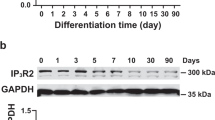
Abstract
Human pluripotent stem cell-derived cardiomyocytes (hPSC-CMs) have great value for studies of human cardiac development, drug discovery, disease modeling, and cell therapy. However, the mixed cardiomyocyte subtypes (ventricular-, atrial-, and nodal-like myocytes) and the maturation heterogeneity of hPSC-CMs restrain their application in vitro and in vivo. Myosin light chain 2 (MYL2, encoding the ventricular/cardiac muscle isoform MLC2v protein) is regarded as a ventricular-specific marker of cardiac myocardium; however, its restricted localization to ventricles during human heart development has been questioned. Consequently, it is currently unclear whether MYL2 definitively marks ventricular hESC-CMs. Here, by using a MYL2-Venus hESC reporter line, we characterized a time-dependent increase of the MYL2-Venus positive (MLC2v-Venus+) hESC-CMs during differentiation. We also compared the molecular, cellular, and functional properties between the MLC2v-Venus+ and MYL2-Venus negative (MLC2v-Venus-) hESC-CMs. At early differentiation stages of hESC-CMs, we reported that both MLC2v-Venus- and MLC2v-Venus+ CMs displayed ventricular-like traits but the ventricular-like cells from MLC2v-Venus+ hESC-CMs displayed more developed action potential (AP) properties than that from MLC2v-Venus- hESC-CMs. Meanwhile, about a half MLC2v-Venus- hESC-CM population displayed atrial-like AP properties, and a half showed ventricular-like AP properties, whereas only ~ 20% of the MLC2v-Venus- hESC-CMs expressed the atrial marker nuclear receptor subfamily 2 group F member 2 (NR2F2, also named as COUPTFII). At late time points, almost all MLC2v-Venus+ hESC-CMs exhibited ventricular-like AP properties. Further analysis demonstrates that the MLC2v-Venus+ hESC-CMs had enhanced Ca2+ transients upon increase of the MLC2v level during cultivation. Concomitantly, the MLC2v-Venus+ hESC-CMs showed more defined sarcomeric structures and better mitochondrial function than those in the MLC2v-Venus- hESC-CMs. Moreover, the MLC2v-Venus+ hESC-CMs were more sensitive to hypoxic stimulus than the MLC2v-Venus- hESC-CMs. These results provide new insights into the development of human ventricular myocytes and reveal a direct correlation between the expression profile of MLC2v and ventricular hESC-CM development. Our findings that MLC2v is predominantly a ventricular marker in developmentally immature hESC-CMs have implications for human development, drug screening, and disease modeling, and this marker should prove useful in overcoming issues associated with hESC-CM heterogeneity.







Similar content being viewed by others
References
Bai HJ, Zhang P, Ma L, Liang H, Wei G, Yang HT (2019) Smyd2 drives mesendodermal differentiation of human embryonic stem cells through mediating the transcriptional activation of key mesendodermal genes. Stem Cells 37:1401–1415. https://doi.org/10.1002/stem.3068
Bao ZZ, Bruneau BG, Seidman JG, Seidman CE, Cepko CL (1999) Regulation of chamber-specific gene expression in the developing heart by irx4. Science 283:1161–1164. https://doi.org/10.1126/science.283.5405.1161
Bizy A, Guerrero-Serna G, Hu B, Ponce-Balbuena D, Willis BC, Zarzoso M, Ramirez RJ, Sener MF, Mundada LV, Klos M, Devaney EJ, Vikstrom KL, Herron TJ, Jalife J (2013) Myosin light chain 2-based selection of human ipsc-derived early ventricular cardiac myocytes. Stem Cell Res 11:1335–1347. https://doi.org/10.1016/j.scr.2013.09.003
