Abstract
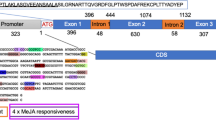
NO and H2O2 are important biological messengers in plants. They are formed during xylem differentiation in Zinnia elegans and apparently play important roles during the xylogenesis. To ascertain the responsiveness of the Z. elegans peroxidase (ZePrx) to these endogenous signals, the effects of NO and H2O2 on ZePrx were studied. The results showed that ZePrx is up-regulated by NO and H2O2, as confirmed by RT-qPCR, and that its promoter contains multiple copies of all the putative cis-elements (ACGT box, OCS box, OPAQ box, L1BX, MYCL box and W box) known to confer regulation by NO and H2O2. Like other OCS elements, the OCS element of ZePrx contains the sequence TACG that is recognized by OBF5, a highly conserved bZIP transcription factor, and the 10 bp sequence, ACAaTTTTGG, which is recognized by OBP1, a Dof domain protein that binds down-stream the OCS element. Furthermore, the ZePrx OCS element is flanked by two CCAAT-like boxes, and encloses one auxin-responsive ARFAT element and two GA3-responsive Pyr boxes. Results also showed that ZePrx may be described as the first protein to be up-regulated by NO and H2O2, whose mRNA contains several short-longevity conferring elements, such as a downstream (DST) sequence analogous to the DSTs contained in the highly unstable SAUR transcripts. The presence of these regulatory elements strongly suggests that ZePrx is finely regulated, as one may expect from an enzyme that catalyzes the last irreversible step of the formation of lignins, the major irreversible sink for the photosynthetically fixed CO2.







Similar content being viewed by others
Abbreviations
- ARFs:
-
Auxin-response factors
- AuxREs:
-
Auxin-responsive cis-elements
- DST:
-
Downstream sequence
- GSNO:
-
S-nitrosoglutathione
- IEF:
-
Isoelectric focusing
- MAPK:
-
Mitogen-activated protein kinase
- NO:
-
Nitric oxide
- PTIO:
-
2-Phenyl-4,4,5,5-tetramethylimidazoline-1-oxyl-3 oxide
- OCS box:
-
Octopine synthase box
- rboh:
-
Respiratory burst oxidase homologue, NADPH-oxidase
- SAUR:
-
Small auxin-up RNA
- TFs:
-
Transcription factors
- ZePrx:
-
Basic peroxidase isoenzyme from Zinnia elegans
References
Abe H, Yamaguchi Shinozaki K, Urao T, Iwasaki T, Hosokawa D, Shinozaki K (1997) Role of Arabidopsis MYC and MYB homologs in drought- and abscisic acid-regulated gene expression. Plant Cell 9:1859–1868
Abe M, Takahashi T, Komeda Y (2001) Identification of a cis-regulatory element for L1 layer-specific gene expression, which is targeted by an L1-specific homeodomain protein. Plant J 26:487–494
Alston TA, Porter DJT, Bright HJ (1985) Generation of nitric oxide by enzymatic oxidation of N-hydroxy-N-nitrosamines. J Biol Chem 260:4069–4074
Besson-Bard A, Pugin A, Wendehenne D (2008) New insights into nitric oxide signaling in plants. Annu Rev Plant Biol 59:21–39
Capone R, Tiwari BS, Levine A (2004) Rapid transmission of oxidative and nitrosative stress signals from roots to shoots in Arabidopsis. Plant Physiol Biochem 42:425–428
Cercos M, Gómez-Cadenas A, Ho THD (1999) Hormonal regulation of a cysteine proteinase gene, EPB-1, in barley aleurone layers: cis- and trans-acting elements involved in the co-ordinated gene expression regulated by gibberellins and abscisic acid. Plant J 19:107–118
Clark D, Durner J, Navarre DA, Klessig DF (2000) Nitric oxide inhibition of tobacco catalase and ascorbate peroxidase. Mol Plant Microbe Interact 13:1380–1384
Corpas FJ, Barroso JB, Carreras A, Valderrama R, Palma JM, Leon AM, Sandalio LM, del Río LA (2006) Constitutive arginine-dependent nitric oxide synthase activity in different organs of pea seedlings during plant development. Planta 224:246–254
Chen WQ, Chao G, Singh KB (1996) The promoter of a H2O2-inducible, Arabidopsis glutathione S-transferase gene contains closely linked OBF- and OBP1-binding sites. Plant J 10:955–966
