Abstract
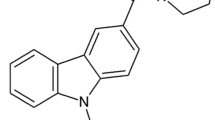
T-type channels are important contributors to the initiation and the maintenance of chronic pain states. Blocking T-type channels is therefore a possible therapeutic strategy for relieving pain. Here, we report the Cav3.2 T-type channel blocking action of a previously reported small organic molecule, KYS-05090S. This compound was able to reduce transiently expressed Cav3.2 currents with low micromolar affinity and mediated a hyperpolarizing shift in half-inactivation potential. KYS-05090S was then tested in models of acute and neuropathic pain. KYS-05090S (10 μg/10 μl delivered intrathecally) significantly reduced acute pain induced by formalin in both the tonic and inflammatory phases. Its antinociceptive effect was not observed when delivered to Cav3.2 null-mice revealing a Cav3.2-dependent mechanism. KYS-05090S also reduced neuropathic pain in a model of partial sciatic nerve injury. Those results indicate that KYS-05090S mediates a potent analgesic effect in inflammatory and neuropathic pain through T-type channel modulation, suggesting that its scaffold could be explored as a new class of analgesic compounds.





Similar content being viewed by others
References
Bladen C, Gadotti VM, Gunduz MG, Berger ND, Simsek R, Safak C, Zamponi GW (2015) 1,4-Dihydropyridine derivatives with T-type calcium channel blocking activity attenuate inflammatory and neuropathic pain. Pflugers Arch - Eur J Physiol 467:1237–1247. doi:10.1007/s00424-014-1566-3
Bladen C, McDaniel SW, Gadotti VM, Petrov RR, Berger ND, Diaz P, Zamponi GW (2015) Characterization of novel cannabinoid based T-type calcium channel blockers with analgesic effects. ACS Chem Neurosci 6:277–287. doi:10.1021/cn500206a
Bourinet E, Alloui A, Monteil A, Barrere C, Couette B, Poirot O, Pages A, McRory J, Snutch TP, Eschalier A et al (2005) Silencing of the Cav3.2 T-type calcium channel gene in sensory neurons demonstrates its major role in nociception. EMBO J 24:315–324. doi:10.1038/sj.emboj.7600515
Cao XH, Byun HS, Chen SR, Pan HL (2011) Diabetic neuropathy enhances voltage-activated Ca2+ channel activity and its control by M4 muscarinic receptors in primary sensory neurons. J Neurochem 119:594–603. doi:10.1111/j.1471-4159.2011.07456.x
Cardenas CG, Del Mar LP, Scroggs RS (1995) Variation in serotonergic inhibition of calcium channel currents in four types of rat sensory neurons differentiated by membrane properties. J Neurophysiol 74:1870–1879
Chen CL, Broom DC, Liu Y, de Nooij JC, Li Z, Cen C, Samad OA, Jessell TM, Woolf CJ, Ma Q (2006) Runx1 determines nociceptive sensory neuron phenotype and is required for thermal and neuropathic pain. Neuron 49:365–377. doi:10.1016/j.neuron.2005.10.036
Choi S, Na HS, Kim J, Lee J, Lee S, Kim D, Park J, Chen CC, Campbell KP, Shin HS (2007) Attenuated pain responses in mice lacking Ca(V)3.2 T-type channels. Genes Brain Behav 6:425–431. doi:10.1111/j.1601-183X.2006.00268.x
De Petrocellis L, Moriello AS, Byun JS, Sohn JM, Lee JY, Vazquez-Romero A, Garrido M, Messeguer A, Zhang FX, Zamponi GW et al (2015) Inhibitory effect of positively charged triazine antagonists of prokinecitin receptors on the transient receptor vanilloid type-1 (TRPV1) channel. Pharmacol Res 99:362–369. doi:10.1016/j.phrs.2015.07.009
