Abstract
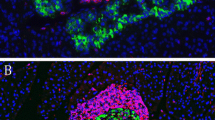
We studied the histo- and immunopathology of the endocrine and exocrine pancreas and a number of other organs in a new insulin-dependent diabetes mellitus (IDDM) rat model (LEW.1AR1/Ztm-iddm rat). The pancreas of the acutely diabetic animals showed an inflammatory infiltrate, involving all islets and ducts. The islet infiltrate was composed mainly of ED1-positive macrophages and T lymphocytes, comprising a large number of CD8+ lymphocytes and a few CD4+ lymphocytes. In addition, the islets displayed apoptotic cells, characterized by condensation and fragmentation of nuclear chromatin. These cells were identified as beta cells by insulin immunostaining. Other endocrine and exocrine glands, including adrenals and thyroid, as well as salivary and submandibular glands, were unaffected. Organs from the digestive tract or systemic circulatory system, including small intestine, liver, heart, and lung also showed no involvement. The kidney was intact in acutely diabetic rats. However, 6 months after diabetes manifestation, pathological changes compatible with a diabetic nephropathy had developed, affecting both the glomerula and the proximal tubular segments. It was concluded that the autoimmune process in this new IDDM rat model is restricted to the endocrine pancreas and leads to apoptotic beta cell destruction.





Similar content being viewed by others
References
Augstein P, Stephens LA, Allison J, Elefanty AG, Ekberg M, Kay TW, Harrison LC (1998) Beta-cell apoptosis in an accelerated model of autoimmune diabetes. Mol Med 4:495–501
Awata T, Guberski DL, Like AA (1995) Genetics of the BB rat: association of autoimmune disorders (diabetes, insulitis, and thyroiditis) with lymphopenia and major histocompatibility complex class II. Endocrinology 136:5731–5735
Bieg S, Lernmark A (1999) Animal models for insulin-dependent diabetes mellitus. In: Volpe R (ed) Contemporary endocrinology: autoimmune endocrinopathies. Humana Press, Totowa, pp 113–139
Christianson SW, Shultz LD, Leiter EH (1993) Adoptive transfer of diabetes into immunodeficient NOD-scid/scid mice. Relative contributions of CD4+ and CD8+ T-cells from diabetic versus prediabetic NOD.NON-Thy-1a donors. Diabetes 42:44–55
Cohen AJ, McGill PD, Rossetti RG, Guberski DL, Like AA (1987) Glomerulopathy in spontaneously diabetic rat. Impact of glycemic control. Diabetes 36:944–951
Eisenbarth GS (2003) Type I diabetes. Molecular, cellular and clinical immunology. Oxford University Press, New York, Oxford
Elsner M, Tiedge M, Guldbakke B, Munday R, Lenzen S (2002) Importance of the GLUT2 glucose transporter for pancreatic beta cell toxicity of alloxan. Diabetologia 45:1542–1549
Hadano H, Suzuki S, Tanigawa K, Ago A (1988) Cell infiltration in various organ and dilatation of the urinary tubule in NON mice. Jikken Dobutsu 37:479–483
Homo-Delarche F (1997) Beta-cell behaviour during the prediabetic stage. Part II. Non-insulin- dependent and insulin-dependent diabetes mellitus. Diabetes Metab 23:473–505
Hsu SM, Raine L, Fanger H (1981) Use of avidin-biotin-peroxidase complex (ABC) in immunoperoxidase techniques: a comparison between ABC and unlabeled antibody (PAP) procedures. J Histochem Cytochem 29:577–580
Imagawa A, Hanafusa T, Itoh N, Miyagawa J, Nakajima H, Namba M, Kuwajima M, Tamura S, Kawata S, Matsuzawa Y, Harlan DM (1996) Islet-infiltrating t lymphocytes in insulin-dependent diabetic patients express CD80 (B7-1) and CD86 (B7-2). J Autoimmun 9:391–396
Jansen A, Homo-Delarche F, Hooijkaas H, Leenen PJ, Dardenne M, Drexhage HA (1994) Immunohistochemical characterization of monocytes-macrophages and dendritic cells involved in the initiation of the insulitis and beta-cell destruction in NOD mice. Diabetes 43:667–675
Jörns A, Tiedge M, Lenzen S (1999) Nutrient-dependent distribution of insulin and glucokinase immunoreactivities in rat pancreatic β cells. Virchows Arch 434:75–82
Jörns A, Tiedge M, Ziv E, Shafrir E, Lenzen S (2002) Gradual loss of pancreatic beta-cell insulin, glucokinase and GLUT2 glucose transporter immunoreactivities during the time course of nutritionally induced type-2 diabetes in Psammomys obesus (sand rat). Virchows Arch 440:63–69
Jörns A, Tiedge M, Lenzen S (2002) Thyroxine induces pancreatic beta cell apoptosis. Diabetologia 45:851–855
Klöppel G, Clemens A (1996) Insulin-dependent diabetes mellitus. Current aspects of morphology, etiology and pathogenesis. Pathologe 17:269–275
Kurrer MO, Pakala SV, Hanson HL, Katz JD (1997) Beta cell apoptosis in T cell-mediated autoimmune diabetes. Proc Natl Acad Sci U S A 94:213–218
Lally FJ, Bone AJ (2002) Animal models of type 1 diabetes. In: Pickup JC, Williams G (eds) Textbook of diabetes, Blackwell Scientific Publications, Oxford, pp 19.1–19.17
Lenzen S, Tiedge M, Elsner M, Lortz S, Weiss H, Jörns A, Klöppel G, Wedekind D, Prokop CM, Hedrich HJ (2001) The LEW.1AR1/Ztm-iddm rat: a new model of spontaneous insulin-dependent diabetes mellitus. Diabetologia 44:1189–1196
Nagata M, Santamaria P, Kawamura T, Utsugi T, Yoon JW (1994) Evidence for the role of CD8+ cytotoxic T cells in the destruction of pancreatic beta-cells in nonobese diabetic mice. J Immunol 152:2042–2050
Sgonc R, Wick G (1994) Methods for the detection of apoptosis. Int Arch Allergy Immunol 105:327–332
Voorbij HA, Jeucken PH, Kabel PJ, De Haan M, Drexhage HA (1989) Dendritic cells and scavenger macrophages in pancreatic islets of prediabetic BB rats. Diabetes 38:1623–1629
Wang RN, Bouwens L, Klöppel G (1996) Beta-cell growth in adolescent and adult rats treated with streptozotocin during the neonatal period. Diabetologia 39:548–557
Acknowledgements
This work has been supported by a grant from the Deutsche Forschungsgemeinschaft (Jo-395 1–1) to A.J. and by the NIH grant 1R21AI55464–01 to S.L. and H.J.H. The technical assistance of M. Böger and U. Sommerfeld is gratefully acknowledged.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jörns, A., Kubat, B., Tiedge, M. et al. Pathology of the pancreas and other organs in the diabetic LEW.1AR1/Ztm-iddm rat, a new model of spontaneous insulin-dependent diabetes mellitus. Virchows Arch 444, 183–189 (2004). https://doi.org/10.1007/s00428-003-0956-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-003-0956-2