Abstract
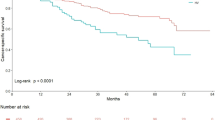
The World Health Organization/International Society of Urological Pathology (2022 WHO/ISUP) classification categorizes noninvasive carcinomas based on the highest grade observed in a pathology sample. According to this classification, a lesion is classified as mixed-grade (MG) if the highest-grade component comprises less than 5% high-grade (HG) carcinoma [14]. This study included 160 cases of low-grade papillary urothelial carcinoma (LGUC) and 160 cases of HG papillary urothelial carcinoma (HGUC), selected randomly. In addition, 160 consecutive and unselected cases of MG papillary urothelial carcinoma (MGUC) were obtained from all bladder transurethral resection specimens diagnosed with papillary urothelial carcinoma between January 2007 and January 2021. The results of the multivariate analysis showed that histologic grade, invasion of the lamina propria, and the presence of carcinoma in situ at presentation were independent prognostic parameters regarding recurrence-free survival (p = 0.002; hazard ratio (HR) = 1.44, 95% confidence interval (CI) = 1.059–1.956, p = 0.02; and HR = 1.76, 95% CI = 1.159–2.684, p = 0.008, respectively). Histologic grade was the only independent prognostic parameter of disease-specific survival (DSS) (p < 0.001). Comparisons between non-muscle invasive (NMI) MGUC and NMI LGUC, as well as between NMI MGUC and NMI HGUC, revealed statistically significant differences in terms of DSS (HR = 0.07, 95% CI = 0.024–0.252, p < 0.001 and HR = 1.59, 95% CI = 1.023–2.460, p = 0.039, respectively). Our study findings demonstrate statistically significant differences regarding DSS between NMI MGUC and NMI HGUC, as well as between NMI MGUC and NMI LGUC. Therefore, we suggested that considering the presence of less than 5% MGUC as a separate category may be appropriate. However, it is important to validate our results in larger cohorts with longer follow-up periods to establish the clinical significance of MGUC and provide guidance for patient management.



Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ajili F, Darouiche A, Chebil M, Boubaker S (2013) The efficiency of the EORTC scoring system for the prediction of recurrence and progression of non-muscle-invasive bladder cancer treated by bacillus Calmette-Guerin immunotherapy. Ultrastruct Pathol 37(4):249–253. https://doi.org/10.3109/01913123.2013.786772
Billis A, Carvalho RB, Mattos AC, Negretti F, Nogueira CR, Oliveira MC, ... & Dinamarco PV (2001) Tumor Grade Heterogeneity in Urothelial Bladder Carcinoma? Proposal of a System Using Combined Numbers. Scand J Urol Nephrol 35(4):275–279. https://doi.org/10.1080/003655901750425837
Chamber M, Wright JL, Haffner M, Reder N, Tretiakova M, ..., True L (2021) Prognosis of -Grade, Non-Muscle Invasive, Papillary Carcinoma of the Bladder: A Survival Analysis. Mod Pathol (Abstract) 34(2):543. https://www.nature.com/articles/s41379-021-00760-7. Accessed 12 Mar 2021
Cheng L, Neumann RM, Nehra A, Spotts BE, Weaver AL, Bostwick DG (2000) Cancer heterogeneity and its biologic implications in the grading of urothelial carcinoma. Cancer 88(7):1663–1670. https://doi.org/10.1002/(sici)1097-0142(20000401)88:7%3C1663::aid-cncr21%3E3.0.co;2-8
Council L, Hameed O (2009) Differential expression of immunohistochemical markers in bladder smooth muscle and myofibroblasts, and the potential utility of desmin, smoothelin, and vimentin in staging of bladder carcinoma. Mod Pathol 22(5):639–650. https://doi.org/10.1038/modpathol.2009.9
Culpan M, Keser F, Iplikci A, Kir G, Atis G, Yildirim A (2021) The Clinical Impact of Tumor Grade Heterogeneity in Non-muscle-invasive Urothelial Carcinoma of the Bladder. Medeni Med J 36(4):310–317. https://doi.org/10.4274/mmj.galenos.2021.48447
Eble JN, Sauter G (2004) Epstein JIet al. World Health Organization Classification of Tumours. Pathology and Genetics of Tumours of the Urinary System and Male Genital Organs. IARC Press, Lyon
Gofrit ON, Pizov G, Shapiro A, Duvdevani M, Yutkin V, Landau EH, ... & Pode D (2014) Mixed high and low grade bladder tumors—are they clinically high or low grade? J Urol 191(6):1693–1696. https://doi.org/10.1016/j.juro.2013.11.056
