Abstract
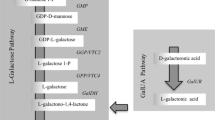
Citrus fruits are an important source of ascorbic acid (AsA) for human nutrition, but the main pathways involved in its biosynthesis and their regulation are still not fully characterized. To study the transcriptional regulation of AsA accumulation, expression levels of 13 genes involved in AsA biosynthesis, 5 in recycling and 5 in degradation were analyzed in peel and pulp of fruit of two varieties with different AsA concentration: Navel orange (Citrus sinensis) and Satsuma mandarin (Citrus unshiu). AsA accumulation in peel and pulp correlated with the transcriptional profiling of the l-galactose pathway genes, and the myo-inositol pathway appeared to be also relevant in the peel of immature-green orange. Differences in AsA content between varieties were associated with differential gene expression of GDP-mannose pyrophosphorylase (GMP), GDP-l-galactose phosphorylase (GGP) and l-galactose-1-phosphate phosphatase (GPP), myo-inositol oxygenase in peel, and GGP and GPP in pulp. Relative expressions of monodehydroascorbate reductase 3 (MDHAR3) and dehydroascorbate reductase1 (DHAR1) correlated with AsA accumulation during development and ripening in peel and pulp, respectively, and were more highly expressed in the variety with higher AsA contents. Collectively, results indicated a differential regulation of AsA concentration in peel and pulp of citrus fruits that may change during the different stages of fruit development. The l-galactose pathway appears to be predominant in both tissues, but AsA concentration is regulated by complex mechanisms in which degradation and recycling also play important roles.






Similar content being viewed by others
Abbreviations
- AsA:
-
Ascorbic acid
- AO:
-
Ascorbate oxidase
- APX:
-
Ascorbate peroxidase
- CP:
-
Crossing point
- DHAR:
-
Dehydroascorbate reductase
- GalLDH:
-
l-Galactono-1,4-lactone dehydrogenase
- GalDH:
-
l-Galactose dehydrogenase
- GalUR:
-
d-Galacturonic acid reductase
- GGP:
-
GDP-l-Galactose phosphorylase
- GLOase:
-
l-Gulono-1,4-lactone oxidase
- GME:
-
GDP-Mannose-3′,5′-epimerase
- GMP:
-
GDP-Mannose pyrophosphorylase
- GPP:
-
l-Galactose-1-phosphate phosphatase
- MDHAR:
-
Monodehydroascorbate reductase
- MIOX:
-
Myo-inositol oxygenase
References
Abeysinghe DC, Li X, Sun CD, Zhang WS, Zhou CH, Chen KS (2007) Bioactive compounds and antioxidant capacities in different edible tissues of citrus fruit of four species. Food Chem 104:1338–1344
Agius F, González-Lamothe R, Caballero JL, Muñoz-Blanco J, Botella MA, Valpuesta V (2003) Engineering increased vitamin C levels in plants by overexpression of a D-galacturonic acid reductase. Nature Biotechnol 21:177–181
Alhagdow M, Mounet F, Gilbert L, Nunes-Nesi A, Garcia V, Just D, Petit J, Beauvoit B, Fernie AR, Rothan C, Baldet P (2007) Silencing of the mitochondrial ascorbate synthesizing enzyme l-galactono-1, 4-lactone dehydrogenase affects plant and fruit development in tomato. Plant Physiol 145:1408–1422
Alós E, Rodrigo MJ, Zacarías L (2013) Transcriptomic analysis of genes involved in the biosynthesis, recycling and degradation of l-ascorbic acid in pepper fruits (Capsicum annuum L). Plant Sci 207:2–11
Atkins CD, Wiederhold E, Moore EL (1945) Vitamin C content of processing residue from Florida citrus waste. Fruit Prod J 24:260–262
Badejo AA, Fujikawa Y, Esaka M (2009) Gene expression of ascorbic acid biosynthesis related enzymes of the Smirnoff–Wheeler pathway in acerola (Malpighia glabra). J Plant Physiol 166:652–660
Badejo AA, Wada K, Gao Y, Maruta T, Sawa Y, Shigeoka S, Ishikawa T (2012) Translocation and the alternative d-galacturonate pathway contribute to increasing the ascorbate level in ripening tomato fruits together with the d-mannose/l-galactose pathway. J Exp Bot 63:229–329