Dziegielewska B, Brautigan DL, Larner JM, Dziegielewski J (2014) T-type Ca2+ channel inhibition induces p53-dependent cell growth arrest and apoptosis through activation of p38-MAPK in colon cancer cells. Mol Cancer Res : MCR 12:348–358. doi:10.1158/1541-7786.MCR-13-0485
Finnerup NB, Attal N, Haroutounian S, McNicol E, Baron R, Dworkin RH, Gilron I, Haanpaa M, Hansson P, Jensen TS et al (2015) Pharmacotherapy for neuropathic pain in adults: a systematic review and meta-analysis. Lancet Neurol 14:162–173. doi:10.1016/S1474-4422(14)70251-0
Francois A, Kerckhove N, Meleine M, Alloui A, Barrere C, Gelot A, Uebele VN, Renger JJ, Eschalier A, Ardid D et al (2013) State-dependent properties of a new T-type calcium channel blocker enhance Ca(V)3.2 selectivity and support analgesic effects. Pain 154:283–293. doi:10.1016/j.pain.2012.10.023
Francois A, Laffray S, Pizzoccaro A, Eschalier A, Bourinet E (2014) T-type calcium channels in chronic pain: mouse models and specific blockers. Pflugers Arch - Eur J Physiol 466:707–717. doi:10.1007/s00424-014-1484-4
Gadotti VM, Bladen C, Zhang FX, Chen L, Gündüz MG, Şimşek R, Şafak C, Zamponi GW (2015) Analgesic effect of a broad spectrum dihydropyridine inhibitor of voltage gated calcium channels. Pfulgers Arch
Gadotti VM, You H, Petrov RR, Berger ND, Diaz P, Zamponi GW (2013) Analgesic effect of a mixed T-type channel inhibitor/CB2 receptor agonist. Mol Pain 9:32. doi:10.1186/1744-8069-9-32
Gadotti VM, Zamponi GW (2011) Cellular prion protein protects from inflammatory and neuropathic pain. Mol Pain 7:59. doi:10.1186/1744-8069-7-59
Gavva NR, Tamir R, Qu Y, Klionsky L, Zhang TJ, Immke D, Wang J, Zhu D, Vanderah TW, Porreca F et al (2005) AMG 9810 [(E)-3-(4-t-butylphenyl)-N-(2,3-dihydrobenzo[b][1,4] dioxin-6-yl)acrylamide], a novel vanilloid receptor 1 (TRPV1) antagonist with antihyperalgesic properties. J Pharmacol Exp Ther 313:474–484. doi:10.1124/jpet.104.079855
Gunthorpe MJ, Chizh BA (2009) Clinical development of TRPV1 antagonists: targeting a pivotal point in the pain pathway. Drug Discov Today 14:56–67. doi:10.1016/j.drudis.2008.11.005
Hildebrand ME, Smith PL, Bladen C, Eduljee C, Xie JY, Chen L, Fee-Maki M, Doering CJ, Mezeyova J, Zhu Y et al (2011) A novel slow-inactivation-specific ion channel modulator attenuates neuropathic pain. Pain 152:833–843. doi:10.1016/j.pain.2010.12.035
Honore P, Wismer CT, Mikusa J, Zhu CZ, Zhong C, Gauvin DM, Gomtsyan A, El Kouhen R, Lee CH, Marsh K et al (2005) A-425619 [1-isoquinolin-5-yl-3-(4-trifluoromethyl-benzyl)-urea], a novel transient receptor potential type V1 receptor antagonist, relieves pathophysiological pain associated with inflammation and tissue injury in rats. J Pharm Exp Ther 314:410–421. doi:10.1124/jpet.105.083915
Hylden JL, Wilcox GL (1980) Intrathecal morphine in mice: a new technique. Eur J Pharmacol 67:313–316
Jacus MO, Uebele VN, Renger JJ, Todorovic SM (2012) Presynaptic Cav3.2 channels regulate excitatory neurotransmission in nociceptive dorsal horn neurons. J Neurosci: Off J Soc Neurosci 32:9374–9382. doi:10.1523/JNEUROSCI.0068-12.2012
Jagodic MM, Pathirathna S, Joksovic PM, Lee W, Nelson MT, Naik AK, Su P, Jevtovic-Todorovic V, Todorovic SM (2008) Upregulation of the T-type calcium current in small rat sensory neurons after chronic constrictive injury of the sciatic nerve. J Neurophysiol 99:3151–3156. doi:10.1152/jn.01031.2007
