Abstract
Main conclusion
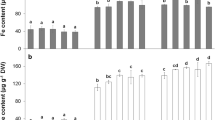
Fe uptake machinery of chloroplasts prefers to utilise Fe(III)–citrate over Fe–nicotianamine complexes.
Iron uptake in chloroplasts is a process of prime importance. Although a few members of their iron transport machinery were identified, the substrate preference of the system is still unknown. Intact chloroplasts of oilseed rape (Brassica napus) were purified and subjected to iron uptake studies using natural and artificial iron complexes. Fe–nicotianamine (NA) complexes were characterised by 5 K, 5 T Mössbauer spectrometry. Expression of components of the chloroplast Fe uptake machinery was also studied. Fe(III)–NA contained a minor paramagnetic Fe(II) component (ca. 9%), a paramagnetic Fe(III) component exhibiting dimeric or oligomeric structure (ca. 20%), and a Fe(III) complex, likely being a monomeric structure, which undergoes slow electronic relaxation at 5 K (ca. 61%). Fe(II)–NA contained more than one similar chemical Fe(II) environment with no sign of Fe(III) components. Chloroplasts preferred Fe(III)–citrate compared to Fe(III)–NA and Fe(II)–NA, but also to Fe(III)–EDTA and Fe(III)–o,o′EDDHA, and the Km value was lower for Fe(III)-citrate than for the Fe–NA complexes. Only the uptake of Fe(III)–citrate was light-dependent. Regarding the components of the chloroplast Fe uptake system, only genes of the reduction-based Fe uptake system showed high expression. Chloroplasts more effectively utilize Fe(III)–citrate, but hardly Fe–NA complexes in Fe uptake.







Similar content being viewed by others
Abbreviations
- FRO:
-
Ferric-chelate reductase/oxidase
- MAR1:
-
Multiple antibiotic resistance 1
- NA:
-
Nicotianamine
- NiCo:
-
Nickel–cobalt transporter
- o,o′EDDHA:
-
Ethylenediamine-N,N′-bis-o-hydroxyphenylacetate
- PIC1:
-
Permease in chloroplast 1
- YSL:
-
Yellow-stripe 1-like
References
Abadía J, Vázquez S, Rellán-Álvarez R, El-Jendoubi H, Abadía A, Álvarez-Fernández A, Flor López-Millán A (2011) Towards a knowledge-based correction of iron chlorosis. Plant Physiol Biochem 4:471–482. https://doi.org/10.1016/j.plaphy.2011.01.026
Álvarez-Fernández A, Díaz-Benito P, Abadía A, López-Millán AF, Abadía J (2014) Metal species involved in long distance metal transport in plants. Front Plant Sci 5:105. https://doi.org/10.3389/fpls.2014.00105
Ariga T, Hazama K, Yanagisawa S, Yoneyama T (2014) Chemical forms of iron in xylem sap from graminaceous and non-graminaceous plants. Soil Sci Plant Nutr 60:460–469. https://doi.org/10.1080/00380768.2014.922406
Balk J, Schaedler TA (2014) Iron cofactor assembly in plants. Annu Rev Plant Biol 65:125–153. https://doi.org/10.1146/annurev-arplant-050213-035759
Bashir K, Rasheed S, Kobayashi T, Seki M, Nishizawa NK (2016) Regulating subcellular metal homeostasis: the key to crop improvement. Front Plant Sci 7:1192. https://doi.org/10.3389/fpls.2016.01192
Becker R, Fritz E, Manteuffel R (1995) Subcellular localization and characterization of excessive iron in the nicotianamine-less tomato mutant chloronerva. Plant Physiol 108:269–275. https://doi.org/10.1104/pp.108.1.269
Bonneau J, Baumann U, Beasley J, Li Y, Johnson AAT (2016) Identification and molecular characterization of the nicotianamine synthase gene family in bread wheat. Plant Biotechnol J 14:2228–2239. https://doi.org/10.1111/pbi.12577
Briat JF, Curie C, Gaymard F (2007) Iron utilization and metabolism in plants. Curr Opin Plant Biol 10:276–282. https://doi.org/10.1016/j.pbi.2007.04.003
