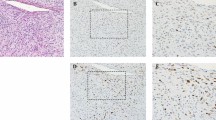
Abstract
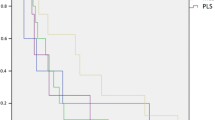
Currently, it is difficult to predict the prognosis of myxoid liposarcoma (MLS) in biopsy specimens. In this study, we determined whether nuclear morphology may be used to predict the prognosis of MLS in primary biopsy specimens. Two pathologists evaluated nuclear morphology using the modified WHO/ISUP and Fuhrman grades. Survival analyses were performed by grouping nuclear high- and low-grades. We examined 53 MLS cases, which included 29 (54.7%) male and 24 (45.3%) female patients with a median age of 46 years (interquartile range, 37 − 60). In total, 7 (13.2%) and 16 (30.2%) cases were assigned to the high nuclear grade group based on the modified WHO/ISUP and Fuhrman gradings, respectively. Survival analyses revealed a significantly worse disease-free survival in the high-grade group (hazard ratio (HR), 7.51; 95% confidence interval (CI), 2.67–21.1, p < 0.001 by the modified WHO/ISUP grading; HR, 4.45; 95% CI, 1.63–12.1, p = 0.001 by the modified Fuhrman grading). Moreover, the modified WHO/ISUP grade showed a significantly worse overall survival in the high-grade group (HR, 4.39; 95% CI, 1.04–18.6, p = 0.028), and the modified Fuhrman grade exhibited a similar, but not significant, trend. Our results indicate that nuclear morphology grading is a good predictor of patient prognosis at the time of biopsy in MLS. Even when cell density is sparse, treatment strategies should be carefully considered when individual tumor cells exhibit atypical nuclei.



Similar content being viewed by others
References
Goldblum J, Weiss S, Folpe AL (2019) Enzinger & Weiss’s soft tissue tumors, 7th edn. Elsevier, Amsterdam
WHO Classification of Tumours Editorial Board (2020) Soft tissue and bone tumours (5th edn). IARC Press
Penel N, Coindre JM, Giraud A, Terrier P, Ranchere-Vince D, Collin F, Guellec SLE, Bazille C, Lae M, de Pinieux G, Ray-Coquard IL, Bonvalot S, Cesne ALE, Robin YM, Stoeckle E, Toulmonde M, Blay JY (2018) Presentation and outcome of frequent and rare sarcoma histologic subtypes a study of 10,262 patients with localized visceral/soft tissue sarcoma managed in reference centers. Cancer 124:1179-1187. https://doi.org/10.1002/cncr.31176
Hoffman A, Ghadimi MP, Demicco EG, Creighton CJ, Torres K, Colombo C, Peng T, Lusby K, Ingram D, Hornick JL, Wang WL, Ravi V, Lazar AJ, Lev D, Pollock RE (2013) Localized and metastatic myxoid/round cell liposarcoma: clinical and molecular observations. Cancer 119:1868–1877. https://doi.org/10.1002/cncr.27847
Moreau LC, Turcotte R, Ferguson P, Wunder J, Clarkson P, Masri B, Isler M, Dion N, Werier J, Ghert M, Deheshi B, Canadian Orthopaedic Oncology S (2012) Myxoid/round cell liposarcoma MRCLS revisited an analysis of 418 primarily managed cases. Ann Surg Oncol 19:1081-1088. https://doi.org/10.1245/s10434-011-2127-z
Haniball J, Sumathi VP, Kindblom LG, Abudu A, Carter SR, Tillman RM, Jeys L, Spooner D, Peake D, Grimer RJ (2011) Prognostic factors and metastatic patterns in primary myxoid/round-cell liposarcoma. Sarcoma 2011:538085. https://doi.org/10.1155/2011/538085
Kilpatrick SE, Doyon J, Choong PFM, Sim FH, Nascimento AG (1996) The clinicopathologic spectrum of myxoid and round cell liposarcoma: a study of 95 cases. Cancer 1450–1458
Nezu Y, Hagiwara K, Yamamoto Y, Fujiwara T, Matsuo K, Yoshida A, Kawai A, Saito T, Ochiya T (2016) miR-135b, a key regulator of malignancy, is linked to poor prognosis in human myxoid liposarcoma Oncogene 35:6177–6188. https://doi.org/10.1038/onc.2016.157
