Abstract
Purpose
This investigation examined the development of neuromuscular fatigue during a simulated soccer match incorporating a period of extra time (ET) and the reliability of these responses on repeated test occasions.
Methods
Ten male amateur football players completed a 120 min soccer match simulation (SMS). Before, at half time (HT), full time (FT), and following a period of ET, twitch responses to supramaximal femoral nerve and transcranial magnetic stimulation (TMS) were obtained from the knee-extensors to measure neuromuscular fatigue. Within 7 days of the first SMS, a second 120 min SMS was performed by eight of the original ten participants to assess the reliability of the fatigue response.
Results
At HT, FT, and ET, reductions in maximal voluntary force (MVC; −11, −20 and −27%, respectively, P ≤ 0.01), potentiated twitch force (−15, −23 and −23%, respectively, P < 0.05), voluntary activation (FT, −15 and ET, −18%, P ≤ 0.01), and voluntary activation measured with TMS (−11, −15 and −17%, respectively, P ≤ 0.01) were evident. The fatigue response was robust across both trials; the change in MVC at each time point demonstrated a good level of reliability (CV range 6–11%; ICC2,1 0.83–0.94), whilst the responses identified with motor nerve stimulation showed a moderate level of reliability (CV range 5–18%; ICC2,1 0.63–0.89) and the data obtained with motor cortex stimulation showed an excellent level of reliability (CV range 3–6%; ICC2,1 0.90–0.98).
Conclusion
Simulated soccer exercise induces a significant level of fatigue, which is consistent on repeat tests, and involves both central and peripheral mechanisms.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Association football (soccer) is a team based, high-intensity, intermittent-sprint sport typically played over 90 min. However, in certain knockout tournament scenarios (e.g., FIFA World Cup or UEFA Champions League) when a match is tied at 90 min, but requires an outright winner, an additional 30 min period of play termed extra time (ET) is required. Recently, negative impacts of this additional period of play have been shown on technical (Harper et al. 2014) and physical (Penas et al. 2015; Russell et al. 2015) performance, as well as aspects of metabolism and hydration status (Harper et al. 2016a, b, d). These negative consequences are concurrent with the greatest occurrence of contact related injuries during this time (Aoki et al. 2012). Participation in soccer results in high levels of metabolic (Rampinini et al. 2011), mechanical (Akenhead et al. 2013), and perceptual stress (Impellizzeri et al. 2004). The aetiology of soccer-specific fatigue, which manifests transiently during simulated and actual match-play, has been hypothesised to be due to several putative mechanisms including, compromised excitation–contraction coupling (Clarke et al. 2015; Rampinini et al. 2011), depletion of endogenous fuel sources (Bendiksen et al. 2012), ionic disturbances (Bangsbo et al. 2006), and dehydration (Laitano et al. 2014). Despite these investigations, the precise mechanisms of fatigue are yet to be delineated.
Fatigue in soccer has been the subject of several reviews (Bangsbo et al. 2007; Mohr et al. 2005; Nedelec et al. 2012) and experimental study (Andersson et al. 2008; Oliver et al. 2008; Rahnama et al. 2006; Robineau et al. 2012), however, a limited number of investigations have attempted to quantify the neuromuscular fatigue response, with equivocal results (Girard et al. 2015; Marshall et al. 2014; Nybo et al. 2013; Rampinini et al. 2011). Fatigue is classically defined as an exercise-induced reduction in the ability of a muscle or muscle group to generate maximal force (Gandevia 2001), which stems from peripheral and central mechanisms. Peripheral fatigue is the loss in muscle force caused by disturbances in sites at or distal to the neuromuscular junction, whereas central fatigue is defined as a progressive, exercised-induced reduction in the voluntary activation (VA) of muscle (Gandevia 2001). Simulated and actual soccer match-play has been shown to elicit substantial peripheral fatigue (Clarke et al. 2015; Girard et al. 2015; Rampinini et al. 2011) likely attributable to alterations in excitation–contraction coupling. Soccer match-play also results in significant central fatigue; a reduced VA of the knee-extensors (~ 8%) following 90 min of football match-play was first reported by Rampinini et al. (2011). Smaller reductions of ~ 1.5% in VA of the plantar flexors have also been reported following 90 min matches in hot (43 °C) and temperate (~ 20 to 21 °C) environments (Girard et al. 2015; Nybo et al. 2013). However, in these investigations, the post-match fatigue assessments were recorded 30–40 min following the match, a time in which the degree of fatigue would have dissipated. Transcranial magnetic stimulation (TMS) can be used to stimulate neural structures (such as the primary motor cortex) to further investigate the central nervous system responses to exercise, and the presence of a supraspinal contribution to central fatigue (Goodall et al. 2014). Of relevance to soccer, TMS has recently been used to demonstrate how maximal repeated-sprint running exercise elicits central fatigue that is partly attributable to sub-optimal output from the motor cortex (Goodall et al. 2015b). Although these data provide some indication of the responses to repeated-sprint activity, akin to soccer, the use of TMS to examine the pattern of fatigue during soccer-specific exercise has not been investigated. Further research is required to elucidate the aetiology of fatigue during soccer, both during regulation 90 min games, and for tournament scenarios where ET periods are common. The potential accumulation of fatigue incurred by ET might explain the previously observed performance reductions (Harper et al. 2014) and increased injury incidence (Aoki et al. 2012). Furthermore, practitioners working in professional soccer have recently highlighted that understanding fatigue responses following ET performance is an important area for future research (Harper et al. 2016c).