Briat JF, Ravet K, Arnaud N, Duc C, Boucherez J, Touraine B, Cellier F, Gaymard F (2010) New insights into ferritin synthesis and function highlight a link between iron homeostasis and oxidative stress in plants. Ann Bot 105:811–822. https://doi.org/10.1093/aob/mcp128
Briat JF, Dubos C, Gaymard F (2015) Iron nutrition, biomass production, and plant product quality. Trends Plant Sci 20:33–40. https://doi.org/10.1016/j.tplants.2014.07.005
Brüggemann W, Maas-Kantel K, Moog PR (1993) Iron uptake by leaf mesophyll-cells: the role of the plasma membrane-bound ferric-chelate reductase. Planta 190:151–155. https://doi.org/10.1007/BF00196606
Bughio N, Takahashi M, Yoshimura E, Nishizawa NK, Mori S (1997) Light-dependent iron transport into isolated barley chloroplasts. Plant Cell Physiol 38:101–105. https://doi.org/10.1093/oxfordjournals.pcp.a029079
Burda K, Kruk J, Borgstadt R, Stanek J, Strzalka K, Schmid GH, Kruse O (2003) Mössbauer studies of the non-heme iron and cytochrome b 559 in a Chlamydomonas reinhardtii PSI-mutant and their interactions with α-tocopherol quinone. FEBS Lett 535:159–165. https://doi.org/10.1016/S0014-5793(02)03895-4
Conte S (2009) The MAR1 transporter of Arabidopsis thaliana has roles in aminoglycoside antibiotic transport and iron homeostasis. Dissertation, The University of Texas at Austin
Conte S, Lloyd A (2010) The MAR1 transporter is an opportunistic entry point for antibiotics. Plant Signal Behav 5:49–52. https://doi.org/10.4161/psb.5.1.10142
Conte S, Stevenson D, Furner I, Lloyd A (2009) Multiple antibiotic resistance in Arabidopsis is conferred by mutations in a chloroplast-localized transport protein. Plant Physiol 151:559–573. https://doi.org/10.1104/pp.109.143487
Curie C, Cassin G, Couch D, Divol F, Higuchi K, le Jean M, Misson J, Schikora A, Czernic P, Mari S (2009) Metal movement within the plant: contribution of nicotianamine and yellow stripe 1-like transporters. Ann Bot 103:1–11. https://doi.org/10.1093/aob/mcn207
Divol F, Couch D, Conéjéro G, Roschzttardtz H, Mari S, Curie C (2013) The Arabidopsis YELLOW STRIPE LIKE4 and 6 transporters control iron release from the chloroplast. Plant Cell 25:1040–1055. https://doi.org/10.1105/tpc.112.107672
Duy D, Wanner G, Meda AR, von Wiren N, Soll J, Philippar K (2007) PIC1, an ancient permease in Arabidopsis chloroplasts, mediates iron transport. Plant Cell 19:986–1006. https://doi.org/10.1105/tpc.106.047407
Duy D, Stübe R, Wanner G, Philippar K (2011) The chloroplast permease PIC1 regulates plant growth and development by directing homeostasis and transport of iron. Plant Physiol 155:1709–1722. https://doi.org/10.1104/pp.110.170233
Flis P, Ouerdane L, Grillet L, Curie C, Mari S, Lobinski R (2016) Inventory of metal complexes circulating in plant fluids: a reliable method based on HPLC coupled with dual elemental and high-resolution molecular mass spectrometric detection. New Phytol 211:1129–1141. https://doi.org/10.1111/nph.13964
Greenwood NN, Gibb TC (1971) Mössbauer spectroscopy. Chapman and Hall Ltd., London
Hantzis LJ, Kroh GE, Jahn CE, Cantrell M, Peers G, Pilon M, Ravet K (2018) A program for iron economy during deficiency targets specific Fe proteins. Plant Physiol 176:596–610. https://doi.org/10.1104/pp.17.01497
Homonnay Z, Szilágyi PÁ, Vértes A, Kuzmann E, Sharma VK, Molnár G, Bousseksou A, Greneche JM, Brausam A, Meier R et al (2008) Iron chelates: a challenge to chemists and Mössbauer spectroscopists. Hyperfine Interact 182:77–86. https://doi.org/10.1007/s10751-008-9713-x