Bermejo A, Llosa MJ, Cano A (2011) Analysis of bioactive compounds in seven citrus cultivars. Food Sci Technol Int 17:55–62
Block G (1991) Dietary guidelines and the results of food consumption surveys. Am J Clin Nutr 53:3565–3575
Bulley SM, Rassam M, Hoser D, Otto W, Schünemann N, Wright M, MacRae E, Gleave A, Laing W (2009) Gene expression studies in kiwifruit and gene over-expression in Arabidopsis indicates that GDP-l-galactose guanyltransferase is a major control point of vitamin C biosynthesis. J Exp Bot 60:765–778
Bulley S, Wright M, Rommens C, Yan H, Rassam M, Lin-Wang K, Andre C, Brewster D, Karunairetnam S, Allan AC, Laing WA (2012) Enhancing ascorbate in fruits and tubers through over-expression of the l-galactose pathway gene GDP-l-galactose phosphorylase. Plant Biotechnol J 10:390–397
Bustin SA, Benes V, Garson JA et al (2009) The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55:611–622
Chen Z, Young TE, Ling J, Chang S, Gallie DR (2003) Increasing vitamin C content of plants through enhanced ascorbate recycling. Proc Natl Acad Sci USA 100:3525–3530
Cruz-Rus E, Botella MA, Valpuesta V, Gomez-Jimenez MC (2010) Analysis of genes involved in l-ascorbic acid biosynthesis during growth and ripening of grape berries. J Plant Physiol 167:739–748
Cruz-Rus E, Amaya I, Sánchez-Sevilla JF, Botella MA, Valpuesta V (2011) Regulation of l-ascorbic acid content in strawberry fruits. J Exp Bot 62:4191–4201
Cruz-Rus E, Amaya I, Valpuesta V (2012) The challenge of increasing vitamin C content in plant foods. Biotechnol J 7:1110–1121
Davey MW, Dekempeener E, Keulemans J (2003) Rocket-powered high-performance liquid chromatographic analysis of plant ascorbate and glutathione. Anal Biochem 316:74–81
Davey MW, Franck C, Keulemans J (2004) Distribution, developmental and stress responses of antioxidant metabolism in malus. Plant Cell Environ 27:1309–1320
Dhuique-Mayer C, Caris-Veyrat C, Ollitrault P, Curk F, Amiot MJ (2005) Varietal and interspecific influence on micronutrient contents in citrus from the Mediterranean area. J Agric Food Chem 52:2140–2145
Endres S, Tenhaken R (2009) Myoinositol oxigenase controls the level of myoinositol in Arabidopsis, but not increase ascorbic acid. Plant Physiol 149:1042–1049
Fukunaga K, Fujikawa Y, Esaka M (2010) Light regulation of ascorbic acid biosynthesis in rice via light responsive cis-elements in genes encoding ascorbic acid biosynthetic enzymes. Biosci Biotechnol Biochem 74:888–891
Gallie DR (2013) The role of l-ascorbic acid recycling in responding to environmental stress and in promoting plant growth. J Exp Bot 64:433–443
Haroldsen VM, Chi-Ham CL, Kulkarni S, Lorence A, Bennett AB (2011) Constitutively expressed DHAR and MDHAR influence fruit, but not foliar ascorbate levels in tomato. Plant Physiol Biochem 49:1244–1249
Imai T, Karita S, Shiratori G, Hattori M, Nunome T, Oba K, Hirai M (1998) l-Galactono-γ-lactone dehydrogenase from sweet potato: purification and cDNA sequence analysis. Plant Cell Physiol 39:1350–1358
Imai T, Ban Y, Terakami S, Yamamoto T, Moriguchi T (2009) l-ascorbate biosynthesis in peach: cloning of six l-galactose pathway-related genes and their expression during peach fruit development. Physiol Plant 136:139–149
Ioannidi E, Kalamaki MS, Engineer C, Pateraki I, Alexandrou D, Mellidou I, Giovannonni J, Kanellis AK (2009) Expression profiling of ascorbic acid-related genes during tomato fruit development and ripening and in response to stress conditions. J Exp Bot 60:663–678
Li MJ, Ma FW, Zhang M, Pu F (2008) Distribution and metabolism of ascorbic acid in apple fruits (Malus domestica Borkh cv. Gala). Plant Sci 174:606–612
Li M, Ma F, Shang P, Zhang M, Hou C, Liang D (2009) Influence of light on ascorbate formation and metabolism in apple fruits. Planta 230:39–51