While there is value in studying the mechanisms of neuromuscular fatigue, the usefulness of such study is dependent on the data demonstrating acceptable reliability. Reliability refers to measurement stability when a testing protocol is undertaken repeatedly (Hopkins 2000). Knowledge of measurement reliability for neuromuscular responses over time is important as these data are rarely provided. Accordingly, the primary aim of this study was to investigate neuromuscular fatigue in response to 120 min of simulated soccer-specific exercise. A secondary aim was to investigate the reliability of the fatigue response.
Methods
Participants
Ten males (age 22 ± 3 year; stature 1.83 ± 0.08 m; body mass 79.3 ± 8.0 kg; estimated maximal oxygen uptake [V̇O2max], 56.0 ± 1.0 mL kg−1 min−1) volunteered to participate in the study. All participants had been competitively playing for the previous 2 years in either the university soccer team or for a semi-professional club. The players were training at least twice a week (with additional strength and conditioning sessions) and were involved with at least one competitive fixture; all testing took place in the late off season of the training year. Participants arrived at the laboratory in a rested and hydrated state, having avoided strenuous exercise in the preceding 48 h. Volunteers also refrained from caffeine for 12 h and alcohol for 24 h prior to each trial. Prior to any experimental procedures, written informed consent was obtained from all participants and the study conformed to the latest revision of the Declaration of Helsinki. The Research Ethics Committee at Northumbria University approved all procedures. All of the participants contributed to a companion study which investigated the reliability of metabolic, perceptual, and performance responses (Harper et al. 2016d); while the data were obtained from the same protocol described below, the primary neuromuscular outcome measures in the current study do not overlap with any of the previous analyses.
Experimental design
Two practice visits preceded the main trial, first, to determine V̇O2max and second, for habituation to the neuromuscular measurement tools and demands of the soccer match simulation (SMS; Russell et al. 2011). Eight of the initial ten participants performed a second main trial to determine the reproducibility of the neuromuscular fatigue measurements. Each visit was separated by at least 7 days to ensure full recovery, and was completed on an indoor running track where the environmental conditions remained constant (temperature 19 ± 1 °C; humidity 34 ± 1%). Neuromuscular function and corticospinal excitability were assessed at baseline and then at HT (45 min), FT (90 min), and following ET (120 min).
Procedures
Practice trials
On the first practice, visit participants completed a standardised warm-up (consisting of running, dynamic stretching, and ball dribbling drills) followed by a progressive shuttle run test to exhaustion to estimate V̇O2max (Ramsbottom et al. 1988). All participants reached at least level 12 on the progressive shuttle test and this result was used to determine the intensity of the main trials (Russell et al. 2011). During the second practice, trial participants were habituated with the techniques to assess neuromuscular function, and completed the 120 min SMS protocol (described below).
Main trial procedures
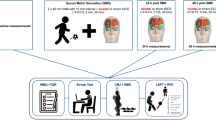
Upon arrival at the laboratory, body mass and stature were recorded (Seca GmbH & Co., Germany) before the consumption of a standardised breakfast (cereals and milk; equating to 10% of participants’ daily calorific intake) and 500 mL of mineral water. Participants then rested for 90 min before baseline neuromuscular function was assessed (described below). Following this assessment, and a standardised warm-up, participants consumed a further 300 mL of water. At HT, 500 mL of water was consumed and at FT, following the recording of all data, participants were given a further 300 mL of water and two 66 g caffeine-free electrolyte gels (IsoGel, High5 Nutrition Ltd., UK). A schematic of the main trial procedure can be seen in Fig. 1.
Schematic of the experimental trial showing when neuromuscular function was assessed at baseline and throughout the soccer match simulation. At each time point, the neuromuscular assessment (NM) involved three knee-extensor maximum voluntary contractions (MVCs) with motor nerve stimulation delivered to the knee-extensors during and 2 s post-MVC to determine voluntary activation and potentiated twitch force. Subsequently, three sets of knee-extensor contractions at varying force levels (100, 75 and 50% MVC) were performed to determine voluntary activation with motor cortex stimulation
Soccer match simulation (SMS)
The SMS involved two 45 min halves of soccer-specific activity separated by a 15 min rest (HT), followed by a 30 min ET period. Each half of the SMS consisted of seven, 4.5 min blocks (3 blocks during each ET period) of intermittent activity, and ball dribbling tests over 20 m. Each 4.5 min block comprised 3 × 3 cycles of intermittent exercise consisting of walks, side steps, dribbles, jogs (40% VO2max), strides (85% VO2max), and maximal intensity sprints over 20 m all in keeping with an audible beep, as previously described and validated by Russell et al. (2011). At FT, following the measurement of neuromuscular function, a 5 min passive recovery period was undertaken before ET. The ET period consisted of two 15 min halves, interspersed by a 2 min break. Due to the inclusion of ET and omission of measuring passing and shooting components, the protocol was a modified version of the previously used SMS, which has been shown to be both valid and reliable when performed over 90 (Russell et al. 2011) and 120 min (Harper et al. 2016d). On completion of the SMS, participants had covered approximately 14.4 km involving 30 dribbles and 30 sprints, which is similar to actual completion of a match requiring ET (Russell et al. 2015). Heart rate (Polar RS400; Polar Electro, Kempele, Finland) and the rating of perceived exertion (RPE, Borg 6–20 scale) were measured throughout the SMS.