Boheler KR, Czyz J, Tweedie D, Yang HT, Anisimov SV, Wobus AM (2002) Differentiation of pluripotent embryonic stem cells into cardiomyocytes. Circ Res 91:189–201. https://doi.org/10.1161/01.res.0000027865.61704.32
Burridge PW, Matsa E, Shukla P, Lin ZC, Churko JM, Ebert AD, Lan F, Diecke S, Huber B, Mordwinkin NM, Plews JR, Abilez OJ, Cui B, Gold JD, Wu JC (2014) Chemically defined generation of human cardiomyocytes. Nat Methods 11:855–860. https://doi.org/10.1038/nmeth.2999
Cai W, Zhang J, de Lange WJ, Gregorich ZR, Karp H, Farrell ET, Mitchell SD, Tucholski T, Lin Z, Biermann M, McIlwain SJ, Ralphe JC, Kamp TJ, Ge Y (2019) An unbiased proteomics method to assess the maturation of human pluripotent stem cell-derived cardiomyocytes. Circ Res 125:936–953. https://doi.org/10.1161/CIRCRESAHA.119.315305
Cao N, Liao J, Liu Z, Zhu W, Wang J, Liu L, Yu L, Xu P, Cui C, Xiao L, Yang HT (2011) In vitro differentiation of rat embryonic stem cells into functional cardiomyocytes. Cell Res 21:1316–1331. https://doi.org/10.1038/cr.2011.48
Cao N, Liu Z, Chen Z, Wang J, Chen T, Zhao X, Ma Y, Qin L, Kang J, Wei B, Wang L, Jin Y, Yang HT (2012) Ascorbic acid enhances the cardiac differentiation of induced pluripotent stem cells through promoting the proliferation of cardiac progenitor cells. Cell Res 22:219–236. https://doi.org/10.1038/cr.2011.195
Chirikian O, Goodyer WR, Dzilic E, Serpooshan V, Buikema JW, McKeithan W, Wu H, Li G, Lee S, Merk M, Galdos F, Beck A, Ribeiro AJS, Paige S, Mercola M, Wu JC, Pruitt BL, Wu SM (2021) Crispr/cas9-based targeting of fluorescent reporters to human ipscs to isolate atrial and ventricular-specific cardiomyocytes. Sci Rep 11:3026. https://doi.org/10.1038/s41598-021-81860-x
de Sousa Chuva, Lopes SM, Hassink RJ, Feijen A, van Rooijen MA, Doevendans PA, Tertoolen L, Brutel de la Riviere A, Mummery CL (2006) Patterning the heart, a template for human cardiomyocyte development. Dev Dyn 235:1994–2002. https://doi.org/10.1002/dvdy.20830
Devalla HD, Schwach V, Ford JW, Milnes JT, El-Haou S, Jackson C, Gkatzis K, Elliott DA, de Sousa Chuva, Lopes SM, Mummery CL, Verkerk AO, Passier R (2015) Atrial-like cardiomyocytes from human pluripotent stem cells are a robust preclinical model for assessing atrial-selective pharmacology. EMBO Mol Med 7:394–410. https://doi.org/10.15252/emmm.201404757
Dubois NC, Craft AM, Sharma P, Elliott DA, Stanley EG, Elefanty AG, Gramolini A, Keller G (2011) Sirpa is a specific cell-surface marker for isolating cardiomyocytes derived from human pluripotent stem cells. Nat Biotechnol 29:1011–1018. https://doi.org/10.1038/nbt.2005
Franco D, Markman MM, Wagenaar GT, Ya J, Lamers WH, Moorman AF (1999) Myosin light chain 2a and 2v identifies the embryonic outflow tract myocardium in the developing rodent heart. Anat Rec 254:135–146. https://doi.org/10.1002/(SICI)1097-0185(19990101)254:1%3c135::AID-AR17%3e3.0.CO;2-S
Fu JD, Jiang P, Rushing S, Liu J, Chiamvimonvat N, Li RA (2010) Na+/ca2+ exchanger is a determinant of excitation-contraction coupling in human embryonic stem cell-derived ventricular cardiomyocytes. Stem Cells Dev 19:773–782. https://doi.org/10.1089/scd.2009.0184
