Abstract
Myonecrosis is a frequent clinical manifestation of envenomings by Viperidae snakes, mainly caused by the toxic actions of secreted phospholipase A2 (sPLA2) enzymes and sPLA2-like homologs on skeletal muscle fibers. A hallmark of the necrotic process induced by these myotoxins is the rapid appearance of hypercontracted muscle fibers, attributed to the massive influx of Ca2+ resulting from cell membrane damage. However, the possibility of myotoxins having, in addition, a direct effect on the contractile machinery of skeletal muscle fibers when internalized has not been investigated. This question is here addressed by using an ex vivo model of single-skinned muscle fibers, which lack membranes but retain an intact contractile apparatus. Rabbit psoas skinned fibers were exposed to two types of myotoxins of Bothrops asper venom: Mt-I, a catalytically active Asp49 sPLA2 enzyme, and Mt-II, a Lys49 sPLA2-like protein devoid of phospholipolytic activity. Neither of these myotoxins affected the main parameters of force development in striated muscle sarcomeres of the skinned fibers. Moreover, no microscopical alterations were evidenced after their exposure to Mt-I or Mt-II. In contrast to the lack of effects on skinned muscle fibers, both myotoxins induced a strong hypercontraction in myotubes differentiated from murine C2C12 myoblasts, with drastic morphological alterations that reproduce those described in myonecrotic tissue in vivo. As neither Mt-I nor Mt-II showed direct effects upon the contractile apparatus of skinned fibers, it is concluded that the mechanism of hypercontraction triggered by both myotoxins in patients involves indirect effects, i.e., the large cytosolic Ca2+ increase after sarcolemma permeabilization.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Envenomings by snakes of the family Viperidae are characterized, among other clinical manifestations, by prominent skeletal muscle necrosis [13, 41]. This effect is primarily due to the direct action of myotoxic secreted phospholipase A2 (sPLA2) enzymes and sPLA2 homologs on skeletal muscle fibers [16, 22]. These two types of viperid myotoxins, which belong to the group II sPLA2s [37], differ in their ability to hydrolyze phospholipids, owing to variations in residues at the catalytic site and the Ca2+-binding loop. Myotoxic Asp49 sPLA2s have the canonical catalytic residues of this group of secreted enzymes and are, therefore, enzymatically active [32]. In contrast, sPLA2 homologs present substitutions at position 49 (numbering according to [36]), where a Lys substitutes Asp in most cases, and in some residues comprising the Ca2+-binding loop [12, 27]. As a consequence, these variants are devoid of enzymatic activity [6, 11]. Despite this variation in their catalytic properties, both types of proteins induce skeletal muscle necrosis [17, 22].
Studies carried out in vivo, in cell culture models, and with artificial membranes have addressed the mechanism of action of these two variants of myotoxic sPLA2s. In both cases, there is a rapid disruption of the plasma membrane of muscle fibers in vivo and of myotubes in culture, as well as of liposomes, followed by a rapid series of intracellular degenerative events in the first two models [9, 14, 17, 23, 40]. In the case of Asp49 sPLA2s, myotoxicity depends on phospholipid hydrolysis, since inhibition of catalysis abolishes their action on muscle cells [33]. In the case of Lys49 sPLA2-like homologs, plasma membrane disruption occurs in the absence of phospholipid hydrolysis and is likely to depend on the ability of these toxins to bind and disrupt the integrity of membranes through the action of cationic and hydrophobic residues [10, 22]. Interestingly, these two types of myotoxins act synergistically [34], hence providing an adaptive advantage for enhanced activity on muscle tissue. In addition to the direct interaction and disruption that these toxins exert in the plasma membrane of muscle fibers, there is evidence indicating that they are also internalized [31, 38], although the implications of such internalization for the mechanisms of cell damage have not been elucidated.
The initial disruption in the integrity of the plasma membrane leads to a number of cellular derangements. The main functional consequence of such alteration is a prominent influx of Ca2+ [8, 40], which, in turn, unleashes a series of intracellular events such as myofilament hypercontraction and mitochondrial alterations [16, 17, 32]. However, it is unknown whether the hypercontraction of myofilaments is only due to the increase of cytosolic Ca2+ or whether the toxins, once internalized, are able to directly affect the contractile apparatus. An experimental strategy to address this question is based on the use of isolated skinned fibers which are devoid of membranes but maintain an intact contractile apparatus [18].
A previous study used this experimental platform to address the action of a myotoxic Lys49 sPLA2 homolog on rat cardiomyocytes [29]. It was observed that this myotoxin induced hypercontraction of cardiomyocytes associated with plasma membrane disruption and increases in cytosolic Ca2+ levels without directly affecting the contractile apparatus. Since the main target of myotoxic sPLA2s is skeletal muscle, in the present study we assessed the effect of a myotoxic Asp49 sPLA2 (Mt-I) and a Lys49 sPLA2 homolog (Mt-II) on skinned skeletal muscle fibers prepared from rabbit psoas muscle. In parallel, we assessed the action of these myotoxins on myotubes in culture. Our findings show that both myotoxins induce cytotoxicity associated with myofilament hypercontraction in myotubes in culture, but do not have a direct effect on the contractile apparatus in skinned muscle fibers.