Assessment of neuromuscular function
To assess changes in neuromuscular function, force and EMG variables were assessed before and immediately after (within 2.5 min) HT, FT and ET. Maximum voluntary contraction (MVC) force was determined from three maximal, 3 s contractions. Femoral nerve stimulation was delivered during each of the contractions and an additional stimulus was delivered at rest, ~2 s after the superimposed stimulus, to determine voluntary activation (Merton 1954) and the potentiated quadriceps twitch force (Q tw,pot). Single pulse TMS was delivered during brief (~3 s) maximal and submaximal voluntary contractions for the measurement of voluntary activation (VATMS). Each set of contractions comprised 100, 75, and 50% MVC efforts separated by ~5 s of rest; the contraction sets were repeated three times with 15 s between each set and mean values for outcome variables were used for analysis. During all contractions, visual feedback of the target force was provided via a computer monitor.
Force and EMG recordings
A calibrated load cell (MuscleLab force sensor 300, Ergotest Technology, Norway) was used to measure knee-extensor force (N) during voluntary and evoked contractions. The load cell was fixed to a custom-built chair and connected to a non-compliant cuff attached around the participant’s right leg, superior to the malleoli. Participants sat upright in the chair with the hips and knees at 90° of flexion and were instructed to grasp the handles on the side of the chair for support during contractions. EMG activity was recorded from the rectus femoris (RF), vastus lateralis (VL), and biceps femoris (BF). Surface Ag/AgCl electrodes (Kendall H87PG/F, Covidien, Mansfield, MA, USA) were placed 2 cm apart over the muscle bellies and a reference electrode was placed over the ipsilateral patella. Electrode placement was marked with permanent ink to ensure a consistent placement between each assessment point. The electrodes were used to record the compound muscle action potential (M-wave) elicited by electrical stimulation of the femoral nerve, motor evoked potentials (MEP) elicited by TMS, and root-mean-square amplitude for maximal voluntary contractions (rmsEMG). The rmsEMG values in the RF and VL were normalised to the corresponding maximal values at each time point (rmsEMG M−1). EMG Signals were amplified (gain × 1000 for EMG and × 300 for force, CED 1902, Cambridge Electronic Design, UK), band-pass filtered (EMG only: 20–2000 Hz), digitised (4 kHz; CED 1401, Cambridge Electronic Design, UK), acquired, and analysed offline (Spike2 v7.12, Cambridge Electronic Design, UK).
Femoral nerve stimulation
Single, electrical stimuli (200 µs pulse width) were delivered to the right femoral nerve through surface electrodes (CF3200, Nidd Valley Medical Ltd., North Yorkshire, UK) using a constant-current stimulator (DS7AH, Digitimer Ltd., Welwyn Garden City, Hertfordshire, UK). In line with the previous investigations from our laboratory (Goodall et al. 2015a, b; Thomas et al. 2015), the cathode was positioned over the nerve, high in the femoral triangle, whilst the anode was placed midway between the greater trochanter and the iliac crest. Single stimuli were delivered to the relaxed muscle beginning at 40 mA, the intensity was increased by 20 mA until a plateau occurred in twitch amplitude and M-wave (M max). Supramaximal stimulation was delivered by increasing the final stimulator output intensity by a further 30% (mean current 190 ± 30 mA). The positions of the stimulating electrodes were marked with indelible ink to ensure consistent placement during the times of assessment. At each time point, muscle contractility was assessed for the peripherally-derived resting twitches as twitch amplitude (Q tw,pot: the maximum twitch tension) and membrane excitability was inferred from the peak-to-peak amplitude and area of the electrically-evoked M max.
Transcranial magnetic stimulation
Single pulse TMS was delivered using a concave double cone coil (110 mm diameter; maximum output 1.4 T), powered by a mono-pulse magnetic stimulator (Magstim 200, The Magstim Company Ltd., Whitland, UK). The coil was held over the vertex to stimulate the left hemisphere (induced current = postero-anterior), in the optimal position to elicit a large MEP in the knee-extensors (RF) and a small MEP in the antagonist (BF). The optimal coil position was marked on the scalp with indelible ink to ensure a reproducible site of stimulation. To measure VATMS and corticospinal excitability, the stimulator output was set to produce the largest possible superimposed twitch force (SIT) during a 50% MVC contraction. The stimulation intensity (62 ± 6%) elicited a large MEP in the RF (pooled average of ~66% of M max area during contractions ≥50% MVC) indicating the TMS stimulus activated a high proportion of knee-extensor motor units, while causing only a small MEP in the antagonist (~20% of RF MEP during knee-extensor contractions). At all-time points corticospinal responsiveness was quantified as the MEP elicited during a 50% MVC expressed relative to the M max elicited at the same contraction strength. We ensured that participants received clear instructions to achieve a plateau in force when contracting at varying force levels whilst receiving TMS (Gruet et al. 2013).
Data analysis
Voluntary activation measured through stimulation of the femoral nerve was quantified using the twitch interpolation technique (Merton 1954). Voluntary activation was quantified by comparing the amplitude of the SIT during MVC with the amplitude of the resting Q tw,pot elicited 2 s post-MVC: motor nerve VA (%) = (1−[SIT/Q tw,pot] × 100). Assessment of VATMS was made by measuring the force responses from motor cortex stimulation during submaximal and maximal contractions. Corticospinal excitability is known to increase during voluntary contraction; thus, it was necessary to estimate the amplitude of the resting twitch (ERT) through linear regression of the SIT force evoked by TMS during the maximal and submaximal contractions. Regression analysis confirmed the linearity of this relationship at all assessment points (mean r 2 ≥ 0.91). Subsequently, VATMS (%) was quantified using the equation: (1−[SIT/ERT] × 100). The peak-to-peak amplitude and area of evoked MEPs and M max were calculated offline.
