Abstract
Main conclusion
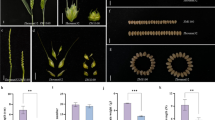
A stable genetic transformation system for Erigeron breviscapus was developed. We cloned the EbYUC2 gene and genetically transformed it into Arabidopsis thaliana and E. breviscapus. The leaf number, YUC2 gene expression, and the endogenous auxin content in transgenic plants were significantly increased.
Abstract
Erigeron breviscapus is a prescription drug for the clinical treatment of cardiovascular and cerebrovascular diseases. The rosette leaves have the highest content of the major active compound scutellarin and are an important component in the yield of E. breviscapus. However, little is known about the genes related to the leaf number and flowering time of E. breviscapus. In our previous study, we identified three candidate genes related to the leaf number and flowering of E. breviscapus by combining resequencing data and genome-wide association study (GWAS). However, their specific functions remain to be characterized. In this study, we cloned and transformed the previously identified full-length EbYUC2 gene into Arabidopsis thaliana, developed the first stable genetic transformation system for E. breviscapus, and obtained the transgenic plants overexpressing EbYUC2. Compared with wild-type plants, the transgenic plants showed a significant increase in the number of leaves, which was correlated with the increased expression of EbYUC2. Consistently, the endogenous auxin content, particularly indole-3-acetic acid, in transgenic plants was also significantly increased. These results suggest that EbYUC2 may control the leaf number by regulating auxin biosynthesis, thereby laying a foundation for revealing the molecular mechanism governing the leaf number and flowering time of E. breviscapus.






Similar content being viewed by others
Data availability
The sequence of EbYUC2 was derived from the E. breviscapus genome deposited at our database (http://medicinalplants.ynau.edu.cn/).
Abbreviations
- IPA:
-
Indole-3-propionic acid
- IAA:
-
Indole-3-acetic acid
- TAA:
-
Tryptophan aminotransferase
- YUC2:
-
Indole-3-pyruvate monooxygenase YUCCA2
References
Akiyama K, Hayashi H (2006) Strigolactones: chemical signals for fungal symbionts and parasitic weeds in plant roots. Ann Bot 97:925–931. https://doi.org/10.1093/aob/mcl063
Alam P, Mohammad A, Ahmad MM, Khan MA, Nadeem M, Khan R, Akmal M, Ahlawat S, Abdin MZ (2014) Efficient method for Agrobacterium mediated transformation of Artemisia annua L. Recent Pat Biotechnol 8:102–107. https://doi.org/10.2174/18722083113079990001
Arney SE (1968) The effect of leaf primordia and auxin on leaf initiation rate in strawberry. Planta 82(3):235–245. https:// doi.org/ https://doi.org/10.1007/BF00398202
Asakura Y, Seki H, Muranaka T, Yamamura Y, Kurosaki F (2008) Enhanced secretory activity of Atropa belladonna hairy root culture over-expressing ADP-ribosylation factor gene. Biol Pharm Bull 31:1465–1468. https://doi.org/10.1248/bpb.31.1465
Cao X, Yang H, Shang C, Ma S, Liu L, Cheng J (2019) The roles of auxin biosynthesis YUCCA gene family in plants. Int J Mol Sci 20(24):6343. https://doi.org/10.3390/ijms20246343
Chen J, Tomes S, Gleave AP, Hall W, Luo Z, Xu J, Yao JL (2022) Significant improvement of apple (Malus domestica Borkh.) transgenic plant production by pre-transformation with a Baby boom transcription factor. Hortic Res 9: uhab014. https://doi.org/10.1093/hr/uhab014
Cheng Y, Dai X, Zhao Y (2006) Auxin biosynthesis by the YUCCA flavin monooxygenases controls the formation of floral organs and vascular tissues in Arabidopsis. Gene Dev 20:1790–1799. https://doi.org/10.1101/gad.1415106
Cheng Y, Dai X, Zhao Y (2007) Auxin synthesized by the YUCCA flavin monooxygenases is essential for embryogenesis and leaf formation in Arabidopsis. Plant Cell 19:2430–2439. https://doi.org/10.1105/tpc.107.053009
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743. https://doi.org/10.1046/j.1365-313x.1998.00343.x
Debernardi JM, Tricoli DM, Ercoli MF, Hayta S, Ronald P, Palatnik JF, Dubcovsky J (2020) A GRF-GIF chimeric protein improves the regeneration efficiency of transgenic plants. Nat Biotechnol 38:1274–1279. https://doi.org/10.1038/s41587-020-0703-0