Garg P, Garg V, Shrestha R, Sanguinetti MC, Kamp TJ, Wu JC (2018) Human induced pluripotent stem cell–derived cardiomyocytes as models for cardiac channelopathies. Circulation Research 123:224–243. https://doi.org/10.1161/circresaha.118.311209
Gintant G, Burridge P, Gepstein L, Harding S, Herron T, Hong C, Jalife J, Wu JC (2019) Use of human induced pluripotent stem cell-derived cardiomyocytes in preclinical cancer drug cardiotoxicity testing: a scientific statement from the American heart association. Circ Res 125:e75–e92. https://doi.org/10.1161/RES.0000000000000291
He JQ, Ma Y, Lee Y, Thomson JA, Kamp TJ (2003) Human embryonic stem cells develop into multiple types of cardiac myocytes: action potential characterization. Circ Res 93:32–39. https://doi.org/10.1161/01.RES.0000080317.92718.99
Huang J, Zhang M, Zhang P, Liang H, Ouyang K, Yang HT (2016) Coupling switch of p2y-ip3 receptors mediates differential ca(2+) signaling in human embryonic stem cells and derived cardiovascular progenitor cells. Purinergic Signal 12:465–478. https://doi.org/10.1007/s11302-016-9512-9
Huang JJ, Wang YJ, Zhang M, Zhang P, Liang H, Bai HJ, Yu XJ, Yang HT (2017) Functional expression of the ca(2+) signaling machinery in human embryonic stem cells. Acta Pharmacol Sin 38:1663–1672. https://doi.org/10.1038/aps.2017.29
Huber I, Itzhaki I, Caspi O, Arbel G, Tzukerman M, Gepstein A, Habib M, Yankelson L, Kehat I, Gepstein L (2007) Identification and selection of cardiomyocytes during human embryonic stem cell differentiation. FASEB J 21:2551–2563. https://doi.org/10.1096/fj.05-5711com
Kane C, Terracciano CMN (2017) Concise review: Criteria for chamber-specific categorization of human cardiac myocytes derived from pluripotent stem cells. Stem Cells 35:1881–1897. https://doi.org/10.1002/stem.2649
Karbassi E, Fenix A, Marchiano S, Muraoka N, Nakamura K, Yang X, Murry CE (2020) Cardiomyocyte maturation: advances in knowledge and implications for regenerative medicine. Nature Reviews Cardiology 17:341–359. https://doi.org/10.1038/s41569-019-0331-x
Kubalak SW, Miller-Hance WC, O’Brien TX, Dyson E, Chien KR (1994) Chamber specification of atrial myosin light chain-2 expression precedes septation during murine cardiogenesis. J Biol Chem 269:16961–16970. https://doi.org/10.1016/s0021-9258(19)89483-8
Lee JH, Protze SI, Laksman Z, Backx PH, Keller GM (2017) Human pluripotent stem cell-derived atrial and ventricular cardiomyocytes develop from distinct mesoderm populations. Cell Stem Cell 21(179–194):e174. https://doi.org/10.1016/j.stem.2017.07.003
Li B, Yang H, Wang X, Zhan Y, Sheng W, Cai H, Xin H, Liang Q, Zhou P, Lu C, Qian R, Chen S, Yang P, Zhang J, Shou W, Huang G, Liang P, Sun N (2017) Engineering human ventricular heart muscles based on a highly efficient system for purification of human pluripotent stem cell-derived ventricular cardiomyocytes. Stem Cell Res Ther 8:202. https://doi.org/10.1186/s13287-017-0651-x
Li Q, Wang J, Wu Q, Cao N, Yang HT (2020) Perspective on human pluripotent stem cell-derived cardiomyocytes in heart disease modeling and repair. Stem Cells Transl Med 9:1121–1128. https://doi.org/10.1002/sctm.19-0340
Li S, Li M, Liu X, Yang Y, Wei Y, Chen Y, Qiu Y, Zhou T, Feng Z, Ma D, Fang J, Ying H, Wang H, Musunuru K, Shao Z, Zhao Y, Ding Q (2018) Genetic and chemical screenings identify hdac3 as a key regulator in hepatic differentiation of human pluripotent stem cells. Stem Cell Reports 11:22–31. https://doi.org/10.1016/j.stemcr.2018.05.001