Material and methods
Isolation of myotoxins
Myotoxin-I (Mt-I; Uniprot P20474) is an Asp49 sPLA2 enzyme, while Myotoxin-II (Mt-II; UniProt P24605) is a Lys49 sPLA2-like protein, both isolated from the venom of Bothrops asper of adult specimens collected in the Pacific versant of Costa Rica. Isolation of the toxins was performed by ion-exchange chromatography on CM-Sephadex C-25 [24] followed by reverse-phase HPLC on a semi-preparative C8 column, as described previously [33].
Solutions for mechanical experiments
Solutions for mechanical experiments were prepared as previously described [4, 5]. All solutions were adjusted to pH 7.0. Free Ca2+ concentrations were expressed as pCa (−LOG 10[Ca2+]) and were in the range of 7.50 (relaxing solution) to 4.47 (activating solution). The skinning solution contained 5.0 mM KH2PO4, 3.0 mM Mg-acetate, 5.0 mM EGTA, 1.0 mM Na2ATP, 50 mM Na-creatine phosphate, 5.0 mM NaN3, 10 mM glutathione, 2.0 mM dithiothreitol, and a protease inhibitor cocktail. Pre-rigor solution (22°C) contained 10 mM imidazole, 2.5 mM EGTA, 7.5 mM EDTA, and 135 mM potassium propionate. Rigor solution (22°C) contained 10 mM imidazole, 2.5 mM EGTA, 2.5 mM EDTA, and 150 mM potassium propionate. Relaxing and activating solutions (5°C) contained 10 mM imidazole, 2.0 mM MgCl2, 1.0 mM MgATP, 1.0 mM EGTA (or CaEGTA), 50 mM sodium creatine phosphate, and 500 U/mL of creatine kinase. Solutions with different pCa values were obtained by mixing appropriate volumes of relaxing and activating solutions. For positive control experiments, increasing concentrations of the myosin inhibitors para-amino-blebbistatin (AmBleb; 6.2, 12.5, 25, and 50 μM) and 2,3-Butandione 2-monoxime (BDM; 10, 20, 30, 40 and 50 mM) were added to activating solution. Chemicals were obtained from Sigma-Aldrich Chemie GmbH (Munich, Germany). AmBleb was obtained from Motorpharma Ltd. (Budapest, Hungary).
Preparation of single-skinned muscle fibers for mechanical experiments
Single fast-twitch muscle fibers were prepared as previously described [2, 43]. Briefly, fiber bundles were isolated from rabbit psoas muscles and incubated for 30 min at 4°C in a skinning solution containing 0.5 % Triton X-100. The skinned fiber bundles were thereafter equilibrated in Triton X-100 free skinning solution containing 3 M sucrose and a protease inhibitor cocktail. The skinned bundles were subsequently shock-frozen in liquid propane and stored in liquid nitrogen in screw cap tubes. For experiments, skinned fiber bundles were thawed in a sucrose-free skinning solution containing a fresh protease inhibitor cocktail. Single-skinned muscle fibers were thereafter gently isolated with forceps and kept for up to 4 days at 4°C without a detectable loss of function [20, 21].
Protocol for mechanical experiments
The methodology and the apparatus used to perform mechanical experiments on isometric-held single-skinned fibers have been previously described [2, 3]. Briefly, single-skinned fibers (8–10 mm long) were mounted between the force transducer and a length driver of the apparatus using cyanoacrylate glue (Histoacryl; B. Braun Surgical GmbH, Melsungen, Germany). The ends of the fibers were thereafter stiffened with glutaraldehyde, using pre-rigor and rigor solutions as previously described [20]. The sarcomere length was set to 2.4 μm in a relaxing solution by means of laser diffraction [2, 3]. The mounted fiber was transferred to activating solution (saturating Ca2+ concentration, pCa 4.47) and maximal isometric force (Forcemax) and the rate constant of isometric force redevelopment after a short period of isotonic shortening (KTRmax) were measured. To this end, the fiber was cycled every 5 s between isometric steady-state contraction and short (330 ms) periods of unloaded isotonic shortening with subsequent restretch to the initial (isometric) sarcomere length; see [28, 30]. Subsequently, the isometric force was also measured at different pCa values corresponding to several sub-saturating Ca2+ concentrations. KTRmax values were obtained by fitting force transients obtained at pCa 4.47 using a single-exponential function. Force−pCa relations were fitted using a sigmoidal Hill equation:
where Fn is the force at pCa = −LOG10 [Ca2+] normalized to maximum force at pCa 4.47, pCa50 is pCa when Fn = 0.5, and nH is the steepness (Hill coefficient) of the force−pCa relation.
