Abstract
Main conclusion
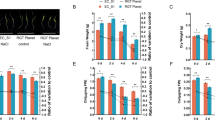
Overexpression and loss of function of OsGEX3 reduce seed setting rates and affect pollen fertility in rice. OsGEX3 positively regulates osmotic stress response by regulating ROS scavenging.
Abstract
GEX3 proteins are conserved in plants. AtGEX3 encodes a plasma membrane protein that plays a crucial role in pollen tube guidance. However, the function of its homolog in rice, OsGEX3, has not been determined. Our results demonstrate that OsGEX3 is localized in the plasma membrane and the nucleus as shown by a transiently transformed assay using Nicotiana benthamiana leaves. The up-regulation of OsGEX3 was detected in response to treatments with polyethylene glycol (PEG) 4000, hydrogen peroxide, and abscisic acid (ABA) via RT-qPCR analysis. Interestingly, we observed a significant decline in the seed setting rates of OsGEX3-OE lines and mutants, compared to the wild type. Further investigations reveal that overexpression and loss of function of OsGEX3 affect pollen maturation. TEM observation revealed a significant decrease in the fertile pollen rates of OsGEX3-OE transgenic lines and Osgex3 mutants due to a delay in pollen development at the late vacuolated stage. Overexpression of OsGEX3 improved osmotic stress and oxidative stress tolerance by enhancing reactive oxygen species (ROS) scavenging in rice seedlings, whereas Osgex3 mutants exhibited an opposite phenotype in osmotic stress. These findings highlight the multifunctional roles of OsGEX3 in pollen development and the response to abiotic stress. The functional characterization of OsGEX3 provides a fundamental basis for rice molecular breeding and can facilitate efforts to cultivate drought resistance and yield-related varieties.










Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the paper and within its supplementary materials published online.
Abbreviations
- GEX3:
-
Gamete-expressed 3 proteins
- GUS:
-
β-Glucuronidase
- MDA:
-
Malondialdehyde
- OsGEX3:
-
Oryza sativa Gamete-expressed 3 protein
- PCD:
-
Programmed cell death
- ROS:
-
Reactive oxygen species
- SOD:
-
Superoxide dismutase
- TEM:
-
Transmission electron microscopy
References
Alandete-Saez M, Ron M, McCormick S (2008) GEX3, expressed in the male gametophyte and in the egg cell of Arabidopsis thaliana, is essential for micropylar pollen tube guidance and plays a role during early embryogenesis. Mol Plant 1(4):586–598. https://doi.org/10.1093/mp/ssn015
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, andsignal transduction. Annu Rev Plant Biol 55:373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Campo S, Baldrich P, Messeguer J, Lalanne E, Coca M, San Segundo B (2014) Overexpression of a calcium-dependent protein kinase confers salt and drought tolerance in rice by preventing membrane lipid peroxidation. Plant Physiol 165(2):688–704. https://doi.org/10.1104/pp.113.230268
Chen S, Songkumarn P, Liu J, Wang GL (2009) A versatile zero background T-vector system for gene cloning and functional genomics. Plant Physiol 150(3):1111–1121. https://doi.org/10.1104/pp.109.137125
Du H, Wang N, Cui F, Li X, Xiao J, Xiong L (2010) Characterization of the beta-carotene hydroxylase gene DSM2 conferring drought and oxidative stress resistance by increasing xanthophylls and abscisic acid synthesis in rice. Plant Physiol 154(3):1304–1318. https://doi.org/10.1104/pp.110.163741
Han MJ, Jung KH, Yi G, Lee DY, An G (2006) Rice immature pollen 1 (RIP1) is a regulator of late pollen development. Plant Cell Physiol 47(11):1457–1472. https://doi.org/10.1093/pcp/pcl013
Hu Q, Tang D, Wang H, Shen Y, Chen X, Ji J et al (2016) The exonuclease homolog OsRAD1 promotes accurate meiotic double-strand break repair by suppressing nonhomologous end joining. Plant Physiol 172(2):1105–1116. https://doi.org/10.1104/pp.16.00831
Jefferson RA (1987) Assaying chimeric genes in plants: the GUS gene fusion system. Plant Mol Biol Rep 5(4):387–405. https://doi.org/10.1007/BF02667740
Joo J, Lee YH, Kim YK, Nahm BH, Song SI (2013) Abiotic stress responsive rice ASR1 and ASR3 exhibit different tissue-dependent sugar and hormone-sensitivities. Mol Cells 35(5):421–435. https://doi.org/10.1007/s10059-013-0036-7
Kidwai M, Dhar YV, Gautam N, Tiwari M, Ahmad IZ, Asif MH et al (2019) Oryza sativa class III peroxidase (OsPRX38) overexpression in Arabidopsis thaliana reduces arsenic accumulation due to apoplastic lignification. J Hazard Mater 362:383–393. https://doi.org/10.1016/j.jhazmat.2018.09.029
Li L, Li Y, Song S, Deng H, Li N, Fu X et al (2015) An anther development F-box (ADF) protein regulated by tapetum degeneration retardation (TDR) controls rice anther development. Planta 241(1):157–166. https://doi.org/10.1007/s00425-014-2160-9
Li Y, Han S, Sun X, Khan NU, Zhong Q, Zhang Z et al (2023) Variations in OsSPL10 confer drought tolerance by directly regulating OsNAC2 expression and ROS production in rice. J Integr Plant Biol 65(4):918–933. https://doi.org/10.1111/jipb.13414
