Abstract
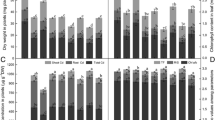
Heme oxygenase (HO, EC 1.14.99.3) catalyzes the oxidative conversion of heme to biliverdin IXα with the concomitant release of carbon monoxide and iron. Recently, HO has been involved in the protection against oxidative stress in plants. The fact that nitric oxide (NO), an endogenous signaling molecule in animals and plants mediates responses to abiotic and biotic stresses, prompted us to study whether this molecule could modulate HO-1 gene transcription. To fulfill this objective leaves of soybean (Glycine max L.) plants were stimulated with Cd, employing an acute intoxication model. Cadmium caused dehydration, chlorophyll loss and ion leakage. Semi-quantitative RT-PCR analysis showed no augmentation of HO-1 transcript levels with respect to controls. Pretreatment with 100 μM sodium nitroprussiate (SNP), a well-known NO donor, prevented the effects caused by Cd. When the HO-1 mRNA levels were analyzed, a significant augmentation (54%) was observed with respect to Cd-treated plants. On the other hand, 50 or 300 μM SNP did not fully prevent the effects elicited by Cd. When HO-1 transcript levels were analyzed, no significant enhancement or a down-regulation was observed. The potassium salt of 2-(4-carboxylphenyl)-4,4,5,5-tetramethylimidazoline-1-oxyl-3-oxide (cPTIO), a specific NO scavenger, arrested NO-mediated protective effects against to Cd-induced oxidative damage. These data provide an understanding of one of the possible roles that NO can play against an oxidative insult. NO is cytoprotective depending on its concentration, and it was further demonstrated that this protection could be, at least in part, mediated by an enhancement of HO-1 mRNA, as it happens with genes associated with the antioxidant defense system.





Similar content being viewed by others
Abbreviations
- BV:
-
Biliverdin IXα
- cPTIO:
-
2-(4-Carboxyphenil)-4,4,5,5-tetramethylimidazoline-1-oxyl-3-oxide
- DAB:
-
3,3′-Diamino benzidine
- HO:
-
Heme oxygenase
- NBT:
-
Nitroblue tetrazolium
- NO:
-
Nitric oxide
- NOS:
-
Nitric oxide synthetase
- SNP:
-
Sodium nitroprussiate
References
Arnaud N, Murgia I, Boucherez J, Briat JF, Cellier F, Gaymard F (2006) An iron-induced nitric oxide burst precedes ubiquitin-dependent protein degradation for Arabidopsis AtFer1 ferritin gene expression. J Biol Chem 281:23579–23588
Balestrasse KB, Gardey L, Gallego SM, Tomaro ML (2001) Response of antioxidative defense system in soybean nodules and roots subjected to cadmium stress. Aust J Plant Physiol 28:497–504
Balestrasse KB, Noriega GO, Batlle A, Tomaro ML (2005) Involvement of heme oxygenase as antioxidant defense in soybean nodules. Free Radic Res 39:145–151
Barroso JB, Corpas FJ, Carreras A, Rodriguez-Serrano M, Esteban FJ, Fernandez-Ocana A, Chaki M, Romero-Puertas MC, Valderrama R, Sandalio LM, del Rio LA (2006) Localization of S-nitrosoglutathione and expression of S-nitrosoglutathione reductase in pea plants under cadmium stress. J Exp Bot 57:1785–1793
Beligni NV, Lamattina L (2001) Nitric oxide: a non-traditional regulator of plant growth. Trends Plant Sci 6:508–509
Boveris A (1984) Oxygen radicals in biological systems. Methods Enzymol 105:429–435
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Clark D, Durner J, Navarre DA, Klessig DF (2000a) Nitric oxide inhibition of tobacco catalase and ascorbate peroxidase. Mol Plant Microbe Interact 13:1380–1384
Clark JE, Foresti R, Green CJ, Motterlini R (2000b) Dynamics of haem-oxygenase-1 expression and bilirubin production in cellular protection against oxidative stress. Biochem J 348:615–619
Colburn NH (1992) Gene regulation by active oxygen and other stress inducers. In: Spatz L, Bloom AD (eds) Biological consequences of oxidative stress. Oxford University Press, New York, pp 121–137
Crawford NM (2006) Mechanisms for nitric oxide synthesis in plants. J Exp Bot 57:471–478
Creissen G, Firmin J, Fryer M (1999) Elevated glutathione biosynthesis capacity in the chloroplast of transgenic tobacco plants paradoxically causes oxidative stress. Plant Cell 11:1277–1291
Gow AJ, Luchsinger BP, Pawloski JR, Singel DJ, Stamler JS (1999) The oxyhemoglobin reaction of nitric oxide. Proc Natl Acad Sci USA 96:9027–9032
Hoagland DR, Arnon DI (1950) The water culture method for growing plants without soil. Calif Agric Exp Stn Circ 347:1–32
Huang X, von Rad U, Durner J (2002) Nitric oxide induces the nitric oxide tolerant alternative oxidase in Arabidopsis suspension cells. Planta 215:914–923
Kikuchi G, Yoshida T, Noguchi M (2005) Heme oxygenase and heme degradation. Biochem Biophys Res Commun 338:558–567
