Abstract
Main conclusion
Genome-wide identification, expression analysis, and functional characterization of previously uncharacterized hexokinase family of oil crop, Brassica napus, underscore the importance of this gene family in plant growth and development.
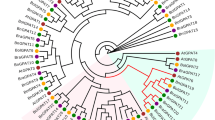
In plants, the multi-gene family of dual-function hexokinases (HXKs) plays important roles in sugar metabolism and sensing that affect growth and development. Rapeseed (Brassica napus L.) is an important oil crop; however, little is known about the B. napus HXK gene family. We identified 19 putative HXKs in B. napus genome. B. rapa and B. oleracea, the two diploid progenitors of B. napus, contributed almost equally to the BnHXK genes. Phylogenetic analysis divided the 19 BnHXKs into four groups. The exon–intron structures of BnHXKs share high similarity to those of HXKs in Arabidopsis and rice. The group III and IV BnHXKs are highly expressed in roots, whereas group I members preferentially express in leaves. Analysis of seed transcriptomes at different developmental stages showed that most of group I and IV HXKs are highly expressed 2-weeks after pollination (2WAP), compared to 4WAP for group III. BnHKXs are differentially expressed in susceptible and tolerant B. napus cultivars after fungal infection, suggesting the possible involvement in defense response. We generated rapeseed RNAi lines for BnHXK9, a member of relatively less characterized group IV, by pollen-mediated gene transformation. The seedlings of BnHXK9-RNAi lines showed delayed growth compared to the wild type. The RNAi plants were dwarf with curly leaves, suggesting the involvement of BnHXK9 in plant development. Collectively, our findings provides a comprehensive account of BnHXK gene family in an important crop and a starting point for further elucidation of their roles in sugar metabolism and sensing, as well as plant growth and development.






Similar content being viewed by others
Change history
21 April 2018
In the original version of this article the name of the second author was misspelled. The correct name is: Xiaomin Wang
Abbreviations
- FA:
-
Fatty acid
- Glc:
-
Glucose
- Glc-6-P:
-
Glc-6-phosphate
- HXK:
-
Hexokinase
- MS:
-
Murashige and Skoog
References
Aders K, Larsson B, von Heijne G, Sonnahammer ELL (2001) Predictingtransmembrane protein topology with a hidden markov model: application to complete genomes. J Mol Biol 305:567–580
Aguilera-Alvarado GP, Sanchez-Nieto S (2017) Plant hexokinases are multifaceted proteins. Plant Cell Physiol 58:1151–1160
Ahuatzi D, Herrero P, de la Cera T, Moreno F (2004) The glucose-regulated nuclear localization of hexokinase 2 in Saccharomyces cerevisiae is Mig1-dependent. J Biol Chem 279:14440–14446
Allen GC, Flores-Vergara MA, Krasynanski S, Kumar S, Thompson WF (2006) A modified protocol for rapid DNA isolation from plant tissues using cetyltrimethylammonium bromide. Nat Protoc 1:2320–2325
Camacho C, Coulouris G, Avagyan V, Ma N, Papadopoulos J, Bealer K, Madden TL (2009) BLAST+: architecture and applications. BMC Bioinform 10:421
Chalhoub B, Denoeud F, Liu S, Parkin IA, Tang H, Wang X, Chiquet J, Belcram H, Tong C, Samans B, Correa M, Da Silva C, Just J, Falentin C, Koh CS, Le Clainche I, Bernard M, Bento P, Noel B, Labadie K, Alberti A, Charles M, Arnaud D, Guo H, Daviaud C, Alamery S, Jabbari K, Zhao M, Edger PP, Chelaifa H, Tack D, Lassalle G, Mestiri I, Schnel N, Le Paslier MC, Fan G, Renault V, Bayer PE, Golicz AA, Manoli S, Lee TH, Thi VH, Chalabi S, Hu Q, Fan C, Tollenaere R, Lu Y, Battail C, Shen J, Sidebottom CH, Wang X, Canaguier A, Chauveau A, Berard A, Deniot G, Guan M, Liu Z, Sun F, Lim YP, Lyons E, Town CD, Bancroft I, Wang X, Meng J, Ma J, Pires JC, King GJ, Brunel D, Delourme R, Renard M, Aury JM, Adams KL, Batley J, Snowdon RJ, Tost J, Edwards D, Zhou Y, Hua W, Sharpe AG, Paterson AH, Guan C, Wincker P (2014) Plant genetics. Early allopolyploid evolution in the post-Neolithic Brassica napus oilseed genome. Science 345:950–953