Hu X, Page MT, Sumida A, Tanaka A, Terry MJ, Tanaka R (2017) The iron–sulfur cluster biosynthesis protein SUFB is required for chlorophyll synthesis, but not phytochrome signaling. Plant J 89:1184–1194. https://doi.org/10.1111/tpj.13455
Jain A, Wilson GT, Connolly EL (2014) The diverse roles of FRO family metalloreductases in iron and copper homeostasis. Front Plant Sci 5:100. https://doi.org/10.3389/fpls.2014.00100
Jeong J, Guerinot ML (2009) Homing in on iron homeostasis in plants. Trends Plant Sci 14:280–285. https://doi.org/10.1016/j.tplants.2009.02.006
Jeong J, Cohu C, Kerkeb L, Pilon M, Connolly EL, Guerinot ML (2008) Chloroplast Fe(III) chelate reductase activity is essential for seedling viability under iron limiting conditions. Proc Natl Acad Sci USA 105:10619–10624. https://doi.org/10.1073/pnas.0708367105
Klencsár Z, Kuzmann E, Vértes A (1996) User-friendly software for Mössbauer spectrum analysis. J Radioanal Nucl Chem 210:105–118. https://doi.org/10.1007/BF02055410
Kovács K, Tóth B, Lévai L, Vértes A, Kuzmann E, Abadía J, Fodor F (2010) Effect of pH and iron complexes on the iron species found in the roots of iron deficient cucumber after iron supply. In: Fodor F (ed) 15th international symposium on iron nutrition and interactions in plants: program and abstracts, Budapest, p 85
Kovács K, Pechoušek J, Machala L, Zbořil R, Klencsár Z, Solti Á, Toth B, Müller B, Pham HD, Kristóf Z et al (2016) Revisiting the iron pools in cucumber roots: identification and localization. Planta 244:167–179. https://doi.org/10.1007/s00425-016-2502-x
Kutrowska A, Szelag M (2014) Low-molecular weight organic acids and peptides involved in the long-distance transport of trace metals. Acta Physiol Plant 36:1957–1968. https://doi.org/10.1007/s11738-014-1576-y
Laemmli UK (1970) Cleavage of structural proteins during assembly of the head of bacteriophage T4. Nature 227:680–685. https://doi.org/10.1038/227680a0
Liu DH, Adler K, Stephan UW (1998) Iron-containing particles accumulate in organelles and vacuoles of leaf and root cells in the nicotianamine-free tomato mutant chloronerva. Protoplasma 201:213–220. https://doi.org/10.1007/BF01287417
López-Millán AF, Duy D, Philippar K (2016) Chloroplast iron transport proteins—function and impact on plant physiology. Front Plant Sci 7:178. https://doi.org/10.3389/fpls.2016.00178
Lucena JJ, Chaney RL (2006) Synthetic iron chelates as substrates of root ferric chelate reductase in green stressed cucumber plants. J Plant Nutr 29:423–439. https://doi.org/10.1080/01904160500524886
Miyakoshi K, Oshita J, Kitahara T (2001) Expeditious synthesis of nicotianamine and 2′-deoxymugineic acid. Tetrahedron 57:3355–3360. https://doi.org/10.1016/S0040-4020(01)00215-0
Nagajyoti PC, Lee KD, Sreekanth TVM (2010) Heavy metals, occurrence and toxicity for plants: a review. Environ Chem Lett 8:199–216. https://doi.org/10.1007/s10311-010-0297-8
Pechoušek J, Prochazka R, Jancik D, Frydrych J, Mashlan M (2010) Universal LabVIEW-powered Mössbauer spectrometer based on USB, PCI or PXI devices. J Phys Conf Ser 217:1–4. https://doi.org/10.1088/1742-6596/217/1/012006/meta
Pechoušek J, Jančík D, Frydrych J, Navařík J, Novák P (2012) Setup of Mössbauer spectrometers at RCPTM. AIP Conf Proc 1489:186–193. https://doi.org/10.1063/1.4759489
Petrouleas V, Diner BA (1982) Investigation of the iron components in photosystem II by Mössbauer spectroscopy. FEBS Lett 147:111–114. https://doi.org/10.1016/0014-5793(82)81022-3