The above-described protocol was performed before and after incubating the mounted skinned fibers in 40 μg/mL Mt-I or Mt-II for 60 min in a relaxing solution. The apparatus used for these mechanical experiments is mounted on an inverted microscope, such that photos of specific sections of the isometric-held skinned fibers could be taken before and after toxin incubations. The experimental protocol is described in Fig. 1.
Skinned fiber length and force generation during isotonic and isometric contraction. a By modifying the position of the length driver, an isometric-held skinned fiber undergoes a transient phase of isotonic shortening (330 ms) and is then re-stretched to its original length, back to isometric contraction. The dotted lines show the time points of the switch from isometric to isotonic (slightly loaded) and back to isometric conditions. The procedure is performed in activating solution (pCa 4.47, i.e., saturating Ca2+ concentrations) and in different ratios of activating and relaxing solutions (i.e., sub-saturating Ca2+ concentrations). The described procedure is repeated several times for every pCa value and the resulting transients are added to generate representative or mean transients (essentially the same transients with even better signal-to-noise ratio; not shown). b Force transients corresponding to the length transients shown in A. Note the clear and fast reduction of force in the transition to the isotonic phase and the fast, pCa-dependent force redevelopment rate after switching back to isometric conditions. kTRmax values were obtained by single-exponential fits of force transients during the force redevelopment phase observed in mean force transients at pCa 4.47 (arrow). For subsequent analysis (Fig. 2a, c, and e), isometric force at sub-saturating Ca2+ concentrations was normalized to isometric force obtained at pCa 4.47 in mean force transients
Positive control for mechanical experiments using skinned fibers
The experiments described in the “Protocol for mechanical experiments” section and Fig. 1. were replicated by incubation of an additional set of skinned fibers in increasing concentrations of the myosin inhibitors AmBleb and BDM at pCa 4.47 (i.e., saturating Ca2+ concentration). Both myosin inhibitors influence the cross-bridge cycle of striated muscle, which in turn is reflected in several parameters of force development [19, 30, 35]. This set of experiments was designed as a positive control in order to show that the experimental protocol used here is sensitive to the effects of molecules that target and influence sarcomeric proteins.
Experiments on myotubes
Differentiation of C2C12 myotubes
The cell culture was performed as described [1]. Briefly, C2C12 mouse progenitor myoblast cells (RRID:CVCL_0188) were seeded on 10 and 18 mm laminin-coated coverslips in DMEM/High Glucose (Capricorn Scientific DMEM-HA) with 1% penicillin and streptomycin (Capricorn Scientific PS-B) containing 15% fetal bovine serum (Gibco 10100147) until confluent (approx. 100,000 cells/cm2). Differentiation was induced by switching to a medium containing 2% horse serum (Gibco 26050088) for 6 days.
Myotoxin-induced morphological changes of myotubes
On the 6th differentiation day, plates containing myotubes were placed on an inverted microscope. The surface of the inverted microscope was previously set at 37°C. Thereafter, pre-warmed aliquots of culture medium containing Mt-I or Mt-II were gently added to the wells, achieving a final concentration of 40 μg/mL. Morphological changes of myotoxin-exposed myotubes were followed by bright-field microscopy for 60 min.
Statistics and data analysis
All values are given as a mean ± SEM (standard error of the mean) with n representing the number of experiments. Mean values were compared using unpaired or paired Student’s t-test (P < 0.05) or one-way analysis of variance (ANOVA, P<0.05) and post hoc analyses of Tukey (multiple comparison test for single effects) as appropriate. Statistical analysis and figure layout were performed using GraphPad Prism (version 5.01; RRID:SCR_002798) for Windows, GraphPad Software, San Diego, CA, USA.