Cheng W, Zhang H, Zhou X, Liu H, Liu Y, Li J, Han S, Wang Y (2011) Subcellular localization of rice hexokinase (OsHXK) family members in the mesophyll protoplasts of tobacco. Biol Plant 55:173–177
Cho JI, Ryoo N, Ko S, Lee SK, Lee J, Jung KH, Lee YH, Bhoo SH, Winderickx J, An G, Hahn TR, Jeon JS (2006a) Structure, expression, and functional analysis of the hexokinase gene family in rice (Oryza sativa L.). Planta 224:598–611
Cho YH, Yoo SD, Sheen J (2006b) Regulatory functions of nuclear hexokinase1 complex in glucose signaling. Cell 127:579–589
Claeyssen E, Rivoal J (2007) Isozymes of plant hexokinase: occurrence, properties and functions. Phytochemistry 68:709–731
da Silva WS, Rezende GL, Galina A (2001) Subcellular distribution and kinetic properties of cytosolic and non-cytosolic hexokinases in maize seedling roots: implications for hexose phosphorylation. J Exp Bot 52:1191–1201
Dai N, Schaffer A, Petreikov M, Shahak Y, Giller Y, Ratner K, Levine A, Granot D (1999) Overexpression of Arabidopsis hexokinase in tomato plants inhibits growth, reduces photosynthesis, and induces rapid senescence. Plant Cell 11:1253–1266
Emanuelsson O, Brunak S, von Heijne G, Nielsen H (2007) Locating proteins in the cell using TargetP, SignalP and related tools. Nat Protoc 2:953–971
Finn RD, Coggill P, Eberhardt RY, Eddy SR, Mistry J, Mitchell AL, Potter SC, Punta M, Qureshi M, Sangrador-Vegas A, Salazar GA, Tate J, Bateman A (2016) The Pfam protein families database: towards a more sustainable future. Nucleic Acids Res 44:D279–D285
Geng MT, Yao Y, Wang YL, Wu XH, Sun C, Li RM, Fu SP, Duan RJ, Liu J, Hu XW, Guo JC (2017) Structure, expression, and functional analysis of the hexokinase gene family in cassava. Int J Mol Sci 18:E1041
Girard IJ, Tong C, Becker MG, Mao X, Huang J, de Kievit T, Fernando WGD, Liu S, Belmonte MF (2017) RNA sequencing of Brassica napus reveals cellular redox control of Sclerotinia infection. J Exp Bot 68:5079–5091
Granot D, Kelly G, Stein O, David-Schwartz R (2014) Substantial roles of hexokinase and fructokinase in the effects of sugars on plant physiology and development. J Exp Bot 65:809–819
Gu Z, Eils R, Schlesner M (2016) Complex heatmaps reveal patterns and correlations in multidimensional genomic data. Bioinformatics 32:2847–2849
Herbers K, Meuwly P, Frommer WB, Metraux JP, Sonnewald U (1996) Systemic acquired resistance mediated by the ectopic expression of invertase: possible hexose sensing in the secretory pathway. Plant Cell 8:793–803
Hill LM, Morley-Smith ER, Rawsthorne S (2003) Metabolism of sugars in the endosperm of developing seeds of oilseed rape. Plant Physiol 131:228–236
Hu B, Jin J, Guo AY, Zhang H, Luo J, Gao G (2015) GSDS 2.0: an upgraded gene feature visualization server. Bioinformatics 31:1296–1297
Jang JC, Leon P, Zhou L, Sheen J (1997) Hexokinase as a sugar sensor in higher plants. Plant Cell 9:5–19
Kandel-Kfir M, Damari-Weissler H, German MA, Gidoni D, Mett A, Belausov E, Petreikov M, Adir N, Granot D (2006) Two newly identified membrane-associated and plastidic tomato HXKs: characteristics, predicted structure and intracellular localization. Planta 224:1341–1352
Karve A, Moore BD (2009) Function of Arabidopsis hexokinase-like1 as a negative regulator of plant growth. J Exp Bot 60:4137–4149
