Abstract
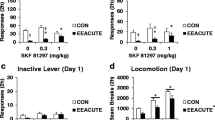
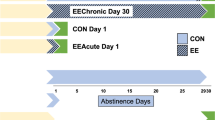
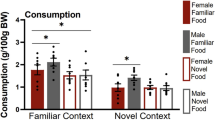
Exposure to environmental enrichment (EE) reduces sucrose seeking by rats with a history of sucrose self-administration. The present experiment examined whether acute or chronic EE also reduces brain Fos levels, a protein marker indicative of neuronal activation. Fos levels were also examined after either 1 or 30 days of forced abstinence to examine whether Fos levels vary with the incubation of sucrose craving. Fos expression was examined in 18 regions and was identified in brain slices using immunohistochemistry. Fos levels were higher in most regions after 30 days of forced abstinence and were decreased in most regions by either acute or chronic EE. Eleven regions had some statistically significant effect and/or interaction of EE or incubation on Fos; the most salient of these are listed here. In the prelimbic cortex, there was an incubation of Fos and EE reduced Fos at both forced abstinence time points. In contrast, in the orbitofrontal cortex, there was no Fos incubation but EE reduced Fos at both forced abstinence time points. An interaction of EE and incubation was observed in the anterior cingulate cortex and nucleus accumbens core and shell where Fos incubated but EE only decreased Fos at the day 30 forced abstinence time point. In contrast, in the dorsolateral striatum Fos incubated, but EE robustly decreased Fos expression at both forced abstinence time points. These differential expression patterns provide rationale for more detailed, site-specific molecular functional studies in how they relate to the ability of EE to reduce sucrose seeking.







Similar content being viewed by others
References
Alexander BK, Coambs RB, Hadaway PF (1978) The effect of housing and gender on morphine self-administration in rats. Psychopharmacology 58:175–179
Bardo MT, Klebaur JE, Valone JM, Deaton C (2001) Environmental enrichment decreases intravenous self-administration of amphetamine in female and male rats. Psychopharmacology 155:278–284
Belin D, Belin-Rauscent A, Murray JE, Everitt BJ (2013) Addiction: failure of control over maladaptive incentive habits. Curr Opin Neurobiol 23:564–572
Bennett LW, Cardone S, Jarczyk J (1998) Effects of a therapeutic camping program on addiction recovery. The algonquin haymarket relapse prevention program. J Subst Abuse Treat 15:469–474
Bindra D (1972) A unified account of classical conditioning and operant training. In: Black HH, Prokasy WF (eds) Classical conditioning II: current research and theory. Appleton-Century-Croft, New York, pp 453–481
Bossert JM, Marchant NJ, Calu DJ, Shaham Y (2013) The reinstatement model of drug relapse: recent neurobiological findings, emerging research topics, and translational research. Psychopharmacology 229:453–476
Brenes JC, Fornaguera J (2008) Effects of environmental enrichment and social isolation on sucrose consumption and preference: associations with depressive-like behavior and ventral striatum dopamine. Neurosci Lett 436:278–282
Carroll ME (1993) The economic context of drug and non-drug reinforcers affects acquisition and maintenance of drug-reinforced behavior and withdrawal effects. Drug Alcohol Depend 33:201–210
Carter BL, Tiffany ST (1999) Meta-analysis of cue-reactivity in addiction research. Addiction 94:327–340
CDC (2014) Overweight and obesity. http://www.cdc.gov/obesity/data/adult.html Accessed February 20, 2015
Chauvet C, Lardeux V, Goldberg SR, Jaber M, Solinas M (2009) Environmental enrichment reduces cocaine seeking and reinstatement induced by cues and stress but not by cocaine. Neuropsychopharmacology 34:2767–2778
Chauvet C, Lardeux V, Jaber M, Solinas M (2011) Brain regions associated with the reversal of cocaine conditioned place preference by environmental enrichment. Neuroscience 184:88–96
Chauvet C, Goldberg SR, Jaber M, Solinas M (2012) Effects of environmental enrichment on the incubation of cocaine craving. Neuropharmacology 63:635–641
Childress AR, Mozley PD, McElgin W, Fitzgerald J, Reivich M, O’Brien CP (1999) Limbic activation during cue-induced cocaine craving. Am J Psychiatry 156:11–18