Results
Effect of Mt-I and Mt-II on single-skinned muscle fibers
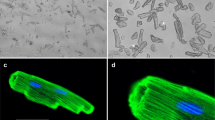
To evaluate the effects of myotoxins on the contractile apparatus of striated muscle, single-skinned muscle fibers were incubated with 40 μg/mL Mt-I or Mt-II for 60 min in a relaxing solution. The results of these experiments are summarized in Fig. 2 and Table 1. Figure 2a and c show the normalized force development of single-skinned fibers before and after incubation with Mt-I or Mt-II, respectively. The exposure to myotoxins did not influence Ca2+ sensitivity nor cooperativity of force development, as evidenced by the similar pCa50 values and Hill coefficients (nH) before and after myotoxin treatment in both experiments (Table 1). On the other hand, the rate constant of force redevelopment at saturating Ca2+ concentration (kTRmax) was significantly lower after Mt-I or Mt-II incubation, as shown in Figs. 2b and d, respectively. To control whether kTRmax decrease was induced by myotoxins or alternatively by the experimental treatment itself (i.e., two full sets of Ca2+-induced activations, including numerous shortening – re-stretch cycles), the experiment was replicated by incubating a third group of skinned fibers in relaxing solution for 60 min without toxins. This experiment also controls possible masked effects in the experiments presented in Figs. 2a and c (i.e., opposite, cancelling effects of the experimental treatment and toxins on pCa50 values and Hill coefficients). As shown in Fig. 2e and Table 1, the results of this additional control experiment are essentially the same as in the experiments including myotoxin treatment (Figs. 2a and c), confirming the absence of any myotoxin-induced effect on Ca2+-sensitivity or cooperativity of force development. Additionally, Fig. 2f shows a significant decrease of kTRmax in this toxin-free control experiment, confirming that this change is not induced by the toxins, but by the experimental protocol. To control for a possible additive effect of toxins and treatment on kTRmax results, the relative magnitude of kTRmax decrease (i.e., ratio Post-/Pre-incubation; kTRmax Post/Pre) in all three experiments was calculated (Fig. 3a). For both toxin and toxin-free experiments, the observed kTRmax decrease was always ~20 % and non-significant differences were detected (Table 2). Finally, Fig. 3b shows maximal force development at saturating Ca2+ concentration (pCa 4.47) after myotoxin or myotoxin-free incubation as compared to its magnitude before incubation (i.e., ratio Post-/Pre-incubation; Forcemax Post/Pre). As for all the previously analyzed variables, no statistically significant differences were detected. In contrast to the lack of effect of both myotoxins on skinned fibers, the set of experiments summarized in Fig. 4 using myosin inhibitors shows that the experimental protocol is able to detect the effects of agents that affect sarcomere proteins. This positive control rules out the possibility of a lack of detection capability in the experiments performed. Altogether, this set of mechanical experiments strongly suggests that Mt-I and Mt-II do not affect the main parameters of force development in striated muscle sarcomeres. In line with the mechanical experiments, bright field microscopy of segments of the skinned fibers used for experiments shown in Figs. 2a and c does not evidence structural changes after incubation with myotoxins (Fig. 5).
Effect of Mt-I and Mt-II on force-pCa relationship and kTRmax of single-skinned muscle fibers. a, c, and e Force-pCa relationship of skinned fibers before and after being exposed to 40 μg/mL of either Mt-I (a) or Mt-II (b) for one hour in a relaxing solution. Ca2+-induced isometric force was not different before (Pre) and after (Mt-I, Mt-II) treatment. The same result was observed for control fibers undergoing the same procedure without Mt exposure (e). See Table 1. b, d, and f kTRmax: Rate constant of force development of skinned fibers at saturating Ca2+ concentration (pCa 4.47). kTRmax was significantly lower after exposition to 40 μg/mL of either Mt-I (b) or MT-II (d) for one hour in a relaxing solution. However, a similar reduction was observed on control fibers undergoing the same procedure without Mt exposure (f). *: significantly lower kTRmax than in the corresponding Pre condition. See bold entries in Table 1
Relative changes of kTRmax and Forcemax after incubation with myotoxins. a kTRmax of skinned fibers after being exposed to 40 μg/mL myotoxins or control incubation for one hour in relaxing solution as normalized to its corresponding magnitude before incubation. b Isometric force at saturating Ca2+ concentration (pCa 4.47) of skinned fibers after being exposed to 40 μg/mL myotoxins or control incubation for one hour in relaxing solution as normalized to its corresponding magnitude before incubation. See Table 2
Effect of myosin inhibitors on force development. a Original force redevelopment transients recorded at saturating Ca2+ concentration. Experimental conditions correspond to the top transient shown in Fig. 1b (pCa 4.47). Note how increasing concentrations of the myosin inhibitor AmBleb (subsequent transients at 6.2, 12.5, 25, and 50 μM [AmBleb]) reduce total force generation, even though the Ca2+ concentration remains the same. b Same experimental protocol as in a., but using a different myosin inhibitor (BDM). Instead of the original force transients, only the resulting force development as a function of [BDM] is shown. As in the case of AmBleb, BDM clearly reduces force generation at saturating Ca2+ concentration in a dose-dependent manner (n = 7). c Comparison of maximum force inhibition achieved by AmBleb and BDM. At those concentrations, force inhibition becomes asymptotic (as shown in b.), such that further increases in the myosin inhibitors did not induce additional force inhibition (n = 5 for AmBleb and 7 for BDM)
Effect of Mt-I and Mt-II on myotubes
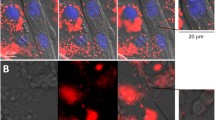
In order to contrast the lack of effects caused by myotoxins on skinned muscle fibers compared to intact skeletal muscle cells with cell membranes, myotubes differentiated from mouse C2C12 myoblasts progenitor cells were exposed to either Mt-I or Mt-II. These experiments are summarized in Fig. 6. In this cell model, both myotoxins induced a strong hypercontraction leading to clear morphological alterations. Hypercontraction originated at a particular point of the cell and was followed by a retraction along its longitudinal axis. Online resources 1 and 2 show an example of the typical hypercontraction of myotubes after myotoxin exposure.