Karve A, Rauh BL, Xia X, Kandasamy M, Meagher RB, Sheen J, Moore BD (2008) Expression and evolutionary features of the hexokinase gene family in Arabidopsis. Planta 228:411–425
Karve R, Lauria M, Virnig A, Xia X, Rauh BL, Moore B (2010) Evolutionary lineages and functional diversification of plant hexokinases. Mol Plant 3:334–346
Kelly G, Moshelion M, David-Schwartz R, Halperin O, Wallach R, Attia Z, Belausov E, Granot D (2013) Hexokinase mediates stomatal closure. Plant J 75:977–988
Kersey PJ, Allen JE, Allot A, Barba M, Boddu S, Bolt BJ, Carvalho-Silva D, Christensen M, Davis P, Grabmueller C, Kumar N, Liu Z, Maurel T, Moore B, McDowall MD, Maheswari U, Naamati G, Newman V, Ong CK, Paulini M, Pedro H, Perry E, Russell M, Sparrow H, Tapanari E, Taylor K, Vullo A, Williams G, Zadissia A, Olson A, Stein J, Wei S, Tello-Ruiz M, Ware D, Luciani A, Potter S, Finn RD, Urban M, Hammond-Kosack KE, Bolser DM, De Silva N, Howe KL, Langridge N, Maslen G, Staines DM, Yates A (2018) Ensembl Genomes 2018: an integrated omics infrastructure for non-vertebrate species. Nucleic Acids Res 46:D802–D808
Kim M, Lim JH, Ahn CS, Park K, Kim GT, Kim WT, Pai HS (2006) Mitochondria-associated hexokinases play a role in the control of programmed cell death in Nicotiana benthamiana. Plant Cell 18:2341–2355
Kim YM, Heinzel N, Giese JO, Koeber J, Melzer M, Rutten T, Von Wiren N, Sonnewald U, Hajirezaei MR (2013) A dual role of tobacco hexokinase 1 in primary metabolism and sugar sensing. Plant Cell Environ 36:1311–1327
Kodama Y, Shumway M, Leinonen R, International Nucleotide Sequence Database Consortium (2012) The Sequence Read Archive: explosive growth of sequencing data. Nucleic Acids Res 40:D54–D56
Kuser PR, Krauchenco S, Antunes OA, Polikarpov I (2000) The high resolution crystal structure of yeast hexokinase PII with the correct primary sequence provides new insights into its mechanism of action. J Biol Chem 275:20814–20821
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9:357–359
Larkin MA, Blackshields G, Brown NP, Chenna R, McGettigan PA, McWilliam H, Valentin F, Wallace IM, Wilm A, Lopez R, Thompson JD, Gibson TJ, Higgins DG (2007) Clustal W and Clustal X version 2.0. Bioinformatics 23:2947–2948
Le BH, Cheng C, Bui AQ, Wagmaister JA, Henry KF, Pelletier J, Kwong L, Belmonte M, Kirkbride R, Horvath S, Drews GN, Fischer RL, Okamuro JK, Harada JJ, Goldberg RB (2010) Global analysis of gene activity during Arabidopsis seed development and identification of seed-specific transcription factors. Proc Natl Acad Sci USA 107:8063–8070
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) method. Methods 25:402–408
Lugassi N, Kelly G, Fidel L, Yaniv Y, Attia Z, Levi A, Alchanatis V, Moshelion M, Raveh E, Carmi N, Granot D (2015) Expression of Arabidopsis hexokinase in citrus guard cells controls stomatal aperture and reduces transpiration. Front Plant Sci 6:1114
Mistry J, Finn RD, Eddy SR, Bateman A, Punta M (2013) Challenges in homology search: HMMER3 and convergent evolution of coiled-coil regions. Nucleic Acids Res 41:e121
Moore B, Zhou L, Rolland F, Hall Q, Cheng WH, Liu YX, Hwang I, Jones T, Sheen J (2003) Role of the Arabidopsis glucose sensor HXK1 in nutrient, light, and hormonal signaling. Science 300:332–336
Nguyen-Quoc B, Foyer CH (2001) A role for ‘futile cycles’ involving invertase and sucrose synthase in sucrose metabolism of tomato fruit. J Exp Bot 52:881–889
Niu Y, Wu GZ, Ye R, Lin WH, Shi QM, Xue LJ, Xu XD, Li Y, Du YG, Xue HW (2009) Global analysis of gene expression profiles in Brassica napus developing seeds reveals a conserved lipid metabolism regulation with Arabidopsis thaliana. Mol Plant 2:1107–1122