Hu Z, Mudaliar K, Quek ML, Paner GP, Barkan GA (2014) Measuring the dimension of invasive component in pT1 urothelial carcinoma in transurethral resection specimens can predict time to recurrence. Ann Diagn Pathol 18(2):49–52. https://doi.org/10.1016/j.anndiagpath.2013.11.002
Jones TD, Cheng L (2006) Papillary urothelial neoplasm of low malignant potential: evolving terminology and concepts. J Urol 175(6):1995–2003. https://doi.org/10.1016/s0022-5347(06)00267-9
Ma S, Han Y, Zhang D, Li Q, Wang E, Wu G, Zhao H (2021) Noninvasive papillary urothelial carcinoma with pathological features in between low and high grades: A case report. Medicine 100(17) https://doi.org/10.1097/md.0000000000025693
Miyamoto H, Miller JS, Fajardo DA, Lee TK, Netto GJ, Epstein JI (2010) Non-invasive papillary urothelial neoplasms: the 2004 WHO/ISUP classification system. Pathol Int 60(1):1–8. https://doi.org/10.1111/j.1440-1827.2009.02477.x
Moch H, Humphrey PA, Ulbright TM, Reuter VE (2016) WHO Classification of tumours of the urinary system and male genital organs, 4th ed.. International Agency for Research on Cancer, Lyon, France
WHO Classification of Tumours (2022) Urinary and male genital tumours, WHO Classification of Tumours Editorial Board, 5th edn. IARC press, Lyon
Mostofi FK, Sobin LH, Torloni H (1973) Histological Typing of Urinary Bladder Tumours. Geneva, Switzerland: World Health Organization. Int Hist Classification Tumours (10)
Neal DJ, Amin MB, Smith SC (2020) CK20 versus AMACR and p53 immunostains in evaluation of Urothelial Carcinoma in Situ and Reactive Atypia. Diagn Pathol 15(1):1–4
Nese N, Gupta R, Bui MH, Amin MB (2009) Carcinoma in situ of the urinary bladder: review of clinicopathologic characteristics with an emphasis on aspects related to molecular diagnostic techniques and prognosis. J Natl Compr Canc Netw 7(1):48–57. https://doi.org/10.6004/jnccn.2009.0004
Pan CC, Chang YH, Chen KK, Yu HJ, Sun CH, Ho DM (2010) Prognostic significance of the 2004 WHO/ISUP classification for prediction of recurrence, progression, and cancer-specific mortality of non–muscle-invasive urothelial tumors of the urinary bladder: a clinicopathologic study of 1,515 cases. Am J Clin Pathol 133(5):788–795. https://doi.org/10.1309/ajcp12mrvvhtckej
Quintero A, Alvarez-Kindelan J, Luque RJ, Gonzalez-Campora R, Requena MJ, Montironi R, Lopez-Beltran A (2006) Ki-67 MIB1 labelling index and the prognosis of primary Ta T1 urothelial cell carcinoma of the bladder. J Clin Pathol 59(1):83–88. https://doi.org/10.1136/jcp.2004.022939
Reis LO, Taheri D, Chaux A, Guner G, Rodriguez MAM, Bivalacqua TJ, ... & Netto GJ (2016) Significance of a minor high-grade component in a low-grade noninvasive papillary urothelial carcinoma of bladder. Hum Pathol 47(1):20–25. https://doi.org/10.1016/j.humpath.2015.09.007
Schubert T, Danzig MR, Kotamarti S, Ghandour RA, Lascano D, Dubow BP, ... & McKiernan JM (2015) Mixed low-and high-grade non-muscle-invasive bladder cancer: a histological subtype with favorable outcome. World J Urol 33:847–852. https://doi.org/10.1007/s00345-014-1383-5
Sylvester RJ, Van Der Meijden AP, Oosterlinck W, Witjes JA, Bouffioux C, Denis L, ... & Kurth K (2006) Predicting recurrence and progression in individual patients with stage Ta T1 bladder cancer using EORTC risk tables: a combined analysis of 2596 patients from seven EORTC trials. Eur Urol 49(3):466–477. https://doi.org/10.1016/j.eururo.2005.12.031
Author information
Authors and Affiliations
Contributions
Gozde Kir: Designed the research study, contributed essential reagents or tools, performed the research, reviewed and corrected manuscript
Gozde Ecem Cecikoglu: Analyzed the data, wrote the draft paper
Muhammet Huseyin Sorkun: Analyzed the data
Cumhur Selçuk Topal, Meftun Culpan, Ferhat Yakup Suceken, Hakan Karaca: Contributed essential reagents or tools
Asif Yıldırım: Reviewed and corrected manuscript
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kir, G., Cecikoglu, G.E., Topal, C.S. et al. Clinicopathologic features and prognostic significance of mixed (Low and high-grade) papillary urothelial carcinoma comparison with low and high-grade papillary urothelial carcinoma. Virchows Arch 483, 621–634 (2023). https://doi.org/10.1007/s00428-023-03625-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-023-03625-3