Effect of Mt-I and Mt-II on morphology of C2C12 myoblast-derived myotubes. a, b No changes in myotubes morphology were observed by light microscopy before (a) and after (b) 60 min control incubation. c, d Clear differences in myotubes morphology were observed before (c) and after (d) incubation with 40 μg/mL Mt-I for 60 min. e, f Same as in c, d for Mt-II. Myotoxins induced a strong hypercontraction of myotubes. White bar 100 μm. See online resources 1 and 2
Discussion
In spite of significant advances towards characterizing and understanding the effects and mode of action of snake venom sPLA2 toxins [15, 25], their molecular, functional, and structural details have been only partially elucidated. In the case of myotoxic sPLA2s from viperids, compelling evidence supports a membrane-permeabilizing action leading to a massive increase of cytoplasmic Ca2+, both for enzymatically active (Asp49) sPLA2s and enzymatically inactive (Lys49) sPLA2-like homologs [7, 8, 32, 40]. Consequent to this Ca2+ increment, a rapid and strong hypercontraction is observed in skeletal muscle fibers after local injection of these myotoxins in mice [17, 26]. Interestingly, recent studies have revealed that the myotoxins are rapidly internalized in myogenic cells in vitro [31] and in mature skeletal muscle fibers in vivo [38], underscoring the relevance of assessing their possible effects on intracellular targets, in addition to their membranotropic actions.
Skinned muscle cells represent a useful ex vivo model to discern between direct and indirect actions of biomolecules on the contractile apparatus and have been recently used to compare the effects of a Lys49 sPLA2 homolog, Mt-II, on isolated intact vs. skinned rat cardiomyocytes [29]. Exposure to Mt-II led to a rapid cytosolic Ca2+ increase and hypercontraction in intact cardiomyocytes, but did not have effects on the contractile apparatus of the skinned cells. Aiming to extend these observations to skeletal muscle and to a catalytically active myotoxin, the present study investigated the effects of Mt-I and Mt-II on rabbit skinned skeletal muscle fibers. Exposure of skinned fibers to these myotoxins did not induce any direct alterations of parameters such as Ca2+ sensitivity or cooperativity of force development. A slight decrease recorded for the rate constant of force redevelopment (kTRmax) after incubation with the myotoxins is attributable to the experimental treatment per se, since it was also observed in control experiments in the absence of toxins.
Experiments with myosin inhibitors showed a strong force inhibition on skinned fibers. AmBleb is a derivative of blebbistatin that prevents myosin from entering the force-generating states by stabilizing the closed state of the switch 2 element of the nucleotide-binding site. In this state, both ADP and Pi are bound to the active site and the formation of the strong actomyosin interaction is inhibited [35, 39, 42, 44]. BDM is also a widely used myosin inhibitor. Recent research has suggested that BDM accelerates ATP cleavage of skeletal myosin subfragment 1 and alters the microenvironment around the phosphorus atoms of myosin-bound ATP analogs, thereby inhibiting active force generation [19]. Our positive controls with both inhibitors confirm the suitability of our setup and experimental protocol to detect changes at the level of the cross-bridge cycle. In addition, we have previously shown that myosin inhibitors can also modify other key variables of force development in our skinned fiber preparation, including its Ca2+ sensitivity and cooperativity (i.e., pCa50 and nH values, respectively) [30].
By microscopical assessment, no morphological changes were evidenced in segments of skinned fibers exposed to the myotoxins. Therefore, it can be concluded that neither Mt-I nor Mt-II has direct effects on the contractile machinery of skeletal muscle fibers and that hypercontraction of fibers results from indirect effects, i.e., the large cytosolic Ca2+ increase after sarcolemma permeabilization. Such hypercontraction observed in skeletal muscle tissue [17, 26] was readily reproduced in myotubes differentiated in vitro from mouse C2C12 myoblasts, after exposure to either Mt-I or Mt-II. It remains to be determined whether these myotoxins, once internalized, affect other intracellular structures, such as the sarcoplasmic reticulum and mitochondria, and whether these alterations contribute to cellular damage. Moreover, the mechanical disruptive effect of hypercontraction in the integrity of the plasma membrane is another possible effect that might contribute to cell damage and needs to be investigated.
Data availability
The datasets generated during the present study are available from the corresponding author upon reasonable request.