Lima-Silva V, Rosado A, Amorim-Silva V et al (2012) Genetic and genome-wide transcriptomic analyses identify co-regulation of oxidative response and hormone transcript abundance with vitamin C content in tomato fruit. BMC Genomics 13:187
Linster CL, Van Schaftingen E (2007) Vitamin C: biosynthesis, recycling and degradation in mammals. FEBS J 274:1–22
Loewus FA, Jang R, Seegmiller CG (1958) The conversion of C14-labeled sugars to l-ascorbic acid in ripening strawberries. IV. A comparative study of d-galacturonic acid and l-ascorbic acid formation. J Biol Chem 232:533–541
Lorence A, Nessler CL (2007) Pathway engineering of the plant vitamin C metabolic network. In: Verpoorte R, Alfermann AW, Johnson TS (eds) Applications of plant metabolic engineering. Springer, Dordrecht, pp 189–209
Martí N, Mena P, Cánovas JA, Micol V, Saura D (2009) Vitamin C and the role of citrus juices as functional food. Nat Prod Commun 4:677–700
Melino VJ, Soole KL, Ford CM (2009) Ascorbate metabolism and the developmental demand for tartaric and oxalic acids in ripening grape berries. BMC Plant Biol 9:145
Mellidou I, Chagné D, Laing WA, Keulemans J, Davey MW (2012a) Allelic variation in paralogs of GDP-l-galactose phosphorylase is a major determinant of vitamin C concentrations in apple fruit. Plant Physiol 160:1613–1629
Mellidou I, Keulemans J, Kanellis AK, Davey MW (2012b) Regulation of fruit ascorbic acid concentrations during ripening in high and low vitamin C tomato cultivars. BMC Plant Biol 17:239
Nagy S (1980) Vitamin C contents of citrus fruit and their products: a review. J Agric Food Chem 28:8–18
Oba K, Ishikawa S, Nishikawa M, Mizuno H, Yamamoto T (1995) Purification and properties of l-galactono-1,4-lactone dehydrogenase, a key enzyme for ascorbic acid biosynthesis, from sweet potato roots. J Biochem 117:120–124
Ostergaard J, Persiau G, Davey MW, Van Montagu M (1997) Isolation of a cDNA coding for l-galactono-γ-lactone dehydrogenase, an enzyme involved in the biosynthesis of ascorbic acid in plants. J Biol Chem 272:30009–30016
Pfaffl MW, Horgan GW, Dempfle L (2002) Relative expression software tool (REST) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acid Res 30:e36
Pfaffl MW, Tichopad A, Prgomet C, Neuvians TP (2004) Determination of stable housekeeping genes, differentially regulated target genes and sample integrity: bestkeeper-excel-based tool using pair-wise correlations. Biotechnol Lett 26:509–515
Qin A, Shi Q, Yu X (2011) Ascorbic acid contents in transgenic potato plants overexpressing two dehydroascorbate reductase genes. Mol Biol Rep 38:1557–1566
Rodrigo MJ, Marcos JF, Zacarías L (2004) Biochemical and molecular analysis of carotenoid biosynthesis in flavedo of orange (Citrus sinensis L.) during fruit development and maturation. J Agric Food Chem 52:6724–6731
Rodrigo MJ, Alquezar B, Alós E, Medina V, Carmona L, Bruno M, Al-Babili S, Zacarías L (2013) A novel carotenoid cleavage activity involved in the biosynthesis of citrus fruit-specific apocarotenoid pigments. J Exp Bot. doi:10.1093/jxb/ert260
Romero P, Lafuente MT, Rodrigo MJ (2012) The Citrus ABA signalosome: identification and transcriptional regulation during sweet orange fruit ripening and leaf dehydration. J Exp Bot 63:4931–4945
Sanmartin M, Pateraki I, Chatzopoulou F, Kanellis AK (2007) Differential expression of the ascorbate oxidase multigene family during fruit development and in response to stress. Planta 225:873–885
Smirnoff N (2011) Vitamin C: the metabolism and functions of ascorbic acid in plants. Adv Bot Res 59:109–177
Stewart I, Wheaton TA (1971) Continuous flow separation of carotenoids by liquid chromatography. J Chromatogr 55:325–336
Tadeo FR, Cercós M, Colmenero-Flores JM et al (2008) Molecular physiology of development and quality of citrus. Adv Bot Res 47:147–223
