Abstract
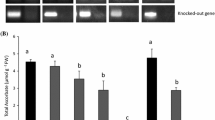
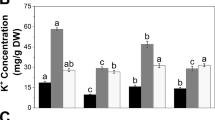
Effects of zinc (Zn) deficiency on shoot metabolites were investigated in contrasting rice (Oryza sativa L.) genotypes with special focus on ascorbic acid (AsA) biosynthesis, recycling, and catabolism. The genotypes IR74 (sensitive) and RIL46 (tolerant) were subjected to −Zn and control treatments for 3 weeks, and samples were taken at three different stages representing the pre-stress phase, emergence of visible stress, and severe visible stress. The emergence of visible symptoms was paralleled by an increase in lipid peroxidation and a decrease in AsA concentration in the sensitive, but not in the tolerant genotype. The tolerant RIL46 showed enhanced transcript levels of several genes involved in the mannose/l-galactose pathway to AsA biosynthesis, and significant up-regulation of a gene involved in the putative alternative myo-inositol pathway under low Zn stress. The level of most AsA precursors was negatively affected by Zn deficiency, but RIL46 had a constitutively higher level of non-phosphorylated precursors. Products of AsA catabolism such as oxalate and threonate did not accumulate in either genotype, suggesting that AsA degradation did not contribute to the stress-induced decline of the AsA pool in IR74. Further factors possibly contributing to tolerance in RIL46 included an almost fivefold higher proline level under −Zn stress and significantly higher trehalose content. The implications of these compounds in AsA metabolism and Zn efficiency thus deserve further attention.





Similar content being viewed by others
Abbreviations
- AsA:
-
Ascorbic acid
- DHA:
-
Dehydroascorbic acid
- DHAR:
-
Dehydroascorbic acid reductase
- MDA:
-
Malondialdehyde
- MDHA:
-
Monodehydroascorbic acid
- MDHAR:
-
Monodehydroascorbic acid reductase
- MIOX:
-
Myo-inositol oxygenase
- PMI:
-
Phosphomannose isomerase
- ROS:
-
Reactive oxygen species
- QTL:
-
Quantitative trait locus
- Zn:
-
Zinc
References
Agius F, Gonzalez-Lamothe R, Caballero JL, Munoz-Blanco J, Botella MA, Valpuesta V (2003) Engineering increased vitamin C levels in plants by overexpression of a d-galacturonic acid reductase. Nat Biotechnol 21:177–181
Ahkami AH, Lischewski S, Haensch K-T, Porfirova S, Hofmann J, Rolletschek H, Melzer M, Franken P, Hause B, Druege U, Hajirezaei MR (2009) Molecular physiology of adventitious root formation in Petunia hybrida cuttings: involvement of wound response and primary metabolism. New Phytol 181:613–625
Alloway BJ (2004) Zinc in soils and crop nutrition. International Fertilizer Industry Association, Paris
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399
Arnold T, Kirk GJD, Wissuwa M, Frei M, Zhao F-J, Mason TFD, Weiss DJ (2010) Evidence for the mechanisms of zinc uptake by rice using isotope fractionation. Plant Cell Environ 33:370–381
Ashraf M, Foolad MR (2007) Roles of glycine betaine and proline in improving plant abiotic stress resistance. Environ Exp Bot 59:206–216
Avonce N, Leyman B, Mascorro-Gallardo JO, Van Dijck P, Thevelein JM, Iturriaga G (2004) The Arabidopsis trehalose-6-P synthase AtTPS1 gene is a regulator of glucose, abscisic acid, and stress signaling. Plant Physiol 136:3649–3659
Bartoli CG, Guiamet JJ, Kiddle G, Pastori GM, Di Cagno R, Theodoulou FL, Foyer CH (2005) Ascorbate content of wheat leaves is not determined by maximal l-galactono-1,4-lactone dehydrogenase (GalLDH) activity under drought stress. Plant Cell Environ 28:1073–1081
Broadley MR, White PJ, Hammond JP, Zelko I, Lux A (2007) Zinc in plants. New Phytol 173:677–702
Cakmak I, Marschner H (1988) Enhanced superoxide radical production in roots of zinc deficient plants. J Exp Bot 39:1449–1460
Cakmak I, Marschner H, Bangerth F (1989) Effect of zinc nutritional status on growth, protein metabolism and levels of indole-3-acetic acid and other phytohormones in bean (Phaseolus vulgaris L.). J Exp Bot 40:405–412
Chen Z, Young TE, Ling J, Chang SC, Gallie DR (2003) Increasing vitamin C content of plants through enhanced ascorbate recycling. Proc Natl Acad Sci USA 100:3525–3530
Conklin PL (2001) Recent advances in the role and biosynthesis of ascorbic acid in plants. Plant Cell Environ 24:383–394
Davey MW, Gilot C, Persiau G, Ostergaard J, Han Y, Bauw GC, Van Montagu MC (1999) Ascorbate biosynthesis in Arabidopsis cell suspension culture. Plant Physiol 121:535–543