Chen WQ, Singh KB (1999) The auxin, hydrogen peroxide and salicylic acid induced expression of the Arabidopsis GST6 promoter is mediated in part by an ocs element. Plant J 19:667–677
de Pinto MC, Paradiso A, Leonetti P, De Gara L (2006) Hydrogen peroxide, nitric oxide and cytosolic ascorbate peroxidase at the crossroad between defence and cell death. Plant J 48:784–795
Delledonne M (2005) NO news is good news for plants. Curr Opin Plant Biol 8:390–396
Demura T, Fukuda H (2007) Transcriptional regulation in wood formation. Trends Plant Sci 12:64–70
Demura T, Tashiro G, Horiguchi G, Kishimoto N, Kubo M, Matsuoka N, Minami A, Nagata-Hiwatashi M, Nakamura K, Okamura Y, Sassa N, Suzuki S, Yazaki J, Kikuchi S, Fukuda H (2002) Visualization by comprehensive microarray analysis of gene expression programs during transdifferentiation of mesophyll cells into xylem cells. Proc Natl Acad Sci USA 99:15794–15799
Drochioiu G (2002) Fast and highly selective determination of cyanide with 2,2-dihydroxy-1,3-indanedione. Talanta 56:1163–1165
Edwards D, Murray JAH, Smith AG (1998) Multiple genes encoding the conserved CCAAT-box transcription factor complex are expressed in Arabidopsis. Plant Physiol 117:1015–1022
Ellis JG, Tokuhisa JG, Llewellyn DJ, Bouchez D, Singh K, Dennis ES, Peacock WJ (1993) Does the ocs-element occur as a functional component of the promoters of plant genes. Plant J 4:433–443
Escamilla-Trevino LL, Shen H, Uppalapati SR, Ray T, Tang Y, Hernandez T, Yin Y, Xu Y, Dixon RA (2010) Switchgrass (Panicum virgatum) possesses a divergent family of cinnamoyl CoA reductases with distinct biochemical properties. New Phytol 185:143–155
Floryszak-Wieczorek J, Milczarek G, Arasimowicz M, Ciszewski A (2006) Do nitric oxide donors mimic endogenous NO-related response in plants? Planta 224:1363–1372
Gabaldón C, Gómez Ros LV, López Núñez-Flores MJ, Esteban Carrasco A, Ros Barceló A (2007) Post-translational modifications of the basic peroxidase isoenzyme from Zinnia elegans. Plant Mol Biol 65:43–61
Gabaldón C, Gómez Ros LV, Pedreño MA, Ros Barceló A (2005a) Nitric oxide production by the differentiating xylem of Zinnia elegans. New Phytol 165:121–130
Gabaldón C, López-Serrano M, Pedreño MA, Ros Barceló A (2005b) Cloning and molecular characterization of the basic peroxidase isoenzyme from Zinnia elegans, an enzyme involved in lignin biosynthesis. Plant Physiol 139:1138–1154
Goldstein S, Russo A, Samuni A (2003) Reactions of PTIO and carboxy-PTIO with ·NO, ·NO2, and O ·−2 . J Biol Chem 278:50949–50955
Gómez Ros LV, Paradiso A, Gabaldón C, Pedreño MA, de Gara L, Ros Barceló A (2006) Two distinct cell sources of H2O2 in the lignifying Zinnia elegans cell culture system. Protoplasma 227:175–183
Gutiérrez J, López Núñez-Flores MJ, Gómez Ros LV, Uzal EN, Esteban Carrasco A, Díaz J, Sottomayor M, Cuello J, Ros Barceló A (2009) Hormonal regulation of the basic peroxidase isoenzyme from Zinnia elegans. Planta 230:767–778
Higo K, Ugawa Y, Iwamoto M, Korenaga T (1999) Plant cis-acting regulatory DNA elements (PLACE) database: 1999. Nucleic Acids Res 27:297–300
Hu RB, Fan CM, Li HY, Zhang QZ, Fu YF (2009) Evaluation of putative reference genes for gene expression normalization in soybean by quantitative real-time RT-PCR. BMC Mol Biol 10:93
Kawaoka A, Kaothien P, Yoshida K, Endo S, Yamada K, Ebinuma H (2000) Functional analysis of tobacco LIM protein Ntlim1 involved in lignin biosynthesis. Plant J 22:289–301
Kim HS, Yu Y, Snesrud EC, Moy LP, Linford LD, Haas BJ, Nierman WC, Quackenbush J (2005) Transcriptional divergence of the duplicated oxidative stress-responsive genes in the Arabidopsis genome. Plant J 41:212–220
Kim SY, Chung HJ, Thomas TL (1997) Isolation of a novel class of bZIP transcription factors that interact with ABA-responsive and embryo-specification elements in the Dc3 promoter using a modified yeast one-hybrid system. Plant J 11:1237–1251