Statistical analysis
Data are presented as means ± SD in the text and figures. One-way repeated-measures analysis of variance (ANOVA) was used to assess changes in all outcome measures. Assumptions of sphericity were explored and controlled for all variables using the Greenhouse–Geisser adjustment, where appropriate. Where significant main effects were detected and pairwise comparisons between time-points are reported, the Tukey method was used to adjust for multiple comparisons (Graphpad Prism, v5.04, La Jolla, CA, USA); statistical significance was assumed at P ≤ 0.05. Effect sizes for selected pairwise comparisons were determined using Cohen’s d. To determine absolute and relative reliability of the fatigue response, typical error expressed as a coefficient of variation (CV, %; Hopkins 2000) and the intraclass correlation coefficient (ICC2,1; SPSS, v22, IBM, Chicago, USA) were determined at each time point, respectively. A CV of ≤5% and an ICC of >0.75 was considered excellent reliability, whereas a CV of 5–10% and ICC 0.60–0.70 were considered good reliability (Duffield et al. 2004; Fleiss 1986).
Results
Heart rate was increased throughout the SMS (F 3,27 = 223.50, P < 0.001) with values at HT, FT, and ET being higher than baseline (all P < 0.001). In addition, RPE increased throughout the protocol (F 3,27 = 20.83, P < 0.001) with values at HT, FT, and ET being higher than that recorded after the first block of the protocol (all P ≤ 0.003). The increase observed at ET was greater than HT (P < 0.001) and FT (P = 0.034) (Table 1).
Neuromuscular function
MVC reduced throughout the SMS (F 3,27 = 18.54, P < 0.001); with HT (−11 ± 6%, P < 0.01; d = 0.86), FT (−20 ± 10%, P < 0.001; d = 1.54), and ET (−27 ± 11%, P < 0.001; d = 1.89) values being lower than baseline (682 ± 92 N). The reduction observed at FT was greater than HT (P = 0.012; d = 0.68) and after ET was greater than FT (P = 0.041; d = 0.47) (Fig. 2a). The reduced MVC was accompanied by significant reductions in Qtw,pot (F 3,27 = 4.03, P = 0.002) indicative of peripheral fatigue. The decline in Qtw,pot amplitude from baseline (189 ± 92 N) was significant at HT (−15 ± 14%, P = 0.019; d = 1.22), but there were no further reductions at FT (−23 ± 15%, vs. HT, P = 0.376; d = 0.51) or ET (−23 ± 19%, vs. HT, P = 0.366) (Fig. 2b). The reduced Q tw,pot amplitude was accompanied by changes in some within-twitch variables, namely contraction and half relaxation time were reduced from HT onwards (Table 1). In conjunction with substantial peripheral fatigue, central fatigue was evident in the form of reductions in VA measured with motor nerve (F 3,27 = 8.02, P < 0.001) and motor cortex stimulation (F 3,27 = 16.10, P < 0.001). In comparison to baseline (93 ± 4%), VA was reduced at FT (−15 ± 15%, P < 0.01; d = 1.56) and following ET (−18 ± 18%, P < 0.001; d = 1.75). The reduction in VA following ET was greater than HT (P = 0.048; d = 0.95) but not FT (Fig. 2c). VATMS was reduced from baseline (93 ± 3%) at HT (−11 ± 8%, P < 0.01; d = 1.75), FT (−15 ± 7%, P < 0.001; d = 2.46) and following ET (−17 ± 9%, P < 0.001; d = 2.33). The reduction in VATMS was not different between HT vs. FT or ET (Fig. 2c). The M max and corticospinal excitability did not change at any time point in both the RF and VL (Table 1). The VL rmsEMG M−1 was unchanged at any time point (F 3,27 = 0.77, P = 0.518); however, the RF rmsEMG M−1 was reduced (F 3,27 = 4.12, P = 0.016); specifically, the RF rmsEMG M−1 was reduced following ET vs. pre (P = 0.039; Table 1).
Maximum voluntary contraction (a), potentiated knee-extensor twitch force (b) and voluntary activation measured with motor nerve (VA, white dot symbol), and motor cortical (VATMS) stimulation (c) at pre-exercise, half time (HT), full time (FT), and following extra time (ET). *P < 0.05 vs. the pre-exercise value, † P < 0.05 vs. HT, ‡ P < 0.05 vs. FT. Values are means ± SD for 10 participants
Reliability data
Reliability data are summarised in Table 2. An excellent level of reliability was evident for all measures of neuromuscular function pre-exercise (CV range 1.6–5.9%; ICC2,1 range 0.87–0.91, Table 2). The fatigue response was robust across both trials; specifically, the changes in MVC at each time point demonstrated a good-to-excellent level of reliability (CV range 6.3–10.8%; ICC2,1 range 0.83–0.94). The fatigue response identified with motor nerve stimulation showed a good-to-moderate level of reliability (CV range 5.2–17.8%; ICC2,1 range 0.63–0.89) and the data obtained with motor cortex stimulation showed an excellent level of reliability (CV range 3.0–5.7%; ICC2,1 range 0.90–0.97).