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Mahender A, Anandan A, Pradhan SK, Pandit E (2016) Rice grain nutritional traits and their enhancement using relevant genes and QTLs through advanced approaches. Springerplus 5(1):2086. https://doi.org/10.1186/s40064-016-3744-6
Mathew IE, Priyadarshini R, Mahto A, Jaiswal P, Parida SK, Agarwal P (2020) SUPER STARCHY1/ONAC025 participates in rice grain filling. Plant Direct 4(9):e00249. https://doi.org/10.1002/pld3.249
Mhamdi A, Queval G, Chaouch S, Vanderauwera S, Van Breusegem F, Noctor G (2010) Catalase function in plants: a focus on Arabidopsis mutants as stress-mimic models. J Exp Bot 61(15):4197–4220. https://doi.org/10.1093/jxb/erq282
Mittler R (2017) ROS are good. Trends Plant Sci 22(1):11–19. https://doi.org/10.1016/j.tplants.2016.08.002
Nielsen ME, Thordal-Christensen H (2012) Recycling of Arabidopsis plasma membrane PEN1 syntaxin. Plant Signal Behav 7(12):1541–1543. https://doi.org/10.4161/psb.22304
Pandey S, Fartyal D, Agarwal A, Shukla T, James D, Kaul T et al (2017) Abiotic stress tolerance in plants: myriad roles of ascorbate peroxidase. Front Plant Sci 8:581. https://doi.org/10.3389/fpls.2017.00581
Qin F, Shinozaki K, Yamaguchi-Shinozaki K (2011) Achievements and challenges in understanding plant abiotic stress responses and tolerance. Plant Cell Physiol 52(9):1569–1582. https://doi.org/10.1093/pcp/pcr106
Russell SD, Jones DS (2015) The male germline of angiosperms: repertoire of an inconspicuous but important cell lineage. Front Plant Sci 6:173. https://doi.org/10.3389/fpls.2015.00173
Russell SD, Gou X, Wong CE, Wang X, Yuan T, Wei X et al (2012) Genomic profiling of rice sperm cell transcripts reveals conserved and distinct elements in the flowering plant male germ lineage. New Phytol 195(3):560–573. https://doi.org/10.1111/j.1469-8137.2012.04199.x
Wang C, Wang Y, Cheng Z, Zhao Z, Chen J, Sheng P et al (2016) The role of OsMSH4 in male and female gamete development in rice meiosis. J Exp Bot 67(5):1447–1459. https://doi.org/10.1093/jxb/erv540
Wang F, Han T, Song Q, Ye W, Song X, Chu J et al (2020) The rice circadian clock regulates tiller growth and panicle development through strigolactone signaling and sugar sensing. Plant Cell 32(10):3124–3138. https://doi.org/10.1105/tpc.20.00289
Wen K, Chen Y, Zhou X, Chang S, Feng H, Zhang J et al (2019) OsCPK21 is required for pollen late-stage development in rice. J Plant Physiol 240:153000. https://doi.org/10.1016/j.jplph.2019.153000
Wu Y, Wang Y, Mi XF, Shan JX, Li XM, Xu JL et al (2016) The QTL GNP1 encodes GA20ox1, which increases grain number and yield by increasing cytokinin activity in rice panicle meristems. PLoS Genet 12(10):e1006386. https://doi.org/10.1371/journal.pgen.1006386
Xiao J, Cheng H, Li X, Xiao J, Xu C, Wang S (2013) Rice WRKY13 regulates cross talk between abiotic and biotic stress signaling pathways by selective binding to different cis-elements. Plant Physiol 163(4):1868–1882. https://doi.org/10.1104/pp.113.226019
Xiong H, Li J, Liu P, Duan J, Zhao Y, Guo X et al (2014) Overexpression of OsMYB48-1, a novel MYB-related transcription factor, enhances drought and salinity tolerance in rice. PLoS ONE 9(3):e92913. https://doi.org/10.1371/journal.pone.0092913
Xiong H, Yu J, Miao J, Li J, Zhang H, Wang X et al (2018) Natural variation in OsLG3 increases drought tolerance in rice by inducing ROS scavenging. Plant Physiol 178(1):451–467. https://doi.org/10.1104/pp.17.01492
Yang S, Xu K, Chen S, Li T, Xia H, Chen L et al (2019) A stress-responsive bZIP transcription factor OsbZIP62 improves drought and oxidative tolerance in rice. BMC Plant Biol 19(1):260. https://doi.org/10.1186/s12870-019-1872-1
Zanella M, Borghi GL, Pirone C, Thalmann M, Pazmino D, Costa A et al (2016) β-amylase 1 (BAM1) degrades transitory starch to sustain proline biosynthesis during drought stress. J Exp Bot 67(6):1819–1826. https://doi.org/10.1093/jxb/erv572
Zhang Z, Hu M, Xu W, Wang Y, Huang K, Zhang C et al (2021) Understanding the molecular mechanism of anther development under abiotic stresses. Plant Mol Biol 105(1–2):1–10. https://doi.org/10.1007/s11103-020-01074-z
Acknowledgements
We acknowledge grants from the Joint Fund of the National Natural Science Foundation of China and the Karst Science Research Center of Guizhou Province (U1812401), and the Major Science and Technology Special Project of Fujian Province (2020NZ08016). Han Bao thank Yanbing Li, Dr. Hai Zhang, and Dr. Yong Cui and for suggestions and technical supports of subcellular localization.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Dorothea Bartels.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bao, H., Cui, Y., Ge, L. et al. OsGEX3 affects anther development and improves osmotic stress tolerance in rice. Planta 259, 68 (2024). https://doi.org/10.1007/s00425-024-04342-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-024-04342-0