Ko JH, Han KH (2004) Arabidopsis whole-transcriptome profiling defines the features of coordinated regulations that occur during secondary growth. Plant Mol Biol 55:433–453
Kubo M, Udagawa M, Nishikubo N, Horiguchi G, Yamaguchi M, Ito J, Mimura T, Fukuda H, Demura T (2005) Transcription switches for protoxylem and metaxylem vessel formation. Gene Dev 19:1855–1860
Lebel E, Heifetz P, Thorne L, Uknes S, Ryals J, Ward E (1998) Functional analysis of regulatory sequences controlling PR-1 gene expression in Arabidopsis. Plant J 16:223–233
López Núñez-Flores MJ, Gutiérrez J, Gómez Ros LV, Uzal EN, Sottomayor M, Ros Barceló A (2010) Downregulation of the basic peroxidase isoenzyme from Zinnia elegans by gibberellic acid. J Integr Plant Biol 52:244–251
McClure BA, Hagen G, Brown CS, Gee MA, Guilfoyle TJ (1989) Transcription, organization, and sequence of an auxin-regulated gene-cluster in soybean. Plant Cell 1:229–239
Meng Z, King PH, Nabors LB, Jackson NL, Chen CY, Emanuel PD, Blume SW (2005) The ELAV RNA-stability factor HuR binds the 5′-untranslated region of the human IGF-IR transcript and differentially represses cap-dependent and IRES-mediated translation. Nucleic Acids Res 33:2962–2979
Nakanomyo I, Kost B, Chua NH, Fukuda H (2002) Preferential and asymmetrical accumulation of a Rac small GTPase mRNA in differentiating xylem cells of Zinnia elegans. Plant Cell Physiol 43:1484–1492
Narsai R, Howell KA, Millar AH, O′Toole N, Small I, Whelan J (2007) Genome-wide analysis of mRNA decay rates and their determinants in Arabidopsis thaliana. Plant Cell 19:3418–3436
Navarre DA, Wendehenne D, Durner J, Noad R, Klessig DF (2000) Nitric oxide modulates the activity of tobacco aconitase. Plant Physiol 122:573–582
Neill SJ, Desikan R, Clarke A, Hurst RD, Hancock JT (2002) Hydrogen peroxide and nitric oxide as signalling molecules in plants. J Exp Bot 53:1237–1247
Newman TC, Ohmetakagi M, Taylor CB, Green PJ (1993) DST sequences, highly conserved among plant SAUR genes, target reporter transcripts for rapid decay in tobacco. Plant Cell 5:701–714
Noriega GO, Yannarelli GG, Balestrasse KB, Batlle A, Tomaro ML (2007) The effect of nitric oxide on heme oxygenase gene expression in soybean leaves. Planta 226:1155–1163
Østergaard L, Pedersen AG, Jespersen HM, Brunak S, Welinder KG (1998) Computational analyses and annotations of the Arabidopsis peroxidase gene family. FEBS Lett 433:98–102
Palmieri MC, Sell S, Huang X, Scherf M, Werner T, Durner J, Lindermayr C (2008) Nitric oxide-responsive genes and promoters in Arabidopsis thaliana: a bioinformatics approach. J Exp Bot 59:177–186
Parani M, Rudrabhatla S, Myers R, Weirich H, Smith B, Leaman DW, Goldman SL (2004) Microarray analysis of nitric oxide responsive transcripts in Arabidopsis. Plant Biotech J 2:359–366
Passardi F, Penel C, Dunand C (2004) Performing the paradoxical: how plant peroxidases modify the cell wall. Trends Plant Sci 9:534–540
Patzlaff A, McInnis S, Courtenay A, Surman C, Newman LJ, Smith C, Bevan MW, Mansfield S, Whetten RW, Sederoff RR, Campbell MM (2003) Characterisation of a pine MYB that regulates lignification. Plant J 36:743–754
Pesquet E, Ranocha P, Legay S, Digonnet C, Barbier O, Pichon M, Goffner D (2005) Novel markers of xylogenesis in Zinnia are differentially regulated by auxin and cytokinin. Plant Physiol 139:1821–1839
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Raes J, Rohde A, Christensen JH, Van de Peer Y, Boerjan W (2003) Genome-wide characterization of the lignification toolbox in Arabidopsis. Plant Physiol 133:1051–1071
Rodríguez-Serrano M, Romero-Puertas MC, Pazmino DM, Testillano PS, Risueno MC, del Río LA, Sandalio LM (2009) Cellular response of pea plants to cadmium toxicity: cross talk between reactive oxygen species, nitric oxide, and calcium. Plant Physiol 150:229–243