Kim HJ, Tsoy I, Park MK, Lee YS, Lee JH, Seo HG, Chang KC (2006) Iron released by sodium nitroprusside contributes to heme oxygenase-1 induction via the cAMP-protein kinase A-mitogen activated protein kinase pathway in RAW 264.7 cells. Mol Pharmacol 69:1633–1640
Llesuy SF, Tomaro ML (1994) Heme oxygenase and oxidative stress. Evidence of involvement of bilirubin as physiological protector against oxidative damage. Biochim Biophys Acta 1223:9–14
Muramoto T, Tsurui N, Terry MJ, Yokota A, Kohchi T (2002) Expression and biochemical properties of a ferredoxin-dependent heme oxygenase required for phytochrome chromophore synthesis. Plant Physiol 130:1958–1966
Murgia I, De Pinto MC, Delledonne M, Soave C, De Gara L (2004) Comparative effects of various nitric oxide donors on ferritin regulation, programmed cell death, and cell redox state in plant cells. J Plant Physiol 161:777–783
Noriega GO, Tomaro ML, Batlle AMC (2003) Bilirubin is highly effective in preventing in vivo d-aminolevulinic acid-induced oxidative cell damage. Biochim Biophys Acta 1638:173–178
Noriega GO, Balestrasse KB, Batlle A, Tomaro ML (2004) Heme oxygenase exerts a protective role against oxidative stress in soybean leaves. Biochem Biophys Res Commun 323:1003–1008
Parani MR, Myers R, Wireich H, Smith B, Leaman DW, Goldman SL (2004) Microarray analysis of nitric oxide responsive transcripts in Arabidopsis. Plant Biotechnol J 2:359–366
Perazzolli M, Dominici P, Romero-Puertas MC, Zago E, Zeier J, Sonoda M, Lamb C, Delledonne M (2004) Arabidopsis non symbiotic hemoglobin AHb1 modulates nitric oxide bioactivity. Plant Cell 16:2785–2794
Reiter SW, Tyrrell RM (2000) The heme synthesis and degradation pathways: role in oxidant sensitivity. Heme oxygenase has both pro- and antioxidant properties. Free Radic Biol Med 28:289–309
Rodriguez-Serrano M, Romero-Puertas MC, Zabalza A, Corpas FJ, Gomez M, Del Rio LA, Sandalio LM (2006) Cadmium effect on oxidative metabolism of pea (Pisum sativum L.) roots. Imaging of reactive oxygen species and nitric oxide accumulation in vivo. Plant Cell Environ 29:1532–1544
Shi S, Wang G, Wang Y, Zhang L, Zhang L (2005) Protective effect of nitric oxide against oxidative stress under ultraviolet-B radiation. Nitric Oxide 13:1–9
Sies H (1993) Strategies of antioxidant defense. Eur J Biochem 215:213–219
Stocker R (1990) Induction of haem oxygenase as a defence against oxidative stress. Free Radic Res Commun 9:101–112
Tenhunen R, Marver HS, Schmid R (1968) The enzymatic conversion of heme to bilirubin by microsomal heme oxygenase. Proc Natl Acad Sci USA 61:748–755
Terry MJ, Linley PJ, Kochi T (2002) Making light of it: the role of plants heme oxygenases in phytochrome chromophore synthesis. Biochem Soc Trans 30:604–609
Tomaro ML, Batlle A (2002) Bilirubin: its role in cytoprotection against oxidative stress. Int J Biochem Cell Biol 34:216–220
Wagner GJ (1993) Accumulation of cadmium in crop plants and its consequences to human health. Adv Agron 51:173–212
Wendehenne D, Durner J, Klessig DF (2004) Nitric oxide: a new player in plant signaling and defence responses. Curr Opin Plant Biol 7:449–455
Wintermans J, de Mots A (1965) Spectrophotometric characteristics of chlorophylls a and b and their pheophytins in ethanol. Biochim Biophys Acta 109:448–453
Yannarelli GG, Noriega GO, Batlle A, Tomaro ML (2006) Heme oxygenase up-regulation in ultraviolet-B irradiated soybean plants involves reactive oxygen species. Planta 224:1154–1162
Zaninotto F, La Camera S, Polverari A, Delledonne M (2006) Cross talk between reactive nitrogen and oxygen species during the hypersensitive disease resistance response. Plant Physiol 141:379–383
Zemojtel T, Frohlich A, Palmieri MC, Kolanczyk M, Mikula I, Wyrwicz LS, Wanker EE, Mundlos S, Vingron M, Martasek P, Durner J (2006) Plant nitric oxide synthase: a never-ending story? Trends Plant Sci 11:524–525
Zhao L, Zhang F, Guo J, Yang Y, Li B, Zhang L (2004) Nitric oxide functions as a signal in salt resistance in the calluses from two ecotypes of reed (Phragmites communis Trin.). Plant Physiol 134:849–857
Acknowledgments
This work was supported by grants from the Universidad de Buenos Aires (Argentina) and from Consejo Nacional de Investigaciones Científicas y Técnicas (CONICET) (Argentina). K.B.B., A.B. and M.L.T. are Career Investigators from CONICET.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Noriega, G.O., Yannarelli, G.G., Balestrasse, K.B. et al. The effect of nitric oxide on heme oxygenase gene expression in soybean leaves. Planta 226, 1155–1163 (2007). https://doi.org/10.1007/s00425-007-0561-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-007-0561-8