Discussion
The primary aim of this study was to investigate the development of neuromuscular fatigue during a 120 min soccer match simulation. Our data demonstrate that 90 min of simulated soccer elicits reductions in the force generating capabilities of the knee-extensors, and this fatigue is a combination of both central and peripheral factors. An additional 30 min period of extra-time induced further fatigue that was primarily of central origin. A secondary aim of the study was to assess the consistency of fatigue development on repeat trials of the 120 min SMS. The development of fatigue was reliable across the two trials with the most variable responses noted following the ET period. Collectively, these data are the first to profile the neuromuscular fatigue response to 120 min of soccer-specific exercise and can help to explain the previously reported reductions in technical performance and physical performance that have been shown to occur during this extended period.
The development of fatigue throughout 120 min of soccer simulation was progressive, with decrements in the ability to generate maximum force evident at successive time-points. Knee-extensor MVC, decrements in which are considered as a global measure of fatigue involving peripheral and central components, was reduced by 11% after 45 min of the simulated match. After 90 min, the ability to generate maximal force was further reduced, and this reduction in strength was similar to the results of simulated and actual intermittent exercise performance (~15%; Clarke et al. 2015; Robineau et al. 2012), but larger than others (Andersson et al. 2008; Ascensao et al. 2008; Ispirlidis et al. 2008; Rampinini et al. 2011; Thorlund et al. 2009). Extra time elicited further reductions in MVC compared to FT (Fig. 2a), a finding which might offer some insight as to why technical performance and injury risk are also known to be affected during this period (Aoki et al. 2012; Harper et al. 2014). In a separate investigation, the loss in maximal force generating capacity of the knee-extensors following the performance of a simulated protocol was not recovered 72 h post-exercise (Thomas et al. 2017). Specifically, the MVC reduction in that study at FT was 16%, similar to that of this study (20%), but following ET, this reduction was further exacerbated (27%). Thus, the fatigue observed following the ET protocol is likely to have persisted for several days’ post-exercise.
The impairment in maximal force production was accompanied by reductions in the Q tw,pot, demonstrative of a contribution from peripheral mechanisms of fatigue (Fig. 2b). The Q tw,pot was reduced from baseline by 15% at HT, and thereafter, no further reduction was observed at FT or ET demonstrating a plateau in the peripheral fatigue response. Such a plateauing of the peripheral fatigue response has previously been demonstrated following self-paced isokinetic exercise (Froyd et al. 2013), intermittent high-intensity cycling (Decorte et al. 2012), and repeated-sprint exercise (Goodall et al. 2015b; Hureau et al. 2014). In line with this study, these previous investigations show a similar biphasic pattern of peripheral fatigue development, whereby most of the decrements in muscle function are manifest early in the exercise bout and are then small thereafter. Such a regulated development of peripheral fatigue can be explained by the recently proposed model based on task-dependency (Thomas et al. 2016). During the first half of the SMS, participants would have met the exercise demand by preferentially exhausting the higher threshold motor units, which are most susceptible to fatigue and change in response to peripheral stimulation. The remaining, smaller degree of fatigue observed at FT and ET was likely attributable to change in the more fatigue-resistant motor units, which exert a smaller reduction in the peripheral twitch but also reduce physical performance (Harper et al. 2016a, d). As with many investigations, the unchanged M max values (Table 1) throughout exercise suggest maintenance of sarcolemmal excitability and a preserved neuromuscular propagation of the action potential. Thus, the peripheral fatigue elicited by simulated soccer performance was likely related to disturbances in the process of excitation–contraction coupling. Specifically, impairments to intracellular Ca2+ regulation in the sarcoplasmic reticulum might reduce Ca2+ sensitivity, leading to a reduction in mechanical output and such muscle fatigue (MacIntosh et al. 2012).
A significant development of central fatigue was also observed, voluntary activation measured with motor nerve stimulation was reduced from baseline throughout the protocol, confirming the previous work showing competitive soccer match-play elicits central fatigue (Rampinini et al. 2011). Following 90 min of match-play reductions in VA of <2% (Girard et al. 2015; Nybo et al. 2013) and ~8% (Rampinini et al. 2011) have been previously reported which is less than observed in this study (~16%, Fig. 2c). A likely explanation for the lack of fatigue in these aforementioned studies is partly due to the investigation of different muscle groups, and the timing of post-exercise measures which might allow some aspects of central fatigue to dissipate (Taylor et al. 1996). In this study, we also quantified VA using TMS of the motor cortex; reductions in VA measured with TMS indicate that some of the observed central fatigue is attributable to supraspinal factors (Gandevia 2001). There was a significant reduction in VA measured with TMS, indicating a reduced capacity for the motor cortex to drive the knee-extensors during, and immediately following 120 min of soccer-specific exercise (Fig. 2c). Over the 120 min simulated soccer match, central fatigue tended to be exacerbated, and this duration-dependent contribution of central processes to fatigue is broadly evident across a range of exercise modes (Lepers et al. 2002; Place et al. 2004; Thomas et al. 2015). In this study, there was a pattern of a progressive decrease in voluntary activation across 120 min of the SMS (pre vs. HT; HT vs. ET), which provides further evidence that central fatigue becomes progressively more limiting as the exercise duration extends.
It is, perhaps, surprising that the ability to produce maximal knee-extensor force dropped following the period of ET compared to FT, but the period of ET did not induce any additional reductions in the Q tw,pot or voluntary activation. The effect sizes for the change in both measurements of VA, and VATMS, between FT and ET were small (d = 0.24 and 0.21), whereas the Q tw,pot showed no effect at all (d = 0.01). Thus, we consider it likely that the additional reductions in MVC following ET are related to central fatigue, which were not detectable by the measurement tools of the study. Taken together, these data support the previous conclusions regarding central fatigue and soccer performance (Rampinini et al. 2011) and, in part, can offer an explanation for the reduced technical and physical performance (Harper et al. 2016b, 2014), and increased risk of injury (Aoki et al. 2012), known to occur during ET.