Ros Barceló A (1998) The generation of H2O2 in the xylem of Zinnia elegans is mediated by an NADPH-oxidase-like enzyme. Planta 207:207–216
Ros Barceló A (2005) Xylem parenchyma cells deliver the H2O2 necessary for lignification in differentiating xylem vessels. Planta 220:747–756
Ros Barceló A, Pomar F, Ferrer MA, Martínez P, Ballesta MC, Pedreño MA (2002a) In situ characterization of a NO-sensitive peroxidase in the lignifying xylem of Zinnia elegans. Physiol Plant 114:33–40
Ros Barceló A, Pomar F, López-Serrano M, Martínez P, Pedreño MA (2002b) Developmental regulation of the H2O2-producing system and of a basic peroxidase isoenzyme in the Zinnia elegans lignifying xylem. Plant Physiol Biochem 40:325–332
Rushton PJ, Somssich IE, Ringler P, Shen QXJ (2010) WRKY transcription factors. Trends Plant Sci 15:247–258
Shishido SM, de Oliveira MG (2001) Photosensitivity of aqueous sodium nitroprusside solutions: nitric oxide release versus cyanide toxicity. Prog React Kinet Mech 26:239–261
Srivastava V, Schinkel H, Witzell J, Hertzberg M, Torp M, Srivastava MK, Karpinska B, Melzer M, Wingsle G (2007) Downregulation of high-isoelectric-point extracellular superoxide dismutase mediates alterations in the metabolism of reactive oxygen species and developmental disturbances in hybrid aspen. Plant J 49:135–148
Sullivan ML, Green PJ (1996) Mutational analysis of the DST element in tobacco cells and transgenic plants: identification of residues critical for mRNA instability. RNA 2:308–315
Ulmasov T, Hagen G, Guilfoyle TJ (1997) ARF1, a transcription factor that binds to auxin response elements. Science 276:1865–1868
Vranova E, Inze D, Van Breusegem F (2002) Signal transduction during oxidative stress. J Exp Bot 53:1227–1236
Welinder KG, Justesen AF, Kjaersgard IVH, Jensen RB, Rasmussen SK, Jespersen HM, Duroux L (2002) Structural diversity and transcription of class III peroxidases from Arabidopsis thaliana. Eur J Biochem 269:6063–6081
Weng JK, Akiyama T, Bonawitz ND, Li X, Ralph J, Chapple C (2010) Convergent evolution of syringyl lignin biosynthesis via distinct pathways in the lycophyte Selaginella and flowering plants. Plant Cell 22:1033–1045
Xu NF, Hagen G, Guilfoyle T (1997) Multiple auxin response modules in the soybean SAUR 15A promoter. Plant Sci 126:193–201
Yanagisawa S (2004) Dof domain proteins: plant-specific transcription factors associated with diverse phenomena unique to plants. Plant Cell Physiol 45:386–391
Yonetani T, Yamamoto H, Erman JE, Leigh JS, Reed GH (1972) Electromagnetic properties of hemoproteins. J Biol Chem 247:2447–2455
Yoshida S, Iwamoto K, Demura T, Fukuda H (2009) Comprehensive analysis of the regulatory roles of auxin in early transdifferentiation into xylem cells. Plant Mol Biol 70:457–469
Zhong R, Lee C, Ye Z-H (2010) Evolutionary conservation of the transcriptional network regulating secondary cell wall biosynthesis. Trends Plant Sci 15:625–632
Acknowledgments
We thank Prof. Alberto Esteban Carrasco for contributing to bioinformatics analysis, drafting and critical reading of the manuscript. This work was supported by grants from the MEC (BFU2006-11577/BFI and BFU2009-08151)-FEDER and CARM (08610/PI/08). Joaquín Herrero and Jorge Gutiérrez hold fellowships (FPU and FPI) from the MEC/MCYT (Spain), respectively.
Author information
Authors and Affiliations
Corresponding author
Additional information
In memoriam of Prof. Alfonso Ros Barceló, be able to continue with your work is an honor.
Rights and permissions
About this article
Cite this article
Gómez-Ros, L.V., Gabaldón, C., López Núñez-Flores, M.J. et al. The promoter region of the Zinnia elegans basic peroxidase isoenzyme gene contains cis-elements responsive to nitric oxide and hydrogen peroxide. Planta 236, 327–342 (2012). https://doi.org/10.1007/s00425-012-1604-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-012-1604-3