Cutter CJ, Schottenfeld RS, Moore BA, Ball SA, Beitel M, Savant JD, Stults-Kolehmainen MA, Doucette C, Barry DT (2014) A pilot trial of a videogame-based exercise program for methadone maintained patients. J Subst Abuse Treat 47:299–305
Epstein DH, Willner-Reid J, Vahabzadeh M, Mezghanni M, Lin JL, Preston KL (2009) Real-time electronic diary reports of cue exposure and mood in the hours before cocaine and heroin craving and use. Arch Gen Psychiatry 66:88–94
Gill MJ, Cain ME (2011) Effects of satiety on operant responding in rats raised in enrichment. Behav Pharmacol 22:40–48
Green TA, Gehrke BJ, Bardo MT (2002) Environmental enrichment decreases intravenous amphetamine self-administration in rats: dose-response functions for fixed- and progressive-ratio schedules. Psychopharmacology 162:373–378
Grimm JW, Kruzich PJ, See RE (2000) Contingent access to stimuli associated with cocaine self-administration is required for reinstatement of drug-seeking behavior. Psychobiology 28:383–386
Grimm JW, Osincup D, Wells B, Manaois M, Fyall A, Buse C, Harkness JH (2008) Environmental enrichment attenuates cue-induced reinstatement of sucrose seeking in rats. Behav Pharmacol 19:777–785
Grimm JW, Weber R, Barnes J, Koerber J, Dorsey K, Glueck E (2013) Brief exposure to novel or enriched environments reduces sucrose cue-reactivity and consumption in rats after 1 or 30 days of forced abstinence from self-administration. PLoS ONE 8:e54164
Harkness JH, Webb S, Grimm JW (2010) Abstinence-dependent transfer of lithium chloride-induced sucrose aversion to a sucrose-paired cue in rats. Psychopharmacology 208:521–530
Herrera DG, Robertson HA (1996) Activation of c-fos in the brain. Prog Neurobiol 50:83–107
Hope BT, Nye HE, Kelz MB, Self DW, Iadarola MJ, Nakabeppu Y, Duman RS, Nestler EJ (1994) Induction of a long-lasting AP-1 complex composed of altered Fos-like proteins in brain by chronic cocaine and other chronic treatments. Neuron 13:1235–1244
Jansen A, Theunissen N, Slechten K, Nederkoorn C, Boon B, Mulkens S, Roefs A (2003) Overweight children overeat after exposure to food cues. Eat Behav 4:197–209
Johnson MW, Bickel WK (2003) The behavioral economics of cigarette smoking: the concurrent presence of a substitute and an independent reinforcer. Behav Pharmacol 14:137–144
Johnson RK, Appel LJ, Brands M, Howard BV, Lefevre M, Lustig RH, Sacks F, Steffen LM, Wylie-Rosett J (2009) Dietary sugars intake and cardiovascular health: a scientific statement from the American heart association. Circulation 120:1011–1020
Kesner RP, Churchwell JC (2011) An analysis of rat prefrontal cortex in mediating executive function. Neurobiol Learn Mem 96:417–431
Konkle AT, Kentner AC, Baker SL, Stewart A, Bielajew C (2010) Environmental-enrichment-related variations in behavioral, biochemical, and physiologic responses of Sprague-Dawley and Long Evans rats. J Am Assoc Lab Anim Sci 49:427–436
Koob GF (2009) Brain stress systems in the amygdala and addiction. Brain Res 1293:61–75
Kupferschmidt DA, Klas PG, Erb S (2012) Cannabinoid CB1 receptors mediate the effects of corticotropin-releasing factor on the reinstatement of cocaine seeking and expression of cocaine-induced behavioural sensitization. Br J Pharmacol 167:196–206
Laddu D, Dow C, Hingle M, Thomson C, Going S (2011) A review of evidence-based strategies to treat obesity in adults. Nutr Clin Pract 26:512–525
LaLumiere RT, Smith KC, Kalivas PW (2012) Neural circuit competition in cocaine-seeking: roles of the infralimbic cortex and nucleus accumbens shell. Eur J Neurosci 35:614–622
Ma YY, Lee BR, Wang X, Guo C, Liu L, Cui R, Lan Y, Balcita-Pedicino JJ, Wolf ME, Sesack SR, Shaham Y, Schluter OM, Huang YH, Dong Y (2014) Bidirectional modulation of incubation of cocaine craving by silent synapse-based remodeling of prefrontal cortex to accumbens projections. Neuron 83:1453–1467
McDannald MA, Jones JL, Takahashi YK, Schoenbaum G (2014) Learning theory: a driving force in understanding orbitofrontal function. Neurobiol Learn Mem 108:22–27
Mendelson JH, Mello NK (1996) Management of cocaine abuse and dependence. N Engl J Med 334:965–972