To substantiate the neuromuscular fatigue responses observed in this study, it is necessary to evaluate the magnitude of change against the reliability of the measurements. Due to there being no preferred, or single statistical approach, the evaluation of measurement reliability is somewhat problematic (Hopkins 2000). In this regard, and in line with other investigations evaluating reliability of responses from the knee-extensors (Bachasson et al. 2013; Rainoldi et al. 2001), we used two approaches to evaluate measurement reliability, the CV and ICC, which provide an absolute and relative assessment, respectively. An excellent level of reliability was evident for measures of neuromuscular function pre-exercise (Table 2), which is in line with the previous work from our laboratory (Goodall et al. 2015b; Thomas et al. 2015), and importantly, enabled us to detect significant changes throughout the soccer-specific exercise. Similar reliability coefficients have been demonstrated in unfatigued states for both upper (Lee et al. 2008; Madsen 1996; Taylor et al. 1996) and lower limb (Amann et al. 2013; Bachasson et al. 2013; Place et al. 2007; Todd et al. 2004) muscle groups, but the reliability of the fatigue response following locomotor exercise is unknown. The fatigue response was consistent across repeated trials of the SMS, though the variability in the response tended to increase with exercise duration, with most variable responses found at the ET assessment point. The change in MVC at each time point demonstrated a good level of reliability (CV range 6.3–10.8%). The fatigue response identified with peripheral stimulation showed a moderate level of reliability (CV range 5.2–17.8%), whilst the data obtained with motor cortex stimulation showed an excellent level of reliability (CV range 3.0–5.7%). As such, our results demonstrate the fatigue response to the SMS is consistent on repeated trials under the present testing conditions. Furthermore, these results are important for future investigations as they could be used to calculate appropriate sample sizes and ascertain worthwhile changes for the variables studied during this mode of exercise.
Limitations and future directions
There are some limitations worthy of consideration in the current study. Most important is the performance of a simulated activity and the level of ecological validity. Participants covered ~14 km during the SMS, which is in line with the distance covered during an actual game (Russell et al. 2015); however, the prescribed nature of the current, and other simulated protocols, differs to that of a real match scenario. Participants exercised to the sound of audio cues throughout the SMS, whereas the intensity of an actual game would fluctuate according to individual motivation and physiological capacity beyond that assessed by a simple aerobic capacity test, and hence potentially impact the fatigue response. Moreover, participants knew that they were partaking in a 120 min exercise protocol, not a 90 min performance then an unbeknown period of ET. Ergometer-based investigations have shown that the physiological and perceptual responses to exercise are different when the duration is known vs. unknown (Baden et al. 2005; Eston et al. 2012). Notwithstanding, the SMS protocol does serve as a valid laboratory based stimulus that allows the assessment of demands akin to soccer (Russell et al. 2011), and the strict control of the activity profile affords a more reproducible exercise stimulus compared to the variable nature of competitive soccer (Carling et al. 2016). To address these limitations, the neuromuscular fatigue response should be determined following actual match-play, and in a way that the period of ET can be blinded. As with all neuromuscular fatigue investigations, we are aware that aspects of corticospinal function have been shown to recover within 1 min following exercise (Taylor et al. 1996). Thus, the present experimental design might not have elucidated the full extent of central fatigue elicited by the SMS. However, our measurement methods were consistent at each time point and the finding that central fatigue was evident at all-time-points demonstrates the robust and reliable nature of the data.
Conclusion
Fatigue develops throughout 120 min of simulated soccer, which is apparent as early as HT, and continues to increase until the cessation of exercise. Both peripheral and central processes contribute to the fatigue observed during simulated soccer, with the additional fatigue induced by a period of ET primarily of central origin. Importantly, the development of fatigue was reliable across repeated trials, with the most variable responses noted following the ET period. These data help to explain the reductions in technical and physical performance that have been shown to occur during this extended period of play.