ten Heuvel SE, Hoekstra HJ, van Ginkel RJ, Bastiaannet E, Suurmeijer AJ (2007) Clinicopathologic prognostic factors in myxoid liposarcoma: a retrospective study of 49 patients with long-term follow-up. Ann Surg Oncol 14:222-229. https://doi.org/10.1245/s10434-006-9043-7
Kawaguchi K, Kohashi K, Iwasaki T, Yamamoto T, Ishihara S, Toda Y, Yamamoto H, Nakashima Y, Oda Y (2023) Prognostic value of nuclear morphometry in myxoid liposarcoma. Cancer Sci 114:2178–2188. https://doi.org/10.1111/cas.15729
WHO Classification of Tumours Editorial Board (2020) Urinary and male genital tumours (5th edn). IARC Press
Fuhrman SA, Lasky LC, Limas C (1982) Prognostic significance of morphologic parameters in renal cell carcinoma. Am J Surg Pathol 6:655–663. https://doi.org/10.1097/00000478-198210000-00007
Chung PW, Deheshi BM, Ferguson PC, Wunder JS, Griffin AM, Catton CN, Bell RS, White LM, Kandel RA, O’Sullivan B (2009) Radiosensitivity translates into excellent local control in extremity myxoid liposarcoma: a comparison with other soft tissue sarcomas. Cancer 115:3254–3261. https://doi.org/10.1002/cncr.24375
Baxter KJ, Govsyeyev N, Namm JP, Gonzalez RJ, Roggin KK, Cardona K (2015) Is multimodality therapy necessary for the management of pure myxoid liposarcomas? A multi-institutional series of pure myxoid liposarcomas of the extremities and torso. J Surg Oncol 111:146–151. https://doi.org/10.1002/jso.23786
Fischer EG (2020) Nuclear morphology and the biology of cancer cells acta cytol. 64:511-519. https://doi.org/10.1159/000508780
Jevtic P, Levy DL (2014) Mechanisms of nuclear size regulation in model systems and cancer. Adv Exp Med Biol 773:537–569. https://doi.org/10.1007/978-1-4899-8032-8_25
Chow KH, Factor RE, Ullman KS (2012) The nuclear envelope environment and its cancer connections. Nat Rev Cancer 12:196–209. https://doi.org/10.1038/nrc3219
Acknowledgements
The authors would like to thank Enago (www.enago.com) for the English language review.
Funding
This work was supported by the Grants-in-Aid for Scientific Research of Japan Society for the Promotion of Science (grant numbers: JP19H03444 and JP21K06887).
Author information
Authors and Affiliations
Contributions
Kengo Kawaguchi, Kenichi Kohashi, and Yoshinao Oda: study concept and design; Kengo Kawaguchi, Kenichi Kohashi, Takeshi Iwasaki, Yasuharu Nakashima, and Yoshinao Oda: development of methodology and writing, review, and revision of the paper; Kengo Kawaguchi, Kenichi Kohashi, and Takeshi Iwasaki: analysis and interpretation of data and statistical analysis; Taro Mori, Hiroshi Furukawa, Chiaki Sato, Hiroki Sonoda, Sakura Shiraishi, and Makoto Endo: acquisition of data and technical and material support; Kengo Kawaguchi and Kenichi Kohashi: writing the manuscript; Kenichi Kohashi, and Yoshinao Oda: funding. All authors read and approved the final paper.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by the Kyushu University Committee of Bioethics (approval number: 29–429).
Competing interests
Yoshinao Oda is an associate editor of Virchows Archiv. The other authors have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kawaguchi, K., Kohashi, K., Iwasaki, T. et al. Nuclear morphological atypia in biopsy accurately reflects the prognosis of myxoid liposarcoma. Virchows Arch (2024). https://doi.org/10.1007/s00428-024-03796-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00428-024-03796-7