Olsson T, Thelander M, Ronne H (2003) A novel type of chloroplast stromal hexokinase is the major glucose-phosphorylating enzyme in the moss Physcomitrella patens. J Biol Chem 278:44439–44447
Pattanaik S, Kong Q, Zaitlin D, Werkman JR, Xie CH, Patra B, Yuan L (2010) Isolation and functional characterization of a floral tissue-specific R2R3 MYB regulator from tobacco. Planta 231:1061–1076
Pelaez R, Herrero P, Moreno F (2010) Functional domains of yeast hexokinase 2. Biochem J 432:181–190
Rojas CM, Senthil-Kumar M, Tzin V, Mysore KS (2014) Regulation of primary plant metabolism during plant–pathogen interactions and its contribution to plant defense. Front Plant Sci 5:17
Rolland F, Moore B, Sheen J (2002) Sugar sensing and signaling in plants. Plant Cell 14(Suppl):S185–S205
Schmieder R, Edwards R (2011) Quality control and preprocessing of metagenomic datasets. Bioinformatics 27:863–864
Sheen J, Zhou L, Jang JC (1999) Sugars as signaling molecules. Curr Opin Plant Biol 2:410–418
Singh SK, Wu Y, Ghosh JS, Pattanaik S, Fisher C, Wang Y, Lawson D, Yuan L (2015) RNA-sequencing reveals global transcriptomic changes in Nicotiana tabacum responding to topping and treatment of axillary-shoot control chemicals. Sci Rep 5:18148
Tadege M, Bucher M, Stahli W, Suter M, Dupuis I, Kuhlemeier C (1998) Activation of plant defense responses and sugar efflux by expression of pyruvate decarboxylase in potato leaves. Plant J 16:661–671
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Mol Biol Evol 30:2725–2729
Troncoso-Ponce MA, Rivoal J, Dorion S, Moisan MC, Garces R, Martinez-Force E (2011) Cloning, biochemical characterization and expression of a sunflower (Helianthus annuus L.) hexokinase associated with seed storage compounds accumulation. J Plant Physiol 168:299–308
Wang JX, Sun Y, Cui GM, Hu JJ (2001) Transgenic maize plants obtained by pollen-mediated transformation. Acta Bot Sin 43:275–279
Wang J, Singh SK, Du C, Li C, Fan J, Pattanaik S, Yuan L (2016) Comparative transcriptomic analysis of two Brassica napus near-isogenic lines reveals a network of genes that influences seed oil accumulation. Front Plant Sci 7:1498
Wiese A, Groner F, Sonnewald U, Deppner H, Lerchl J, Hebbeker U, Flugge U, Weber A (1999) Spinach hexokinase I is located in the outer envelope membrane of plastids. FEBS Lett 461:13–18
Xiao W, Sheen J, Jang JC (2000) The role of hexokinase in plant sugar signal transduction and growth and development. Plant Mol Biol 44:451–461
Xu FQ, Li XR, Ruan YL (2008) RNAi-mediated suppression of hexokinase gene OsHXK10 in rice leads to non-dehiscent anther and reduction of pollen germination. Plant Sci 175:674–684
Zhang ZW, Yuan S, Xu F, Yang H, Zhang NH, Cheng J, Lin HH (2010) The plastid hexokinase pHXK: a node of convergence for sugar and plastid signals in Arabidopsis. FEBS Lett 584:3573–3579
Acknowledgements
The authors thank Chunfeng Du and Shuanshi Xian, Cotton Research Institute of Shanxi Academy of Agricultural Sciences, for providing the plant material. This project is supported by Shanxi Provincial Science and Technology Research Grant (20140311010-4), China. The work is supported in part by the National Science Foundation under Cooperative Agreement no. 1355438 to L.Y.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
The original version of this article was revised: Spelling of second author was incorrect. Now it has been corrected.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, J., Wang, X., Geng, S. et al. Genome-wide identification of hexokinase gene family in Brassica napus: structure, phylogenetic analysis, expression, and functional characterization. Planta 248, 171–182 (2018). https://doi.org/10.1007/s00425-018-2888-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-018-2888-8