Abbreviations
- ANOVA:
-
Analysis of variance
- BF:
-
Biceps femoris
- CV:
-
Coefficient of variation
- EMG:
-
Electromyography
- ERT:
-
Estimated resting twitch
- ET:
-
Extra time
- FT:
-
Full time
- HT:
-
Half time
- ICC:
-
Intraclass correlation coefficient
- M max :
-
Maximal M-wave
- MVC:
-
Maximum voluntary contraction
- MEP:
-
Motor evoked potential
- Q tw,pot :
-
Potentiated knee-extensor twitch force
- RF:
-
Rectus femoris; rms, root-mean-squared
- SIT:
-
Superimposed twitch
- SMS:
-
Soccer match simulation
- TMS:
-
Transcranial magnetic stimulation
- VA:
-
Voluntary activation measured using motor nerve stimulation
- VATMS :
-
Voluntary activation measured using motor cortex stimulation
- VL:
-
Vastus lateralis
- VO2max :
-
Maximal oxygen uptake
References
Akenhead R, Hayes PR, Thompson KG, French D (2013) Diminutions of acceleration and deceleration output during professional football match play. J Sci Med Sport 16:556–561
Amann M, Goodall S, Twomey R, Subudhi AW, Lovering AT, Roach RC (2013) AltitudeOmics: on the consequences of high-altitude acclimatization for the development of fatigue during locomotor exercise in humans. J Appl Physiol 115:634–642
Andersson H, Raastad T, Nilsson J, Paulsen G, Garthe I, Kadi F (2008) Neuromuscular fatigue and recovery in elite female soccer: effects of active recovery. Med Sci Sports Exerc 40: 372–380
Aoki H, O’Hata N, Kohno T, Morikawa T, Seki J (2012) A 15-year prospective epidemiological account of acute traumatic injuries during official professional soccer league matches in Japan. Am J Sports Med 40:1006–1014
Ascensao A, Rebelo A, Oliveira E, Marques F, Pereira L, Magalhaes J (2008) Biochemical impact of a soccer match—analysis of oxidative stress and muscle damage markers throughout recovery. Clin Biochem 41:841–851
Bachasson D, Millet GY, Decorte N, Wuyam B, Levy P, Verges S (2013) Quadriceps function assessment using an incremental test and magnetic neurostimulation: a reliability study. J Electromyog Kinesiol 23:649–658
Baden DA, McLean TL, Tucker R, Noakes TD, St Clair Gibson A (2005) Effect of anticipation during unknown or unexpected exercise duration on rating of perceived exertion, affect, and physiological function. Br J Sports Med 39: 742–746
Bangsbo J, Mohr M, Krustrup P (2006) Physical and metabolic demands of training and match-play in the elite football player. J Sports Sci 24:665–674
Bangsbo J, Iaia FM, Krustrup P (2007) Metabolic response and fatigue in soccer. Int J Sports Physiol Perf 2:111–127
Bendiksen M, Bischoff R, Randers MB, Mohr M, Rollo I, Suetta C, Bangsbo J, Krustrup P (2012) The Copenhagen Soccer Test: physiological response and fatigue development. Med Sci Sports Exerc 44:1595–1603
Carling C, Bradley P, McCall A, Dupont G (2016) Match-to-match variability in high-speed running activity in a professional soccer team. J Sports Sci 34:2215–2223
Clarke N, Farthing JP, Lanovaz JL, Krentz JR (2015) Direct and indirect measurement of neuromuscular fatigue in Canadian football players. Appl Physiol Nutr Metab 40:464–473
Decorte N, Lafaix PA, Millet GY, Wuyam B, Verges S (2012) Central and peripheral fatigue kinetics during exhaustive constant-load cycling. Scand J Med Sci Sports 22:381–391
Duffield R, Dawson B, Pinnington HC, Wong P (2004) Accuracy and reliability of a Cosmed K4b2 portable gas analysis system. J Sci Med Sport 7:11–22
Eston R, Stansfield R, Westoby P, Parfitt G (2012) Effect of deception and expected exercise duration on psychological and physiological variables during treadmill running and cycling. Psychophysiol 49: 462–469
Fleiss J (1986) The design and analysis of clinical experiments. Wiley, New York
Froyd C, Millet GY, Noakes TD (2013) The development of peripheral fatigue and short-term recovery during self-paced high-intensity exercise. J Physiol 591:1339–1346
Gandevia SC (2001) Spinal and supraspinal factors in human muscle fatigue. Physiol Rev 81:1725–1789
Girard O, Nybo L, Mohr M, Racinais S (2015) Plantar flexor neuromuscular adjustments following match-play football in hot and cool conditions. Scand J Med Sci Sports 25(Suppl 1):154–163
Goodall S, Howatson G, Romer L, Ross E (2014) Transcranial magnetic stimulation in sport science: a commentary. Eur J Sport Sci 14(Suppl 1):S332–S340
Goodall S, Charlton K, Hignett C, Prichard J, Barwood M, Howatson G, Thomas K (2015a) Augmented supraspinal fatigue following constant-load cycling in the heat. Scand J Med Sci Sports 25(Suppl 1):164–172
Goodall S, Charlton K, Howatson G, Thomas K (2015b) Neuromuscular fatigability during repeated-sprint exercise in male athletes. Med Sci Sports Exerc 47: 528–536
Gruet M, Temesi J, Rupp T, Millet GY, Verges S (2013) Effect of different approaches to target force on transcranial magnetic stimulation responses. Muscle Nerve 48:430–432
Harper LD, West DJ, Stevenson E, Russell M (2014) Technical performance reduces during the extra-time period of professional soccer match-play. PLoS One 9:e110995
Harper LD, Briggs MA, McNamee G, West DJ, Kilduff LP, Stevenson E, Russell M (2016a) Physiological and performance effects of carbohydrate gels consumed prior to the extra-time period of prolonged simulated soccer match-play. J Sci Med Sport 19:509–514
Harper LD, Clifford T, Briggs MA, McNamee G, West DJ, Stevenson E, Russell M (2016b) The effects of 120 min of simulated match play on indices of acid-base balance in professional academy soccer players. J Strength Cond Res 30:1517–1524
