexpected new roles of parvalbumins. PLoS ONE 7:7–14. https://doi.org/10.1371/journal.pone.0039560
Starck D (1979) Cranio-cerebral relations in recent reptiles. Biology of the reptilia, 9 neurology A. Academic Press Inc. Ltd, London, pp 1–38
Stevens M (2013) Sensory ecology, behavior, and evolution. Oxford University Press, Oxford
Thewissen JGM, Nummela S (2008) Sensory evolution on the threshold: adaptations in secondarily aquatic vertebrates. University of California Press, London
Vincent SE, Shine R, Brown GP (2005) Does foraging mode influence sensory modalities for prey detection in male and female filesnakes, Acrochordus arafurae? Anim Behav 70:715–721. https://doi.org/10.1016/j.anbehav.2005.01.002
Voris HK, Voris HH, Liat LB (1978) The food and feeding behavior of a marine snake, Enhydrina schistosa (Hydrophiidae). Copeia 1978:134–146
Weisbecker V, Rowe T, Wroe S et al (2021) Global elongation and high shape flexibility as an evolutionary hypothesis of accommodating mammalian brains into skulls. Evolution (n Y) 75:625–640. https://doi.org/10.1111/evo.14163
Westhoff G, Fry BG, Bleckmann H (2005) Sea snakes (Lapemis curtus) are sensitive to low-amplitude water motions. Zoology 108:195–200. https://doi.org/10.1016/j.zool.2005.07.001
Yopak KE, Pakan JMP, Wylie D (2017) The cerebellum of nonmammalian vertebrates. In: Evolution of nervous systems, 2nd edn, Elsevier, pp 373–385
Young BA (2003) Snake bioacoustics: toward a richer understanding of the behavioral ecology of snakes. Q Rev Biol 78:303–325. https://doi.org/10.1360/zd-2013-43-6-1064
Young BA (2007) Response of the yellow anaconda (Eunectes notaeus) to aquatic acoustic stimuli. In: Henderson RW, Powell R (eds) Biology of the boas and pythons. Eagle Mountain Publishing, Utah, pp 199–205
Young BA, Aguiar A, Lillywhite HB (2008) Foraging cues used by insular Florida cottonmouths, Agkistrodon piscivorus conanti. South Am J Herpetol 3:135–144. https://doi.org/10.2994/1808-9798(2008)3[135:fcubif]2.0.co;2
Acknowledgements
We thank the Fyssen foundation for funding this study. Special thanks to the herpetological collections staff of the American Museum of Natural History: David Kizirian, David Dickey, Margaret Arnold and especially Lauren Vonnahme, but also Alan Resetar (Field Museum of Natural History), Erica Ely and Lauren Scheinberg (California Academy of Sciences) for their help and patience in carefully choosing specimens that fit our study and quickly processing specimen loans. Another special thanks to Morgan Hill Chase and Andrew Smith, from the Microscopy and Imaging and Facility who did all the CT scanning involved in this study. We would like to warmly thank Vera Weisbecker and an anonymous reviewer for their comments on our manuscript that greatly improved its quality, but also for their insightful comments on endocranial evolution in other vertebrate groups.
Funding
We thank the Fyssen Foundation for funding this research.
Author information
Authors and Affiliations
Contributions
MS conceived the ideas, designed the methodology and collected the data. MS and RC analyzed the data. MS led the writing of the manuscript. All authors contributed to the interpretation and discussion of the results, and to the editing of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
Not applicable to this study.
Consent to participate
Not applicable to this study.
Consent for publication
All authors contributed critically to the drafts and gave final approval for publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Segall, M., Cornette, R., Rasmussen, A.R. et al. Inside the head of snakes: influence of size, phylogeny, and sensory ecology on endocranium morphology. Brain Struct Funct 226, 2401–2415 (2021). https://doi.org/10.1007/s00429-021-02340-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-021-02340-6