Abstract
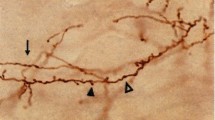
The subcortical white matter (SWM) has been traditionally considered as a site for passive–neutral–information transfer through cerebral cortex association and projection fibers. Yet, the presence of subcortical neuronal and glial “interstitial” cells expressing immunolabelled neurotransmitters/neuromodulators and synaptic vesicular proteins, and recent immunohistochemical and electrophysiological observations on the rat visual cortex as well as interactive regulation of myelinating processes support the possibility that SWM nests subcortical, regionally variable, distributed neuronal–glial circuits, that could influence information transfer. Their hypothetical involvement in regulating the timing and signal transfer probability at the SWM axonal components ought to be considered and experimentally analysed. Thus, the “interstitial” neuronal cells—associated with local glial cells—traditionally considered to be vestigial and functionally inert under normal conditions, they may well turn to be critical in regulating information transfer at the SWM.
Similar content being viewed by others
References
Akbarian S, Kim JJ, Potkin SG, Hetrick WP, Bunney WE Jr, Jones EG (1996) Maldistribution of interstitial neurons in prefrontal white matter of the brains of schizophrenic patients. Arch Gen Psych 53:425–436
Anderson SA, Volk DW, Lewis DA (1996) Increased density of microtubule associated protein 2-immunoreactive neurons in the prefrontal white matter of schizophrenic subjects. Schizophr Res 19:111–119
Bezzi P, Gundersen V, Galbete JL, Seifert G, Steinhäuser C, Pilati E, Volterra A (2004) Astrocytes contain a vesicular compartment that is competent for regulated exocytosis of glutamate. Nat Neurosci 7:613–620
Butt AM, Fern RF, Matute C (2014) Neurotransmitter signaling in white matter. Glia 62:1762–1779
Chun JJM, Shatz CJ (1989) Interstitial cells of the adult neocortical white matter are the remnant of the early generated subplate neuron population. J Comp Neurol 282:555–569
Colombo JA, Bentham C (2006) Immunohistochemical analysis of subcortical white matter astroglia of infant and adult primates, with a note on resident neurons. Brain Res 1100:93–103
Fields RD (2010) Change in the Brain’s white matter. Science 330:768–769
Fields RD (2015) A new mechanism of nervous system plasticity: activity-dependent myelination. Nat Rev Neurosci 16:756–767
Ford MC, Alexandrova O, Cossell L, Stange-Marten A, Sinclair J, Kopp-Scheinpflug C, Pecka M, Attwell D, Grothe B (2015) Tuning of Ranvier node and internode properties in myelinated axons to adjust action potential timing. Nat Commun 6:8073. https://doi.org/10.1038/ncomms9073
Friedlander MJ, Torres-Reveron J (2009) The changing roles of neurons in the cortical subplate. Front Neuroanat. https://doi.org/10.3389/neuro.05.015.2009
García-Marin V, Blazquez-Llorca L, Rodriguez JR, Gonzalez-Soriano J, DeFelipe J (2010) Differential distribution of neurons in the gyral white matter of the human cerebral cortex. J Comp Neurol 518:4740–4759. https://doi.org/10.1002/cne.22485
Gusel’nikova VV, Korzhevskiy DE (2015) NeuN as a neuronal nuclear antigen and neuron differentiaition marker. Acta Naturae 7:42–47
Hendry SHC, Jones EG, Emsons PC (1984) Morphology, distribution, and synaptic relations of somatostatin-and neuropeptide Y-immunoreactive neurons in rat and monkey neocortex. J Neurosci 4:2497–2517
Jeftinija SD, Jeftinija KV, Stefanovic G (1997) Cultured astrocytes express proteins involved in vesicular glutamate release. Brain Res 750:41–47
Judas M, Sedmak G, Pletikos M, Jovanov-Milosevic N (2010) Populations of subplate and interstitial neurons in fetal and adult human telecephalon. J Anat 217:381–399
Kirkpatrick B, Messias NC, Conley RR, Roberts RC (2003) Interstitial cells of the white matter in the dorsolateral prefrontal cortex in deficit and nondeficit schizophrenia. J Nerv Ment Dis 191:563–567