References
Amrute-Nayak M, Gand LV, Khan B, Holler T, Kefalakes E, Kosanke M, Kraft T, Nayak A (2022) SENP7 deSUMOylase-governed transcriptional program coordinates sarcomere assembly and is targeted in muscle atrophy. Cell Rep 41:111702. https://doi.org/10.1016/j.celrep.2022.111702
Brenner B (1983) Technique for stabilizing the striation pattern in maximally calcium-activated skinned rabbit psoas fibers. Biophys J 41:99–102. https://doi.org/10.1016/S0006-3495(83)84411-7
Brenner B (1988) Effect of Ca2+ on cross-bridge turnover kinetics in skinned single rabbit psoas fibers: implications for regulation of muscle contraction. Proc Natl Acad Sci U S A 85:3265–3269. https://doi.org/10.1073/pnas.85.9.3265
Brenner B, Chalovich JM (1999) Kinetics of thin filament activation probed by fluorescence of N-((2-(iodoacetoxy)ethyl)-N-methyl)amino-7-nitrobenz-2-oxa-1,3-diazole-labeled troponin I incorporated into skinned fibers of rabbit psoas muscle: implications for regulation of muscle contraction. Biophys J 77:2692–2708. https://doi.org/10.1016/S0006-3495(99)77103-1
Brenner B, Kraft T, Yu LC, Chalovich JM (1999) Thin filament activation probed by fluorescence of N-((2-(iodoacetoxy)ethyl)-N-methyl)amino-7-nitrobenz-2-oxa-1,3-diazole-labeled troponin I incorporated into skinned fibers of rabbit psoas muscle. Biophys J 77:2677–2691. https://doi.org/10.1016/S0006-3495(99)77102-X
Chioato L, De Oliveira AH, Ruller R, Sa JM, Ward RJ (2002) Distinct sites for myotoxic and membrane-damaging activities in the C-terminal region of a Lys49-phospholipase A2. Biochem J 366:971–976. https://doi.org/10.1042/BJ20020092
Cintra-Francischinelli M, Pizzo P, Angulo Y, Gutierrez JM, Montecucco C, Lomonte B (2010) The C-terminal region of a Lys49 myotoxin mediates Ca2+ influx in C2C12 myotubes. Toxicon 55:590–596. https://doi.org/10.1016/j.toxicon.2009.10.013
Cintra-Francischinelli M, Pizzo P, Rodrigues-Simioni L, Ponce-Soto LA, Rossetto O, Lomonte B, Gutierrez JM, Pozzan T, Montecucco C (2009) Calcium imaging of muscle cells treated with snake myotoxins reveals toxin synergism and presence of acceptors. Cell Mol Life Sci 66:1718–1728. https://doi.org/10.1007/s00018-009-9053-2
Diaz C, Gutierrez JM, Lomonte B, Gene JA (1991) The effect of myotoxins isolated from Bothrops snake venoms on multilamellar liposomes: relationship to phospholipase A2, anticoagulant and myotoxic activities. Biochim Biophys Acta 1070:455–460. https://doi.org/10.1016/0005-2736(91)90086-n
Fernandes CA, Borges RJ, Lomonte B, Fontes MR (2014) A structure-based proposal for a comprehensive myotoxic mechanism of phospholipase A2-like proteins from viperid snake venoms. Biochim Biophys Acta 1844:2265–2276. https://doi.org/10.1016/j.bbapap.2014.09.015
Fernandez J, Caccin P, Koster G, Lomonte B, Gutierrez JM, Montecucco C, Postle AD (2013) Muscle phospholipid hydrolysis by Bothrops asper Asp49 and Lys49 phospholipase A2 myotoxins--distinct mechanisms of action. FEBS J 280:3878–3886. https://doi.org/10.1111/febs.12386
Francis B, Gutierrez JM, Lomonte B, Kaiser II (1991) Myotoxin II from Bothrops asper (Terciopelo) venom is a lysine-49 phospholipase A2. Arch Biochem Biophys 284:352–359. https://doi.org/10.1016/0003-9861(91)90307-5
Gutierrez JM, Calvete JJ, Habib AG, Harrison RA, Williams DJ, Warrell DA (2017) Snakebite envenoming. Nat Rev Dis Primers 3:17063. https://doi.org/10.1038/nrdp.2017.63
Gutierrez JM, Chaves F, Gene JA, Lomonte B, Camacho Z, Schosinsky K (1989) Myonecrosis induced in mice by a basic myotoxin isolated from the venom of the snake Bothrops nummifer (jumping viper) from Costa Rica. Toxicon 27:735–745. https://doi.org/10.1016/0041-0101(89)90040-8
Gutierrez JM, Lomonte B (2013) Phospholipases A2: unveiling the secrets of a functionally versatile group of snake venom toxins. Toxicon 62:27–39. https://doi.org/10.1016/j.toxicon.2012.09.006
Gutierrez JM, Ownby CL (2003) Skeletal muscle degeneration induced by venom phospholipases A2: insights into the mechanisms of local and systemic myotoxicity. Toxicon 42:915–931. https://doi.org/10.1016/j.toxicon.2003.11.005
Gutierrez JM, Ownby CL, Odell GV (1984) Pathogenesis of myonecrosis induced by crude venom and a myotoxin of Bothrops asper. Exp Mol Pathol 40:367–379. https://doi.org/10.1016/0014-4800(84)90054-6