Di D, Wu L, Zhang L, An C, Zhang T, Luo P, Gao H, Kriechbaumer V, Guo G (2016) Functional roles of Arabidopsis CKRC2/YUCCA8 gene and the involvement of PIF4 in the regulation of auxin biosynthesis by cytokinin. Sci Rep 6:36866. https://doi.org/10.1038/srep36866
Dong L, Ma Y, Chen C, Shen L, Sun W, Cui G, Naqvi NI, Deng YZ (2022) Identification and characterization of auxin/IAA biosynthesis pathway in the rice blast fungus Magnaporthe oryzae. J Fungi 8:208. https://doi.org/10.3390/jof8020208
Firsov A, Mitiouchkina T, Shaloiko L, Pushin A, Vainstein A, Dolgov S (2020) Agrobacterium-mediated transformation of Chrysanthemum with artemisinin biosynthesis pathway genes. Plants-Basel 9(4):537. https://doi.org/10.3390/plants9040537
Gomez-Roldan V, Fermas S, Brewer PB, Puech-Pages V, Dun EA, Pillot JP, Letisse F, Matusova R, Danoun S, Portais JC, Bouwmeester H, Becard G, Beveridge CA, Rameau C, Rochange SF (2008) Strigolactone inhibition of shoot branching. Nature 455: 189–194. https://doi.org/10.1038/nature07271
Gawel NJ, Jarret RL (1991) A modified CTAB DNA extraction procedure for Musa and Ipomoea. Plant Mol Biol Rep 9: 262–266. https://doi.org/10.1007/BF02672076
Hadfi K, Speth V, Neuhaus G (1998) Auxin-induced developmental patterns in Brassica juncea embryos. Development 125(5):879–887 https://doi.org/10.1242/dev.125.5.879
He S, Dong X, Zhang G, Fan W, Duan S, Shi H, Li D, Li R, Chen G, Long G, Zhao Y, Chen M, Yan M, Yang J, Lu Y, Zhou Y, Chen W, Dong Y, Yang S (2021) High quality genome of Erigeron breviscapus provides a reference for herbal plants in Asteraceae. Mol Ecol Resour 21:153–169. https://doi.org/10.1111/1755-0998.13257
Hou X, Liu S, Pierri F, Dai X, Qu L, Zhao Y (2011) Allelic analyses of the Arabidopsis YUC1 locus reveal residues and domains essential for the functions of YUC family of flavin monooxygenases. J Integr Plant Biol 53:54–62. https://doi.org/10.1111/j.1744-7909.2010.01007.x
Kalve S, De Vos D, Beemster GT (2014) Leaf development: a cellular perspective. Front Plant Sci 5:362. https://doi.org/10.3389/fpls.2014.00362
Liu X, Cheng J, Zhang G, Ding W, Duan L, Yang J, Kui L, Cheng X, Ruan J, Fan W, Chen J, Long G, Zhao Y, Cai J, Wang W, Ma Y, Dong Y, Yang S, Jiang H (2018) Engineering yeast for the production of breviscapine by genomic analysis and synthetic biology approaches. Nat Commun 9:448. https://doi.org/10.1038/s41467-018-02883-z
Liu Y, Yang N, Yuan H, Chen P, Gu R, Zhang Y (2023) BraVRG, a novel protein of Brassica rapa, is induced by vernalization and promotes flowering in Arabidopsis thaliana. Plant Sci 327:111544. https://doi.org/10.1016/j.plantsci.2022.111544
Ljung K, Sveriges L (2013) Auxin metabolism and homeostasis during plant development. Development (cambridge) 140:943–950. https://doi.org/10.1242/dev.086363
Ma R, Yu Z, Cai Q, Li H, Dong Y, Oksman-Caldentey KM, Rischer H (2020) Agrobacterium-mediated genetic transformation of the medicinal plant Veratrum dahuricum. Plants-Basel 9(2):191. https://doi.org/10.3390/plants9020191
Mashiguchi K, Tanaka K, Sakai T, Sugawara S, Kawaide H, Natsume M, Hanada A, Yaeno T, Shirasu K, Yao H, McSteen P, Zhao Y, Hayashi K, Kamiya Y, Kasahara H (2011) The main auxin biosynthesis pathway in Arabidopsis. Proc Natl Acad Sci USA 108:18512–18517. https://doi.org/10.1073/pnas.1108434108
Matusova R, Rani K, Verstappen FW, Franssen MC, Beale MH, Bouwmeester HJ (2005) The strigolactone germination stimulants of the plant-parasitic Striga and Orobanche spp. are derived from the carotenoid pathway. Plant Physiol 139:920–934. https://doi.org/10.1104/pp.105.061382
McSteen P, Laudencia-Chingcuanco D, Colasanti J (2000) A floret by any other name: control of meristem identity in maize. Trends Plant Sci 5:61–66. https://doi.org/10.1016/s1360-1385(99)01541-1
Mookkan M, Nelson-Vasilchik K, Hague J, Kausch A, Zhang ZJ (2018) Morphogenic regulator-mediated transformation of maize inbred B73. Curr Protoc Plant Biol 3:e20075. https://doi.org/10.1002/cppb.20075
Pan J, Zhao F, Zhang G, Pan Y, Sun L, Bao N, Qin P, Chen L, Yu J, Zhang Y, Xu L (2019) Control of de novo root regeneration efficiency by developmental status of Arabidopsis leaf explants. J Genet Genomics 46:133–140. https://doi.org/10.1016/j.jgg.2019.03.001