Nader J, Claudia C, Rawas RE, Favot L, Jaber M, Thiriet N, Solinas M (2012) Loss of environmental enrichment increases vulnerability to cocaine addiction. Neuropsychopharmacology 37:1579–1587
O’Brien CP, Childress AR, Arndt IO, McLellan AT, Woody GE, Maany I (1988) Pharmacological and behavioral treatments of cocaine dependence: controlled studies. J Clin Psychiatry 49(Suppl):17–22
Ogden CL, Yanovski SZ, Carroll MD, Flegal KM (2007) The epidemiology of obesity. Gastroenterology 132:2087–2102
Paxinos G, Watson C (2007) The rat brain in stereotaxic coordinates, 6th edn. Burlington, Elsevier
PHS (2002) Policy on humane care and use of laboratory animals. http://grants.nih.gov/grants/olaw/references/phspol.htm Accessed February 20, 2015
Reynolds GS (1961) Behavioral contrast. J Exp Anal Behav 4:57–71
Shalev U, Grimm JW, Shaham Y (2002) Neurobiology of relapse to heroin and cocaine seeking: a review. Pharmacol Rev 54:1–42
Shaw RG, Mitchell-Olds T (1993) ANOVA for unbalanced data: an overview. Ecology 74:1638–1645
Shenhav A, Botvinick MM, Cohen JD (2013) The expected value of control: an integrative theory of anterior cingulate cortex function. Neuron 79:217–240
Sobik L, Hutchison K, Craighead L (2005) Cue-elicited craving for food: a fresh approach to the study of binge eating. Appetite 44:253–261
Solinas M, Thiriet N, Chauvet C, Jaber M (2010) Prevention and treatment of drug addiction by environmental enrichment. Prog Neurobiol 92:572–592
Thiel KJ, Sanabria F, Pentkowski NS, Neisewander JL (2009) Anti-craving effects of environmental enrichment. Int J Neuropsychopharmacol 12:1151–1156
Thiel KJ, Pentkowski NS, Peartree NA, Painter MR, Neisewander JL (2010) Environmental living conditions introduced during forced abstinence alter cocaine-seeking behavior and Fos protein expression. Neuroscience 171:1187–1196
Thiel KJ, Painter MR, Pentkowski NS, Mitroi D, Crawford CA, Neisewander JL (2011) Environmental enrichment counters cocaine abstinence-induced stress and brain reactivity to cocaine cues but fails to prevent the incubation effect. Addict Biol 17:365–377
Tuten M, Fitzsimons H, Chisolm MS, Nuzzo PA, Jones HE (2012) Contingent incentives reduce cigarette smoking among pregnant, methadone-maintained women: results of an initial feasibility and efficacy randomized clinical trial. Addiction 107:1868–1877
Velez-Hernandez ME, Padilla E, Gonzalez-Lima F, Jimenez-Rivera CA (2014) Cocaine reduces cytochrome oxidase activity in the prefrontal cortex and modifies its functional connectivity with brainstem nuclei. Brain Res 1542:56–69
Volkow ND, Tomasi D, Wang GJ, Fowler JS, Telang F, Goldstein RZ, Alia-Klein N, Wong C (2011) Reduced metabolism in brain “control networks” following cocaine-cues exposure in female cocaine abusers. PLoS One 6:e16573
Wadden TA (1993) Treatment of obesity by moderate and severe caloric restriction. Results of clinical research trials. Ann Intern Med 119:688–693
WHO (2015) Obesity and overweight. http://www.who.int/mediacentre/factsheets/fs311/en/Accessed February 20, 2015
Wise RA (2004) Dopamine, learning and motivation. Nat Rev Neurosci 5:483–494
Zhou L, Pruitt C, Shin CB, Garcia AD, Zavala AR, See RE (2013) Fos expression induced by cocaine-conditioned cues in male and female rats. Brain Struct Funct 219:1831–1840
Acknowledgments
The authors wish to thank Kylan Dorsey and Dr. Blair Duncan for help with data collection and Dr. Bruce Hope for technical advice on immunohistochemistry. This study was supported by national institutes of health grant DA016285-03 and Western Washington University.
Conflict of interest
The authors declare that they have no conflict of interest. The manuscript does not contain clinical studies or patient data.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Grimm, J.W., Barnes, J.L., Koerber, J. et al. Effects of acute or chronic environmental enrichment on regional Fos protein expression following sucrose cue-reactivity testing in rats. Brain Struct Funct 221, 2817–2830 (2016). https://doi.org/10.1007/s00429-015-1074-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-015-1074-z