Tausz M, Sircelj H, Grill D (2004) The glutathione system as a stress marker in plant ecophysiology: is a stress-response concept valid? J Exp Bot 55:1955–1962
Wheeler GL, Jones MA, Smirnoff N (1998) The biosynthetic pathway of vitamin C in higher plants. Nature 393:365–369
Wolucka BA, Van Montagu M (2003) GDP-mannose 3′, 5′-epimerase forms GDP-l-gulose, a putative intermediate for the de novo biosynthesis of vitamin C in plants. J Biol Chem 278:47483–47490
Wolucka BA, Van Montagu M (2007) The VTC2 cycle and the de novo biosynthesis pathways for vitamin C in plants: an opinion. Phytochemistry 68:2602–2613
Xu Q, Chen LL, Ruan X et al (2013) The draft genome of sweet orange (Citrus sinensis). Nat Genet 45:59–66
Yabuta Y, Yoshimura K, Takeda T, Shigeoka S (2000) Molecular characterization of tobacco mitochondrial l-galactono-γ-lactone dehydrogenase and its expression in Escherichia coli. Plant Cell Physiol 41:666–675
Yabuta Y, Mieda T, Rapolu M, Nakamura A, Motoki T, Maruta T, Yoshimura K, Ishikawa T, Shigeoka S (2007) Light regulation of ascorbate biosynthesis is dependent on the photosynthetic electron transport chain but independent of sugars in Arabidopsis. J Exp Bot 58:2661–2671
Yang X, Xie J, Wang F, Zhong J, Liu Y, Li G, Peng S (2011) Comparison of ascorbate metabolism in fruits of two citrus species with obvious difference in ascorbate content in pulp. J Plant Physiol 168:2196–2205
Yang X-Y, Wang F–F, Teixeira da Silva JA, Zhong J, Liu Y-Z, Peng S-A (2013) Branch girdling at fruit green mature stage affects fruit ascorbic acid contents and expression of genes involved in L-galactose pathway in citrus. N Z J Crop Hort Sci. doi:10.1080/01140671.2012.724429
Zhang Y, Li H, Shu W, Zhang C, Zhang W, Ye Z (2011) Suppressed expression of ascorbate oxidase gene promotes ascorbic acid accumulation in tomato fruit. Plant Mol Biol Rep 29:638–645
Acknowledgments
This work was supported by research grants FP7-PEOPLE-2011-CIG-2011-303652 (Marie Curie Actions, European Union), Proyectos de I + D para Grupos de Investigación Emergentes GV/2012/036 (Generalitat Valenciana, Spain). Enriqueta Alós was recipient of a JAE-Doc (CSIC) post-doctoral contract which is co-funded by Fondo Social Europeo de Desarrollo Regional (FEDER). Financial support by PROMETEO2010/010 (Generalitat Valenciana, Spain), Fun-C-Food (CSD2007-0063C, Ministerio de Ciencia e Innovación, Spain) and AGL-2009-11558 and AGL-2012-34573 (Ministerio Economía y Competitividad, Spain) are also acknowledged. We thank Dr. Luis Navarro (IVIA, Moncada, Valencia) for the use of the Citrus Germplasm Bank and Dr. Jose Vicente Gil (Universitat de Valencia; IATA-CSIC) for allowing the use of the HPLC equipment. We also gratefully acknowledge the assistance of Arancha Gurrea, Joanna Lado and Amparo Beneyto.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
425_2014_2044_MOESM2_ESM.pptx
Supplementary Figure S1 Changes in fruit diameter, external color (a/b ratio) and maturity index of the juice (Total soluble solids/total acidity) in fruits of Satsuma Owari (white circles) and Washington Navel (black circles) during fruit development and ripening. Data of fruit diameter and external color are mean ± SD of 20 replicates, and those of juice maturity index are mean ± SD of 3 replicates. JN, JL, A, S, O, N, D indicate June, July, August, September, October, November and December, respectively. (PPTX 72 kb)
Rights and permissions
About this article
Cite this article
Alós, E., Rodrigo, M.J. & Zacarías, L. Differential transcriptional regulation of l-ascorbic acid content in peel and pulp of citrus fruits during development and maturation. Planta 239, 1113–1128 (2014). https://doi.org/10.1007/s00425-014-2044-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-014-2044-z