Lian X, Hsiao C, Wilson G, Zhu K, Hazeltine LB, Azarin SM, Raval KK, Zhang J, Kamp TJ, Palecek SP (2012) Robust cardiomyocyte differentiation from human pluripotent stem cells via temporal modulation of canonical wnt signaling. Proc Natl Acad Sci U S A 109:E1848-1857. https://doi.org/10.1073/pnas.1200250109
Liang H, Zhang P, Bai HJ, Huang J, Yang HT (2020) Tata box-binding protein-related factor 3 drives the mesendoderm specification of human embryonic stem cells by globally interacting with the tata box of key mesendodermal genes. Stem Cell Res Ther 11:196. https://doi.org/10.1186/s13287-020-01711-w
Liu G, Liu Z, Cao N (2021) Human pluripotent stem cell-based cardiovascular disease modeling and drug discovery. Pflugers Arch. https://doi.org/10.1007/s00424-021-02542-1
Liu Y, Bai H, Guo F, Thai PN, Luo X, Zhang P, Yang C, Feng X, Zhu D, Guo J, Liang P, Xu Z, Yang H, Lu X (2020) Pgc-1alpha activator zln005 promotes maturation of cardiomyocytes derived from human embryonic stem cells. Aging (Albany NY) 12:7411–7430. https://doi.org/10.18632/aging.103088
Ma J, Guo L, Fiene SJ, Anson BD, Thomson JA, Kamp TJ, Kolaja KL, Swanson BJ, January CT (2011) High purity human-induced pluripotent stem cell-derived cardiomyocytes: electrophysiological properties of action potentials and ionic currents. Am J Physiol Heart Circ Physiol 301:H2006-2017. https://doi.org/10.1152/ajpheart.00694.2011
Maltsev VA, Rohwedel J, Hescheler J, Wobus AM (1993) Embryonic stem cells differentiate in vitro into cardiomyocytes representing sinusnodal, atrial and ventricular cell types. Mech Dev 44:41–50. https://doi.org/10.1016/0925-4773(93)90015-p
Maltsev VA, Wobus AM, Rohwedel J, Bader M, Hescheler J (1994) Cardiomyocytes differentiated in vitro from embryonic stem cells developmentally express cardiac-specific genes and ionic currents. Circ Res 75:233–244. https://doi.org/10.1161/01.res.75.2.233
Nelson DO, Lalit PA, Biermann M, Markandeya YS, Capes DL, Addesso L, Patel G, Han T, John MC, Powers PA, Downs KM, Kamp TJ, Lyons GE (2016) Irx4 marks a multipotent, ventricular-specific progenitor cell. Stem Cells 34:2875–2888. https://doi.org/10.1002/stem.2486
O’Brien TX, Lee KJ, Chien KR (1993) Positional specification of ventricular myosin light chain 2 expression in the primitive murine heart tube. Proc Natl Acad Sci U S A 90:5157–5161. https://doi.org/10.1073/pnas.90.11.5157
Pan Z, Ebert A, Liang P (2020) Human-induced pluripotent stem cells as models for rare cardiovascular diseases: from evidence-based medicine to precision medicine. Pflugers Arch. https://doi.org/10.1007/s00424-020-02486-y
Poon EN, Luo XL, Webb SE, Yan B, Zhao R, Wu SCM, Yang Y, Zhang P, Bai H, Shao J, Chan CM, Chan GC, Tsang SY, Gundry RL, Yang HT, Boheler KR (2020) The cell surface marker cd36 selectively identifies matured, mitochondria-rich hpsc-cardiomyocytes. Cell Res 30:626–629. https://doi.org/10.1038/s41422-020-0292-y
Protze SI, Lee JH, Keller GM (2019) Human pluripotent stem cell-derived cardiovascular cells: from developmental biology to therapeutic applications. Cell Stem Cell 25:311–327. https://doi.org/10.1016/j.stem.2019.07.010