Jagodic MM, Pathirathna S, Nelson MT, Mancuso S, Joksovic PM, Rosenberg ER, Bayliss DA, Jevtovic-Todorovic V, Todorovic SM (2007) Cell-specific alterations of T-type calcium current in painful diabetic neuropathy enhance excitability of sensory neurons. J Neurosci: Off J Soc Neurosci 27:3305–3316. doi:10.1523/JNEUROSCI.4866-06.2007
Jensen TS, Madsen CS, Finnerup NB (2009) Pharmacology and treatment of neuropathic pains. Curr Opin Neurol 22:467–474. doi:10.1097/WCO.0b013e3283311e13
Jung SY, Lee SH, Kang HB, Park HA, Chang SK, Kim J, Choo DJ, Oh CR, Kim YD, Seo JH et al (2010) Antitumor activity of 3,4-dihydroquinazoline dihydrochloride in A549 xenograft nude mice. Bioorg Med Chem Lett 20:6633–6636. doi:10.1016/j.bmcl.2010.09.020
Kang HB, Rim HK, Park JY, Choi HW, Choi DL, Seo JH, Chung KS, Huh G, Kim J, Choo DJ et al (2012) In vivo evaluation of oral anti-tumoral effect of 3,4-dihydroquinazoline derivative on solid tumor. Bioorg Med Chem Lett 22:1198–1201. doi:10.1016/j.bmcl.2011.11.083
Malmberg AB, Basbaum AI (1998) Partial sciatic nerve injury in the mouse as a model of neuropathic pain: behavioral and neuroanatomical correlates. Pain 76:215–222
Mantyh P (2013) Bone cancer pain: causes, consequences, and therapeutic opportunities. Pain 154(Suppl 1):S54–S62. doi:10.1016/j.pain.2013.07.044
Messinger RB, Naik AK, Jagodic MM, Nelson MT, Lee WY, Choe WJ, Orestes P, Latham JR, Todorovic SM, Jevtovic-Todorovic V (2009) In vivo silencing of the Ca(V)3.2 T-type calcium channels in sensory neurons alleviates hyperalgesia in rats with streptozocin-induced diabetic neuropathy. Pain 145:184–195. doi:10.1016/j.pain.2009.06.012
Petruska JC, Napaporn J, Johnson RD, Gu JG, Cooper BY (2000) Subclassified acutely dissociated cells of rat DRG: histochemistry and patterns of capsaicin-, proton-, and ATP-activated currents. J Neurophysiol 84:2365–2379
Rim HK, Cho S, Shin DH, Chung KS, Cho YW, Choi JH, Lee JY, Lee KT (2014) T-type Ca2+ channel blocker, KYS05090 induces autophagy and apoptosis in A549 cells through inhibiting glucose uptake. Molecules 19:9864–9875. doi:10.3390/molecules19079864
Snider WD, McMahon SB (1998) Tackling pain at the source: new ideas about nociceptors. Neuron 20:629–632
Stucky CL, Lewin GR (1999) Isolectin B(4)-positive and -negative nociceptors are functionally distinct. J Neurosci: Off J Soc Neurosci 19:6497–6505
Takahashi T, Aoki Y, Okubo K, Maeda Y, Sekiguchi F, Mitani K, Nishikawa H, Kawabata A (2010) Upregulation of Ca(v)3.2 T-type calcium channels targeted by endogenous hydrogen sulfide contributes to maintenance of neuropathic pain. Pain 150:183–191. doi:10.1016/j.pain.2010.04.022
Todorovic SM, Jevtovic-Todorovic V (2007) Regulation of T-type calcium channels in the peripheral pain pathway. Channels 1:238–245
Wen XJ, Xu SY, Chen ZX, Yang CX, Liang H, Li H (2010) The roles of T-type calcium channel in the development of neuropathic pain following chronic compression of rat dorsal root ganglia. Pharmacology 85:295–300. doi:10.1159/000276981
Yue J, Liu L, Liu Z, Shu B, Zhang Y (2013) Upregulation of T-type Ca2+ channels in primary sensory neurons in spinal nerve injury. Spine 38:463–470. doi:10.1097/BRS.0b013e318272fbf8
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
M’Dahoma, S., Gadotti, V.M., Zhang, FX. et al. Effect of the T-type channel blocker KYS-05090S in mouse models of acute and neuropathic pain. Pflugers Arch - Eur J Physiol 468, 193–199 (2016). https://doi.org/10.1007/s00424-015-1733-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-015-1733-1