Rajasekaran D, Groning S, Schmitz C, Zierow S, Drucker N, Bakou M, Kohl K, Mertens A, Lue H, Weber C, Xiao A, Luker G, Kapurniotu A, Lolis E, Bernhagen J (2016) Macrophage migration inhibitory factor-CXCR4 receptor interactions: EVIDENCE FOR PARTIAL ALLOSTERIC AGONISM IN COMPARISON WITH CXCL12 CHEMOKINE. J Biol Chem 291:15881–15895. https://doi.org/10.1074/jbc.M116.717751
Reinhardt D, Mandel T, Kuhlemeier C (2000) Auxin regulates the initiation and radial position of plant lateral organs. Plant Cell 12: 507–518. https://doi.org/10.1105/tpc.12.4.507
Song WL, Lu YC, Yang SC (2018) Correlation analysis between agronomic traits and economic characters of Erigeron breviscapus. Mol Plant Breed 7148–7158. https://doi.org/10.13271/j.mpb.016.007148
Umehara M, Hanada A, Yoshida S, Akiyama K, Arite T, Takeda-Kamiya N, Magome H, Kamiya Y, Shirasu K, Yoneyama K, Kyozuka J, Yamaguchi S (2008) Inhibition of shoot branching by new terpenoid plant hormones. Nature 455:195–200. https://doi.org/10.1038/nature07272
Won C, Shen X, Mashiguchi K, Zheng Z, Dai X, Cheng Y, Kasahara H, Kamiya Y, Chory J, Zhao Y (2011) Conversion of tryptophan to indole-3-acetic acid by TRYPTOPHAN AMINOTRANSFERASES OF ARABIDOPSIS and YUCCAs in Arabidopsis. Proc Natl Acad Sci USA 108:18518–18523. https://doi.org/10.1073/pnas.1108436108
Xiong Y, Jiao Y (2019) The diverse roles of auxin in regulating leaf development. Plants-Basel 8(7):243. https://doi.org/10.3390/plants8070243
Xu X, E Z, Zhang D, Yun Q, Zhou Y, Niu B, Chen C (2021) OsYUC11-mediated auxin biosynthesis is essential for endosperm development of rice. Plant Physiol 185: 934-950. https://doi.org/10.1093/plphys/kiaa057
Yan Y, Wang Z (2007) Genetic transformation of the medicinal plant Salvia miltiorrhiza by Agrobacterium tumefaciens-mediated method. Plant Cell Tissue Organ Cult 88:175–184. https://doi.org/10.1007/s11240-006-9187-y
Yang G, Liang K, Zhou Z, Wang X, Huang G (2020) UPLC-ESI-MS/MS-based widely targeted metabolomics analysis of wood metabolites in teak (Tectona grandis). Molecule 25(9):2189. https://doi.org/10.3390/molecules25092189
Zhang W, Wei X, Meng HL, Ma CH, Jiang NH, Zhang GH, Yang SC (2015) Transcriptomic comparison of the self-pollinated and cross-pollinated flowers of Erigeron breviscapus to analyze candidate self-incompatibility-associated genes. BMC Plant Biol 15: 248. https://doi.org/10.1186/s12870-015-0627-x
Zhang Z, Runions A, Mentink RA, Kierzkowski D, Karady M, Hashemi B, Huijser P, Strauss S, Gan X, Ljung K, Tsiantis M (2020) A WOX/auxin biosynthesis module controls growth to shape leaf form. Curr Biol 30:4857–4868. https://doi.org/10.1016/j.cub.2020.09.037
Zhao Y (2010) Auxin biosynthesis and its role in plant development. Annu Rev Plant Biol 61:49–64. https://doi.org/10.1146/annurev-arplant-042809-112308
Zhao Y (2012) Auxin biosynthesis: a simple two-step pathway converts tryptophan to indole-3-acetic acid in plants. Mol Plant 5:334–338. https://doi.org/10.1093/mp/ssr104
Zhao Y (2018) Essential roles of local auxin biosynthesis in plant development and in adaptation to environmental changes. Annu Rev Plant Biol 69: 417–435. https://doi.org/10.1146/annurev-arplant-042817-040226
Zhao Y, Yu Y, Guo J, Zhang Y, Huang L (2023) Rapid and efficient optimization method for a genetic transformation system of medicinal plants Erigeron breviscapus. Int J Mol Sci 24(6):5611. https://doi.org/10.3390/ijms24065611
Zheng L, Zhang L, Duan K, Zhu ZP, Ye ZW, Gao QH (2016) YUCCA type auxin biosynthesis genes encoding flavin monooxygenases in melon: Genome-wide identification and developmental expression analysis. S Afr J Bot 102:142–152. https://doi.org/10.1016/j.sajb.2015.06.012
Acknowledgements
This work was supported by the National Natural Science Foundation of China (81960689).
Author information
Authors and Affiliations
Contributions
HSM, SBC, and YSC designed the experiment. ZQ, XJL, and YYH performed the experiments. LYC and ZGH analyzed the data. ZQ, HSM, and YJL prepared the manuscript.
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhu, Q., Lu, YC., Xiong, JL. et al. Development of a stable genetic transformation system in Erigeron breviscapus: a case study with EbYUC2 in relation to leaf number and flowering time. Planta 259, 98 (2024). https://doi.org/10.1007/s00425-024-04351-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-024-04351-z