Kalakoutis M, Di Giulio I, Douiri A, Ochala J, Harridge SDR, Woledge RC (2021) Methodological considerations in measuring specific force in human single skinned muscle fibres. Acta Physiol (Oxf) 233:e13719. https://doi.org/10.1111/apha.13719
Komatsu H, Koseki Y, Kanno T, Aoki S, Kodama T (2017) 2,3-Butandione 2-monoxime inhibits skeletal myosin II by accelerating ATP cleavage. Biochem Biophys Res Commun 490:849–854. https://doi.org/10.1016/j.bbrc.2017.06.130
Kraft T, Chalovich JM, Yu LC, Brenner B (1995) Parallel inhibition of active force and relaxed fiber stiffness by caldesmon fragments at physiological ionic strength and temperature conditions: additional evidence that weak cross-bridge binding to actin is an essential intermediate for force generation. Biophys J 68:2404–2418. https://doi.org/10.1016/S0006-3495(95)80423-6
Kraft T, Messerli M, Rothen-Rutishauser B, Perriard JC, Wallimann T, Brenner B (1995) Equilibration and exchange of fluorescently labeled molecules in skinned skeletal muscle fibers visualized by confocal microscopy. Biophys J 69:1246–1258. https://doi.org/10.1016/S0006-3495(95)80018-4
Lomonte B (2023) Lys49 myotoxins, secreted phospholipase A2-like proteins of viperid venoms: A comprehensive review. Toxicon 224:107024. https://doi.org/10.1016/j.toxicon.2023.107024
Lomonte B, Angulo Y, Rufini S, Cho W, Giglio JR, Ohno M, Daniele JJ, Geoghegan P, Gutierrez JM (1999) Comparative study of the cytolytic activity of myotoxic phospholipases A2 on mouse endothelial (tEnd) and skeletal muscle (C2C12) cells in vitro. Toxicon 37:145–158. https://doi.org/10.1016/s0041-0101(98)00171-8
Lomonte B, Gutierrez JM (1989) A new muscle damaging toxin, myotoxin II, from the venom of the snake Bothrops asper (terciopelo). Toxicon 27:725–733. https://doi.org/10.1016/0041-0101(89)90039-1
Lomonte B, Križaj I (2021) Snake venom phospholipase A2 toxins. In: Handbook of Venoms and Toxins of Reptiles. In: Mackessy SP (ed) Handbook of Venoms and Toxins of Reptiles, 2nd edn. CRC Press, Boca Raton, pp 389–411
Lomonte B, Lundgren J, Johansson B, Bagge U (1994) The dynamics of local tissue damage induced by Bothrops asper snake venom and myotoxin II on the mouse cremaster muscle: an intravital and electron microscopic study. Toxicon 32:41–55. https://doi.org/10.1016/0041-0101(94)90020-5
Lomonte B, Rangel J (2012) Snake venom Lys49 myotoxins: From phospholipase A2 to non-enzymatic membrane disruptors. Toxicon 60:520–530. https://doi.org/10.1016/j.toxicon.2012.02.007
Lopez-Davila AJ, Chalovich JM, Zittrich S, Piep B, Matinmehr F, Malnasi-Csizmadia A, Rauscher AA, Kraft T, Brenner B, Stehle R (2020) Cycling cross-bridges contribute to thin filament activation in human slow-twitch fibers. Front Physiol 11:144. https://doi.org/10.3389/fphys.2020.00144
Lopez-Davila AJ, Weber N, Kraft T, Matinmehr F, Arias-Hidalgo M, Fernandez J, Lomonte B, Gutierrez JM (2021) Cytotoxicity of snake venom Lys49 PLA2-like myotoxin on rat cardiomyocytes ex vivo does not involve a direct action on the contractile apparatus. Sci Rep 11:19452. https://doi.org/10.1038/s41598-021-98594-5
Lopez Davila AJ, Zhu L, Fritz L, Kraft T, Chalovich JM (2020) The positively charged C-terminal region of human skeletal troponin T retards activation and decreases calcium sensitivity. Biochemistry 59:4189–4201. https://doi.org/10.1021/acs.biochem.0c00499
Massimino ML, Simonato M, Spolaore B, Franchin C, Arrigoni G, Marin O, Monturiol-Gross L, Fernandez J, Lomonte B, Tonello F (2018) Cell surface nucleolin interacts with and internalizes Bothrops asper Lys49 phospholipase A2 and mediates its toxic activity. Sci Rep 8:10619. https://doi.org/10.1038/s41598-018-28846-4
Montecucco C, Gutierrez JM, Lomonte B (2008) Cellular pathology induced by snake venom phospholipase A2 myotoxins and neurotoxins: common aspects of their mechanisms of action. Cell Mol Life Sci 65:2897–2912. https://doi.org/10.1007/s00018-008-8113-3
Mora-Obando D, Diaz C, Angulo Y, Gutierrez JM, Lomonte B (2014) Role of enzymatic activity in muscle damage and cytotoxicity induced by Bothrops asper Asp49 phospholipase A2 myotoxins: are there additional effector mechanisms involved? PeerJ 2:e569. https://doi.org/10.7717/peerj.569
Mora-Obando D, Fernandez J, Montecucco C, Gutierrez JM, Lomonte B (2014) Synergism between basic Asp49 and Lys49 phospholipase A2 myotoxins of viperid snake venom in vitro and in vivo. PLoS One 9:e109846. https://doi.org/10.1371/journal.pone.0109846