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acid Res 29:e45. https://doi.org/10.1093/nar/29.9.e45
Pich A, Manteuffel R, Hillmer S, Scholz G, Schmidt W (2001) Fe homeostasis in plant cells: does nicotianamine play multiple roles in the regulation of cytoplasmic Fe concentration? Planta 213:967–976. https://doi.org/10.1007/s004250100573
Porra RJ, Thompson WA, Kriedemann PE (1989) Determination of accurate extinction coefficients and simultaneous equations for assaying chlorophylls a and b extracted with 4 different solvents: verification of the concentration of chlorophyll standards by atomic absorption spectroscopy. Biochim Biophys Acta 975:384–394. https://doi.org/10.1016/S0005-2728(89)80347-0
Ravet K, Pilon M (2013) Copper and iron homeostasis in plants: the challenges of oxidative stress. Antioxid Redox Sign 19:919–932. https://doi.org/10.1089/ars.2012.5084
Reichman SM, Parker DR (2002) Revisiting the metal-binding chemistry of nicotianamine and 2′-deoxymugineic acid. Implications for iron nutrition in strategy II plants. Plant Physiol 129:1435–1438. https://doi.org/10.1104/pp.005009
Reinbothe C, Bartsch S, Eggink LL, Hoober JK, Brusslan J, Andrade-Paz R, Monnet J, Reinbothe S (2006) A role for chlorophyllide a oxygenase in the regulated import and stabilization of light-harvesting chlorophyll a/b proteins. Proc Natl Acad Sci USA 103:4777–4782. https://doi.org/10.1073/pnas.0511066103
Rellán-Álvarez R, Abadía J, Álvarez-Fernández A (2008) Formation of metal–nicotianamine complexes as affected by pH, ligand exchange with citrate and metal exchange. A study by electrospray ionization time-of-flight mass spectrometry. Rapid Commun Mass Spectrom 22:1553–1562. https://doi.org/10.1002/rcm.3523
Rellán-Álvarez R, Giner-Martinez-Sierra J, Orduna J, Orera I, Angel Rodríguez-Castrillón J, Ignacio García-Alonso J, Abadía J, Álvarez-Fernández A (2010) Identification of a tri-iron(III), tri-citrate complex in the xylem sap of iron-deficient tomato resupplied with iron: new insights into plant iron long-distance transport. Plant Cell Physiol 51:91–102. https://doi.org/10.1093/pcp/pcp170
Schuler M, Rellán-Álvarez R, Fink-Straube C, Abadía J, Bauer P (2012) Nicotianamine functions in the phloem-based transport of iron to sink organs, in pollen development and pollen tube growth in Arabidopsis. Plant Cell 24:2380–2400. https://doi.org/10.1105/tpc.112.099077
Shingles R, North M, McCarty RE (2002) Ferrous ion transport across chloroplast inner envelope membranes. Plant Physiol 128:1022–1030. https://doi.org/10.1104/pp.010858
Smith GF, McCurdy WH, Diehl H (1952) The colorimetric determination of iron in raw and treated municipal water supplies by use of 4:7-diphenyl-1:10-phenanthroline. Analyst 77:418–422
Solti Á, Kovács K, Basa B, Vértes A, Sárvári É, Fodor F (2012) Uptake and incorporation of iron in sugar beet chloroplasts. Plant Physiol Biochem 52:91–97. https://doi.org/10.1016/j.plaphy.2011.11.010
Solti Á, Müller B, Czech V, Sárvári É, Fodor F (2014) Functional characterization of the chloroplast ferric chelate oxidoreductase enzyme. New Phytol 202:920–928. https://doi.org/10.1111/nph.12715
Solti Á, Kovács K, Müller B, Vázquez S, Hamar É, Pham HD, Tóth B, Abadía J, Fodor F (2016) Does a voltage-sensitive outer envelope transport mechanism contributes to the chloroplast iron uptake? Planta 244:1303–1313. https://doi.org/10.1007/s00425-016-2586-3