Protze SI, Liu J, Nussinovitch U, Ohana L, Backx PH, Gepstein L, Keller GM (2017) Sinoatrial node cardiomyocytes derived from human pluripotent cells function as a biological pacemaker. Nat Biotechnol 35:56–68. https://doi.org/10.1038/nbt.3745
Ruan H, Liao Y, Ren Z, Mao L, Yao F, Yu P, Ye Y, Zhang Z, Li S, Xu H, Liu J, Diao L, Zhou B, Han L, Wang L (2019) Single-cell reconstruction of differentiation trajectory reveals a critical role of ets1 in human cardiac lineage commitment. BMC Biol 17:89. https://doi.org/10.1186/s12915-019-0709-6
Shafaattalab S, Lin E, Christidi E, Huang H, Nartiss Y, Garcia A, Lee J, Protze S, Keller G, Brunham L, Tibbits GF, Laksman Z (2019) Ibrutinib displays atrial-specific toxicity in human stem cell-derived cardiomyocytes. Stem Cell Reports 12:996–1006. https://doi.org/10.1016/j.stemcr.2019.03.011
Uosaki H, Fukushima H, Takeuchi A, Matsuoka S, Nakatsuji N, Yamanaka S, Yamashita JK (2011) Efficient and scalable purification of cardiomyocytes from human embryonic and induced pluripotent stem cells by vcam1 surface expression. PLoS One 6:e23657. https://doi.org/10.1371/journal.pone.0023657
van den Heuvel NH, van Veen TA, Lim B, Jonsson MK (2014) Lessons from the heart: Mirroring electrophysiological characteristics during cardiac development to in vitro differentiation of stem cell derived cardiomyocytes. J Mol Cell Cardiol 67:12–25. https://doi.org/10.1016/j.yjmcc.2013.12.011
Veevers J, Farah EN, Corselli M, Witty AD, Palomares K, Vidal JG, Emre N, Carson CT, Ouyang K, Liu C, van Vliet P, Zhu M, Hegarty JM, Deacon DC, Grinstein JD, Dirschinger RJ, Frazer KA, Adler ED, Knowlton KU, Chi NC, Martin JC, Chen J, Evans SM (2018) Cell-surface marker signature for enrichment of ventricular cardiomyocytes derived from human embryonic stem cells. Stem Cell Reports 11:828–841. https://doi.org/10.1016/j.stemcr.2018.07.007
Wong AO, Gurung B, Wong WS, Mak SY, Tse WW, Li CM, Lieu DK, Costa KD, Li RA, Hajjar RJ (2020) Adverse effects of hydroxychloroquine and azithromycin on contractility and arrhythmogenicity revealed by human engineered cardiac tissues. J Mol Cell Cardiol 153:106–110. https://doi.org/10.1016/j.yjmcc.2020.12.014
Wu SP, Cheng CM, Lanz RB, Wang T, Respress JL, Ather S, Chen W, Tsai SJ, Wehrens XH, Tsai MJ, Tsai SY (2013) Atrial identity is determined by a coup-tfii regulatory network. Dev Cell 25:417–426. https://doi.org/10.1016/j.devcel.2013.04.017
Yamauchi K, Li J, Morikawa K, Liu L, Shirayoshi Y, Nakatsuji N, Elliott DA, Hisatome I, Suemori H (2018) Isolation and characterization of ventricular-like cells derived from nkx2-5(egfp/w) and mlc2v(mcherry/w) double knock-in human pluripotent stem cells. Biochem Biophys Res Commun 495:1278–1284. https://doi.org/10.1016/j.bbrc.2017.11.133
Zhang JZ, Termglinchan V, Shao NY, Itzhaki I, Liu C, Ma N, Tian L, Wang VY, Chang ACY, Guo H, Kitani T, Wu H, Lam CK, Kodo K, Sayed N, Blau HM, Wu JC (2019) A human ipsc double-reporter system enables purification of cardiac lineage subpopulations with distinct function and drug response profiles. Cell Stem Cell 24(802–811):e805. https://doi.org/10.1016/j.stem.2019.02.015
Zhang P, Huang JJ, Ou-Yang KF, Liang H, Li ML, Wang YJ, Yang HT (2020) Minimal contribution of ip3r2 in cardiac differentiation and derived ventricular-like myocytes from human embryonic stem cells. Acta Pharmacol Sin 41:1576–1586. https://doi.org/10.1038/s41401-020-00528-w