DeBolt S, Hardie J, Tyerman S, Ford CM (2004) Composition and synthesis of raphide crystals and druse crystals in berries of Vitis vinifera L. cv. Cabernet Sauvignon: ascorbic acid as precursor for both oxalic and tartaric acids as revealed by radiolabelling studies. Aust J Grape Wine R 10:134–142
Dobermann A, Fairhurst T (2000) Rice: nutrient disorders & nutrient management. International Rice Research Institute, Los Banos
Duan J, Zhang M, Zhang H, Xiong H, Liu P, Ali J, Li J, Li Z (2012) OsMIOX, a myo-inositol oxygenase gene, improves drought tolerance through scavenging of reactive oxygen species in rice (Oryza sativa L.). Plant Sci 196:143–151
Endres S, Tenhaken R (2009) Myoinositol oxygenase controls the level of myoinositol in Arabidopsis, but does not increase ascorbic acid. Plant Physiol 149:1042–1049
Frei M, Tanaka JP, Chen CP, Wissuwa M (2010a) Mechanisms of ozone tolerance in rice: characterization of two QTLs affecting leaf bronzing by gene expression profiling and biochemical analyses. J Exp Bot 61:1405–1417
Frei M, Wang Y, Ismail AM, Wissuwa M (2010b) Biochemical factors conferring shoot tolerance to oxidative stress in rice grown in low zinc soil. Funct Plant Biol 37:74–84
French D (1954) The raffinose family of oligosaccharides. Adv Carbohyd Chem Bi 9:149–184
Garg AK, Kim JK, Owens TG, Ranwala AP, Do Choi Y, Kochian LV, Wu RJ (2002) Trehalose accumulation in rice plants confers high tolerance levels to different abiotic stresses. Proc Natl Acad Sci USA 99:15898–15903
Goddijn OJM, van Dun K (1999) Trehalose metabolism in plants. Trends Plant Sci 4:315–319
Gracy RW, Noltmann EA (1968a) Studies on phosphomannose isomerase. I. Isolation homogeneity measurements and determination of some physical properties. J Biol Chem 243:3161–3168
Gracy RW, Noltmann EA (1968b) Studies on phosphomannose isomerase: II. Characterization as a zinc metalloenzyme. J Biol Chem 243:4109–4116
Green MA, Fry SC (2005) Vitamin C degradation in plant cells via enzymatic hydrolysis of 4-O-oxalyl-l-threonate. Nature 433:83–87
Guo ZF, Tan HQ, Zhu ZH, Lu SY, Zhou BY (2005) Effect of intermediates on ascorbic acid and oxalate biosynthesis of rice and in relation to its stress resistance. Plant Physiol Bioch 43:955–962
Hacisalihoglu G, Kochian LV (2003) How do some plants tolerate low levels of soil zinc? Mechanisms of zinc efficiency in crop plants. New Phytol 159:341–350
Hancock RD, Viola R (2005) Biosynthesis and catabolism of l-ascorbic acid in plants. Crit Rev Plant Sci 24:167–188
Hodges DM, DeLong JM, Forney CF, Prange RK (1999) Improving the thiobarbituric acid-reactive-substances assay for estimating lipid peroxidation in plant tissues containing anthocyanin and other interfering compounds. Planta 207:604–611
Hoffland E, Wei C, Wissuwa M (2006) Organic anion exudation by lowland rice (Oryza sativa L.) at zinc and phosphorus deficiency. Plant Soil 283:155–162
Hossain MA, Asada K (1984) Purification of dehydroascorbate reductase from spinch and its characterization as a thiol enzyme. Plant Cell Physiol 25:85–92
Hossain B, Hirata N, Nagatomo Y, Akashi R, Takaki H (1997) Internal zinc accumulation is correlated with increased growth in rice suspension culture. J Plant Growth Regul 16:239–243
Ishikawa T, Dowdle J, Smirnoff N (2006) Progress in manipulating ascorbic acid biosynthesis and accumulation in plants. Physiol Plant 126:343–355
Jo Y, Hyun TK (2011) Genome-wide identification of antioxidant component biosynthetic enzymes: comprehensive analysis of ascorbic acid and tocochromanols biosynthetic genes in rice. Comput Biol Chem 35:261–268
Kitagishi K, Obata H (1986) Effects of zinc deficiency on the nitrogen-metabolism of meristematic tissue of rice plants with reference to protein synthesis. Soil Sci Plant Nutr 32:397–405
Linster CL, Clarke SG (2008) l-Ascorbate biosynthesis in higher plants: the role of VTC2. Trends Plant Sci 13:567–573
Loewus FA, Kelly S (1961) Identity of l-ascorbic acid formed by d-glucose by strawberry (Fragaria). Nature 191:1059–1061
Lorence A, Chevone BI, Mendes P, Nessler CL (2004) myo-Inositol oxygenase offers a possible entry point into plant ascorbate biosynthesis. Plant Physiol 134:1200–1205
Luo Y, Li W-M, Wang W (2008) Trehalose: protector of antioxidant enzymes or reactive oxygen species scavenger under heat stress? Environ Exp Bot 63:378–384
Maruta T, Yonemitsu M, Yabuta Y, Tamoi M, Ishikawa T, Shigeoka S (2008) Arabidopsis phosphomannose isomerase 1, but not phosphomannose isomerase 2 is essential for ascorbic acid biosynthesis. J Biol Chem 283:28842–28851