Spiller SC, Castelfranco AM, Castelfranco PA (1982) Effects of iron and oxygen on chlorophyll biosynthesis I. In vivo observations on iron and oxygen-deficient plants. Plant Physiol 69:107–111. https://doi.org/10.1104/pp.69.1.107
Sugiura Y, Tanaka H, Mino Y, Ishida T, Ota N, Inoue M, Nomoto K, Yoshioka H, Takemoto T (1981) Structure, properties, and transport mechanism of iron(III) complex of mugineic acid, a possible phytosiderophore. J Am Chem Soc 103:6979–6982. https://doi.org/10.1021/ja00413a043
Terry N, Abadía J (1986) Function of iron in chloroplasts. J Plant Nutr 9:609–646. https://doi.org/10.1080/01904168609363470
Terry N, Low G (1982) Leaf chlorophyll content and its relation to the intracellular localization of iron. J Plant Nutr 5:301–310. https://doi.org/10.1080/01904168209362959
Vértes A, Nagy DL (1990) Mössbauer spectroscopy of frozen solutions. Akadémiai Kiadó, Budapest
Vértes A, Nagy-Czako I, Burger K (1978) Moessbauer study of equilibrium constants of solvates 3. Solvent–solute interactions in nonaqueous solutions of iron(III) chloride. J Phys Chem 82:1469–1473. https://doi.org/10.1021/j100502a004
von Wirén N, Klair S, Bansal S, Briat JF, Khodr H, Shioiri T, Leigh RA, Hider RC (1999) Nicotianamine chelates both FeIII and FeII. Implications for metal transport in plants. Plant Physiol 119:1107–1114. https://doi.org/10.1104/pp.119.3.1107
Weber G, von Wirén N, Hayen H (2008) Investigation of ascorbate-mediated iron release from ferric phytosiderophores in the presence of nicotianamine. Biometals 21:503–513. https://doi.org/10.1007/s10534-008-9137-8
Zanin L, Tomasi N, Rizzardo C, Gottardi S, Terzano R, Alfeld M, Janssens K, de Nobili M, Mimmo T, Cesco S (2015) Iron allocation in leaves of Fe-deficient cucumber plants fed with natural Fe complexes. Physiol Plant 154:82–94. https://doi.org/10.1111/ppl.12296
Acknowledgements
This work was supported by the grants financed by the National Research, Development and Innovation Office, Hungary (NKFIH PD-112047, PD-111979 and K-124159), VEKOP-2.3.3-15-2016-00008 and the Spanish Ministry of Economy and Competitiveness (MINECO; project AGL2016-75226-R, co-financed with FEDER). Á.S. was also supported by the Bolyai János Research Scholarship of the Hungarian Academy of Sciences (BO/00207/15/4). The authors (JP, LM, and RZ) gratefully acknowledge the financial support provided by the project LO1305 of the Ministry of Education, Youth and Sports of the Czech Republic.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest in relation with the submitted manuscript.
Electronic supplementary material
Below is the link to the electronic supplementary material.
425_2018_3037_MOESM1_ESM.pdf
Supplementary material 1 Online Resource S1 Extended evaluation of the 57Fe Mössbauer spectra of the Fe(III)–nicotianamine frozen solution (PDF 28 kb)
425_2018_3037_MOESM2_ESM.pdf
Supplementary material 2 Online Resource S2 Table on the 57Fe Mössbauer parameters associated with the spectra of the Fe(III)–nicotianamine frozen solution (PDF 33 kb)
425_2018_3037_MOESM3_ESM.pdf
Supplementary material 3 Online Resource S3 Figure on the expression of BnYsl4 in tissues and organs of different developmental stages expressed in normalised relative quantities (NRQ) (PDF 48 kb)
Rights and permissions
About this article
Cite this article
Müller, B., Kovács, K., Pham, HD. et al. Chloroplasts preferentially take up ferric–citrate over iron–nicotianamine complexes in Brassica napus. Planta 249, 751–763 (2019). https://doi.org/10.1007/s00425-018-3037-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-018-3037-0