Zhang Q, Jiang J, Han P, Yuan Q, Zhang J, Zhang X, Xu Y, Cao H, Meng Q, Chen L, Tian T, Wang X, Li P, Hescheler J, Ji G, Ma Y (2010) Direct differentiation of atrial and ventricular myocytes from human embryonic stem cells by alternating retinoid signals. Cell Research 21:579–587. https://doi.org/10.1038/cr.2010.163
Zhou W, Bos JM, Ye D, Tester DJ, Hrstka S, Maleszewski JJ, Ommen SR, Nishimura RA, Schaff HV, Kim CS, Ackerman MJ (2019) Induced pluripotent stem cell-derived cardiomyocytes from a patient with myl2-r58q-mediated apical hypertrophic cardiomyopathy show hypertrophy, myofibrillar disarray, and calcium perturbations. J Cardiovasc Transl Res 12:394–403. https://doi.org/10.1007/s12265-019-09873-6
Zhou Z, Huang W, Liang J, Szczesna-Cordary D (2016) Molecular and functional effects of a splice site mutation in the myl2 gene associated with cardioskeletal myopathy and early cardiac death in infants. Front Physiol 7:240. https://doi.org/10.3389/fphys.2016.00240
Zwartsen A, de Korte T, Nacken P, de Lange DW, Westerink RHS, Hondebrink L (2019) Cardiotoxicity screening of illicit drugs and new psychoactive substances (nps) in human ipsc-derived cardiomyocytes using microelectrode array (mea) recordings. J Mol Cell Cardiol 136:102–112. https://doi.org/10.1016/j.yjmcc.2019.09.007
Acknowledgments
The authors thank Dr. Kenneth R. Boheler from the Johns Hopkins University for the constructive suggestions and the manuscript editing and also Dr. Ouyang K from Peking University Shenzhen Graduate School for writing the program for Ca2+ transient measurements. We also acknowledge technical support from Xiang Miao, Yu-Jia Zhai, Shu-Yang Yan, and Hua-Jun Bai from Shanghai Institute of Nutrition and Health. We thank WiCell Research Institute for providing the H7 hESCs.
Funding
This work was supported by grants from the National Key R&D Program of China (2017YFA0103700 to H.T.Y. and Q.D.), Strategic Priority Research Program of the CAS (No. XDA16010201 to H.T.Y.), Major Program of Development Fund for Shanghai Zhangjiang National Innovation Demonstration Zone (ZJ2018-ZD-004 to H.T.Y.), and National Natural Science Foundation of China (81520108004, 81470422 to H.T.Y.).
Author information
Authors and Affiliations
Contributions
X.L.L, P.Z., and H.T.Y. conceived and designed the research; X.L.L., and P.Z. collected the data; X.L, S.H., and P.Z. constructed the hESC reporter cell line; X.L.L., P.Z., and H.T.Y. analyzed the data; S.L.R. participated in the AP analysis; X.L.L, P.Z., and H.T.Y. interpreted the experimental data and wrote the manuscript; Q.D. supervised the construction of hESC reporter cell line and provided financial support; H.T.Y. approved the manuscript and provided the funding support.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is submitted to the special issue on Recent Progresses in hiPSCs for Cardiac Repair, Disease Models, and Drug Discovery in Pflügers Archiv-European Journal of Physiology
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Luo, XL., Zhang, P., Liu, X. et al. Myosin light chain 2 marks differentiating ventricular cardiomyocytes derived from human embryonic stem cells. Pflugers Arch - Eur J Physiol 473, 991–1007 (2021). https://doi.org/10.1007/s00424-021-02578-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-021-02578-3