Kostovic I, Rakic P (1980) Cytology and time of origin of interstitial neurons in the white matter in infant and adult human and monkey telencephalon. J Neurocytol 9:219–242
Kostovic I, Rakic P (1990) Developmental history of the transient subplate zone in the visual and somatosensory cortex of the macaque momkey and human brain. J Comp Neurol 297:441–470
Kriegler S, Chiu SY (1993) Calcium signaling of glial cells along mammalian axons. J Neurosci 13:4229–4245
Kukley M, Capetillo-Zarate E, Dietrich D (2007) Vesicular glutamate release from axons in white matter. Nat Neurosci 10:311–320
Maienschein V, Marxen M, Volknandt W, Zimmermann H (1999) A plethora of presynaptic proteins associated with ATP-storing organelles in cultured astrocytes. Glia 26:233–244
Mortazavi F, Wang X, Rosene DL, Rockland KS (2016) White matter neurons in young adult and aged Rhesus minkeys. Front Neuroanat. https://doi.org/10.3389/fnana.2016.00015
Mortazavi F, Romano SE, Rosene DL, Rockland KS (2017) A survey of white matter neurons at the gyral crowns and sulcal depths in the rhesus monkey. Front Neuroanat. https://doi.org/10.3389/fnana.2017.00069
Mullen RJ, Buck CR, Smith AM (1992) NeuN, a neuronal specific nuclear protein in vertebrates. Development 116:201–211
Palmero-Gallagher N, Zilles K (2017) Cortical layers: cyto-, myelo-, receptor- and synaptic architecture in human cortical areas. Neuroimage. https://doi.org/10.1016/j.neuroimage.2017.08.035
Parpura V, Fang Y, Basarsky T, Jahn R, Haydon PG (1995) Expression of synaptobrevin II, cellubrevin and syntaxin but not SNAP-25 in cultured astrocytes. FEBS Lett 377:489–492
Rosier A, Arckens L, Orban GA, Vandesande F (1993) Immunocytochemical detection of astrocyte GABAA receptors in cat visual cortex. J Histochem Cytochem 41:685–692
Sarnat HB, Nochlin D, Born DE (1998) Neuronal nuclear antigen (NeuN): a marker of neuronal maturation in early human fetal nervous system. Brain Dev 20:88–94
Seidl AH (2014) Regulation of conduction time along axons. Neuroscience 276:126–134. https://doi.org/10.1016/j.neuroscience.2013.06.047
Steinhauser C, Gallo V (1996) News on glutamate receptors in glial cells. Trends Neurosci 19:339–345
Susuki K, Kuba H (2016) Activity-dependent regulation of excitable axonal domains. J Physiol Sci 66:99–104. https://doi.org/10.1007/s12576-015-0413-4
Torres-Reveron JE, Friedlander MJ (2007) Properties of persistent postnatal cortical subplate neurons. J Neurosci 27:9962–9974
Valverde F, Facal-Valverde MV (1988) Postnatal development of interstitial (subplate) cells in the white matter of the temporal cortex of kittens: a correlated Golgi and electron microscopic study. J Comp Neurol 269:168–192
Verkhratsky A, Butt A (2013) Glial physiology and pathophysiology. Wiley, UK
Wake H, Lee PR, Fields RD (2011) Control of local protein synthesis and initial events in myelination by action potentials. Science 333:1647–1651
Wake H, Ortiz FC, Woo DH, Lee PR, Angulo MC, Fields RD (2015) Nonsynaptic junctions on myelinating glia promote preferential myelination of electrically active axons. Nat Commun. https://doi.org/10.1038/ncomms8844
Waxman SG, Ritchie JM (1996) Molecular dissection of the myelinated axon. Ann Neurol 33:121–136
Yang Y, Fung SJ, Rothwell A, Tianmei S, Weickert CS (2011) Increased interstitial white matter neuron density in the DLPFC of people with schizophrenia. Biol Psychiat 69:63–70
Zhang C-L, Wilson JA, Williams J, Chiu SY (2006) Action potentials induce uniform calcium influx in mammalian myelinated optic nerves. J Neurophysiol 96:695–609. https://doi.org/10.1152/jn.00083.2006
Acknowledgements
Support by Fundación Conectar.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author declares compliance with ethical standards.
Conflict of interest
The author declares no conflict of interest.
Rights and permissions
About this article
Cite this article
Colombo, J.A. Cellular complexity in subcortical white matter: a distributed control circuit?. Brain Struct Funct 223, 981–985 (2018). https://doi.org/10.1007/s00429-018-1609-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-018-1609-1