Matysik J, Alia, Bhalu B, Mohanty P (2002) Molecular mechanisms of quenching of reactive oxygen species by proline under stress in plants. Curr Sci India 82:525–532
Molinari HBC, Marur CJ, Daros E, De Campos MKF, De Carvalho JFRP, Pereira LFP, Vieira LGE (2007) Evaluation of the stress-inducible production of proline in transgenic sugarcane (Saccharum spp.): osmotic adjustment, chlorophyll fluorescence and oxidative stress. Physiol Plant 130:218–229
Naqvi S, Zhu C, Farre G, Ramessar K, Bassie L, Breitenbach J, Perez Conesa D, Ros G, Sandmann G, Capell T, Christou P (2009) Transgenic multivitamin corn through biofortification of endosperm with three vitamins representing three distinct metabolic pathways. Proc Natl Acad Sci USA 106:7762–7767
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49:249–279
Parsons HT, Yasmin T, Fry SC (2011) Alternative pathways of dehydroascorbic acid degradation in vitro and in plant cell cultures: novel insights into vitamin C catabolism. Biochem J 440:375–383
Patel RK, Jain M (2011) PlantRGS: a web server for the identification of most suitable candidate reference genes for quantitative gene expression studies in plants. DNA Res 18:463–470
Possingham JV (1956) The effect of mineral nutrition on the content of free amino acids and amides in tomato plants. Aust J Biol Sci 9:539–551
Rolland F, Baena-Gonzalez E, Sheen J (2006) Sugar sensing and signaling in plants: conserved and novel mechanisms. Annu Rev Plant Biol 57:675–709
Rontein D, Basset G, Hanson AD (2002) Metabolic engineering of osmoprotectant accumulation in plants. Metab Eng 4:49–56
Rose MT, Rose TJ, Pariasca-Tanaka J, Widodo, Wissuwa M (2011) Revisiting the role of organic acids in the bicarbonate tolerance of zinc-efficient rice genotypes. Funct Plant Biol 38:493–504
Rose MT, Rose TJ, Pariasca-Tanaka J, Yoshihashi T, Neuweger H, Goesmann A, Frei M, Wissuwa M (2012) Root metabolic response of rice (Oryza sativa L.) genotypes with contrasting tolerance to zinc deficiency and bicarbonate excess. Planta 236:959–973
Smirnoff N (1996) Botanical briefing: the function and metabolism of ascorbic acid in plants. Ann Bot 78:661–669
Smirnoff N, Wheeler GL (2000) Ascorbic acid in plants: biosynthesis and function. Crit Rev Plant Sci 19:267–290
Szekely G, Abraham E, Cseplo A, Rigo G, Zsigmond L, Csiszar J, Ayaydin F, Strizhov N, Jasik J, Schmelzer E, Koncz C, Szabados L (2008) Duplicated P5CS genes of Arabidopsis play distinct roles in stress regulation and developmental control of proline biosynthesis. Plant J 53:11–28
Tausz M, Sircelj H, Grill D (2004) The glutathione system as a stress marker in plant ecophysiology: is a stress-response concept valid? J Exp Bot 55:1955–1962
Tripathi B, Gaur J (2004) Relationship between copper-and zinc-induced oxidative stress and proline accumulation in Scenedesmus sp. Planta 219:397–404
Ueda Y, Wu LB, Frei M (2013) A critical comparison of two high throughput ascorbate analyses methods for plant samples. Plant Physiol Bioch 70:418–423
Wheeler GL, Jones MA, Smirnoff N (1998) The biosynthetic pathway of vitamin C in higher plants. Nature 393:365–369
Widodo B, Broadley MR, Rose T, Frei M, Pariasca-Tanaka J, Yoshihashi T, Thomson M, Hammond JP, Aprile A, Close TJ, Ismail AM, Wissuwa M (2010) Response to zinc deficiency of two rice lines with contrasting tolerance is determined by root growth maintenance and organic acid exudation rates, and not by zinc-transporter activity. New Phytol 186:400–414
Wingler A, Wingler (2002) The function of trehalose biosynthesis in plants. Phytochemistry 60:437–440
Wissuwa M, Ismail AM, Yanagihara S (2006) Effects of zinc deficiency on rice growth and genetic factors contributing to tolerance. Plant Physiol 142:731–741
Wolucka BA, Van Montagu M (2003) GDP-mannose 3′, 5′-epimerase forms GDP-l-gulose, a putative intermediate for the de novo biosynthesis of vitamin C in plants. J Biol Chem 278:47483–47490
Acknowledgments
This study was partly funded by Deutsche Forschungsgemeinschaft (FR2952/1-1).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Höller, S., Hajirezaei, MR., von Wirén, N. et al. Ascorbate metabolism in rice genotypes differing in zinc efficiency. Planta 239, 367–379 (2014). https://doi.org/10.1007/s00425-013-1978-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-013-1978-x