Rauscher AA, Gyimesi M, Kovacs M, Malnasi-Csizmadia A (2018) Targeting myosin by blebbistatin derivatives: optimization and pharmacological potential. Trends Biochem Sci 43:700–713. https://doi.org/10.1016/j.tibs.2018.06.006
Renetseder R, Brunie S, Dijkstra BW, Drenth J, Sigler PB (1985) A comparison of the crystal structures of phospholipase A2 from bovine pancreas and Crotalus atrox venom. J Biol Chem 260:11627–11634
Six DA, Dennis EA (2000) The expanding superfamily of phospholipase A(2) enzymes: classification and characterization. Biochim Biophys Acta 1488:1–19. https://doi.org/10.1016/s1388-1981(00)00105-0
Vargas-Valerio S, Robleto J, Chaves-Araya S, Monturiol-Gross L, Lomonte B, Tonello F, Fernandez J (2021) Localization of Myotoxin I and Myotoxin II from the venom of Bothrops asper in a murine model. Toxicon 197:48–54. https://doi.org/10.1016/j.toxicon.2021.04.006
Varkuti BH, Kepiro M, Horvath IA, Vegner L, Rati S, Zsigmond A, Hegyi G, Lenkei Z, Varga M, Malnasi-Csizmadia A (2016) A highly soluble, non-phototoxic, non-fluorescent blebbistatin derivative. Sci Rep 6:26141. https://doi.org/10.1038/srep26141
Villalobos JC, Mora R, Lomonte B, Gutierrez JM, Angulo Y (2007) Cytotoxicity induced in myotubes by a Lys49 phospholipase A2 homologue from the venom of the snake Bothrops asper: evidence of rapid plasma membrane damage and a dual role for extracellular calcium. Toxicol In Vitro 21:1382–1389. https://doi.org/10.1016/j.tiv.2007.04.010
Warrell DA (2010) Snake bite. Lancet 375:77–88. https://doi.org/10.1016/S0140-6736(09)61754-2
Xu S, White HD, Offer GW, Yu LC (2009) Stabilization of helical order in the thick filaments by blebbistatin: further evidence of coexisting multiple conformations of myosin. Biophys J 96:3673–3681. https://doi.org/10.1016/j.bpj.2009.01.049
Yu LC, Brenner B (1989) Structures of actomyosin crossbridges in relaxed and rigor muscle fibers. Biophys J 55:441–453. https://doi.org/10.1016/S0006-3495(89)82838-3
Zhao FQ, Padron R, Craig R (2008) Blebbistatin stabilizes the helical order of myosin filaments by promoting the switch 2 closed state. Biophys J 95:3322–3329. https://doi.org/10.1529/biophysj.108.137067
Acknowledgements
The authors thank the excellent technical support of Faramarz Matinmehr, U. Dávila, and S. Kerschensteiner.
Funding
The authors thank Vicerrectoría de Investigación (VI-741-B5-602 and VI-741-B4-100), Universidad de Costa Rica for partial support to these studies (to JF, JMG, and BL). Parts of that work were additionally funded by Deutsche Forschungsgemeinschaft (TRR267; to TT) and Collaborative Research Center (1470 “HFpEF”; to TT). We thank Deutsche Gesellschaft für Kardiologie for Research’s Stipend (to NW) and Förderstiftung Medizinische Hochschule Hannover (to NW). The sponsors had no role in the study design, in the collection, analysis, and interpretation of data, in the writing of the report, and in the decision to submit the article for publication.
Author information
Authors and Affiliations
Contributions
AJLD, NW, JF, BL, and JMG conceived the experimental design. AJLD and LF performed and analyzed mechanical experiments on skinned fibers. NW and KRM performed and analyzed experiments on myotubes. AJLD, NW, BL, and JMG wrote the first draft of the manuscript. AN and LVG cultured myoblasts and differentiated them into myotubes. E.W. performed BDM-mechanical experiments. JF, BL, JMG, TK, and TT supervised the project. All authors commented on the manuscript and approved its last version.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable
Consent to publish
Not applicable
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Online Resource 1 Baseline: Myotubes morphology observed by light microscopy 1 min after incubation with 40 μg/mL Mt-II. The myotoxin has not exerted its effect.
Online Resource 2 Clear differences in some myotubes morphology were observed after 17 min incubation with 40 μg/mL Mt-II. Note the fast hypercontraction of some myotubes.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
López-Dávila, A.J., Weber, N., Nayak, A. et al. Skeletal muscle fiber hypercontraction induced by Bothrops asper myotoxic phospholipases A2 ex vivo does not involve a direct action on the contractile apparatus. Pflugers Arch - Eur J Physiol 475, 1193–1202 (2023). https://doi.org/10.1007/s00424-023-02840-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-023-02840-w