Harper LD, Fothergill M, West DJ, Stevenson E, Russell M (2016c) Practitioners’ perceptions of the soccer extra-time period: implications for future research. PLoS One 11:e0157687
Harper LD, Hunter R, Parker P, Goodall S, Thomas K, Howatson G, West DJ, Stevenson E, Russell M (2016d) Test-retest reliability of physiological and performance responses to 120 min of simulated soccer match-play. J Strength Cond Res 30:3178–3186
Hopkins WG (2000) Measures of reliability in sports medicine and science. Sports Med 30:1–15
Hureau TJ, Olivier N, Millet GY, Meste O, Blain GM (2014) Exercise performance is regulated during repeated sprints to limit the development of peripheral fatigue beyond a critical threshold. Exp Physiol 99:951–963
Impellizzeri FM, Rampinini E, Coutts AJ, Sassi A, Marcora SM (2004) Use of RPE-based training load in soccer. Med Sci Sports Exerc 36: 1042–1047
Ispirlidis I, Fatouros IG, Jamurtas AZ, Nikolaidis MG, Michailidis I, Douroudos I, Margonis K, Chatzinikolaou A, Kalistratos E, Katrabasas I, Alexiou V, Taxildaris K (2008) Time-course of changes in inflammatory and performance responses following a soccer game. Clin J Sport Med 18:423–431
Laitano O, Luiz Runco J, Baker L (2014) Hydration science and strategies in football. Sports Sci Exerc 27: 1–7
Lee M, Gandevia SC, Carroll TJ (2008) Cortical voluntary activation can be reliably measured in human wrist extensors using transcranial magnetic stimulation. Clin Neurophysiol 119:1130–1138
Lepers R, Maffiuletti NA, Rochette L, Brugniaux J, Millet GY (2002) Neuromuscular fatigue during a long-duration cycling exercise. J Appl Physiol 92:1487–1493
MacIntosh BR, Holash RJ, Renaud JM (2012) Skeletal muscle fatigue–regulation of excitation-contraction coupling to avoid metabolic catastrophe. J Cell Sci 125:2105–2114
Madsen OR (1996) Torque, total work, power, torque acceleration energy and acceleration time assessed on a dynamometer: reliability of knee and elbow extensor and flexor strength measurements. Eur J Appl Physiol 74:206–210
Marshall PW, Lovell R, Jeppesen GK, Andersen K, Siegler JC (2014) Hamstring muscle fatigue and central motor output during a simulated soccer match. PLoS One 9:e102753
Merton PA (1954) Voluntary strength and fatigue. J Physiol 123:553–564
Mohr M, Krustrup P, Bangsbo J (2005) Fatigue in soccer: a brief review. J Sports Sci 23:593–599
Nedelec M, McCall A, Carling C, Legall F, Berthoin S, Dupont G (2012) Recovery in soccer: part I - post-match fatigue and time course of recovery. Sports Med 42:997–1015
Nybo L, Girard O, Mohr M, Knez W, Voss S, Racinais S (2013) Markers of muscle damage and performance recovery after exercise in the heat. Med Sci Sports Exerc 45: 860–868
Oliver J, Armstrong N, Williams C (2008) Changes in jump performance and muscle activity following soccer-specific exercise. J Sports Sci 26:141–148
Penas CL, Dellal AO, Owen AL, Gomez-Ruano MA (2015) The influence of the extra-time period on physical performance in elite soccer. Int J Perf Anal Sport 15:830–839
Place N, Lepers R, Deley G, Millet GY (2004) Time course of neuromuscular alterations during a prolonged running exercise. Med Sci Sports Exerc 36: 1347–1356
Place N, Maffiuletti NA, Martin A, Lepers R (2007) Assessment of the reliability of central and peripheral fatigue after sustained maximal voluntary contraction of the quadriceps muscle. Muscle Nerve 35:486–495
Rahnama N, Lees A, Reilly T (2006) Electromyography of selected lower-limb muscles fatigued by exercise at the intensity of soccer match-play. J Electromyog Kinesiol 16:257–263
Rainoldi A, Bullock-Saxton JE, Cavarretta F, Hogan N (2001) Repeatability of maximal voluntary force and of surface EMG variables during voluntary isometric contraction of quadriceps muscles in healthy subjects. J Electromyog Kinesiol 11:425–438
Rampinini E, Bosio A, Ferraresi I, Petruolo A, Morelli A, Sassi A (2011) Match-related fatigue in soccer players. Med Sci Sports Exerc 43: 2161–2170
Ramsbottom R, Brewer J, Williams C (1988) A progressive shuttle run test to estimate maximal oxygen uptake. Br J Sports Med 22:141–144
Robineau J, Jouaux T, Lacroix M, Babault N (2012) Neuromuscular fatigue induced by a 90-minute soccer game modeling. J Strength Cond Res 26:555–562
Russell M, Rees G, Benton D, Kingsley M (2011) An exercise protocol that replicates soccer match-play. Int J Sports Med 32:511–518
Russell M, Sparkes W, Northeast J, Kilduff LP (2015) Responses to a 120 min reserve team soccer match: a case study focusing on the demands of extra time. J Sports Sci 33:2133–2139
Taylor JL, Butler JE, Allen GM, Gandevia SC (1996) Changes in motor cortical excitability during human muscle fatigue. J Physiol 490:519–528
Thomas K, Goodall S, Stone M, Howatson G, St Clair Gibson A, Ansley L (2015) Central and peripheral fatigue in male cyclists after 4-, 20-, and 40-km time trials. Med Sci Sports Exerc 47: 537–546
Thomas K, Elmeua M, Howatson G, Goodall S (2016) Intensity-dependent contribution of neuromuscular fatigue after constant-load cycling. Med Sci Sports Exerc 48: 1751–1760
Thomas K, Dent J, Howatson G, Goodall S (2017) Etiology and recovery of neuromuscular fatigue following simulated soccer match-play. Med Sci Sports Exerc (in press)
Thorlund JB, Aagaard P, Madsen K (2009) Rapid muscle force capacity changes after soccer match play. Int J Sports Med 30:273–278
Todd G, Gorman RB, Gandevia SC (2004) Measurement and reproducibility of strength and voluntary activation of lower-limb muscles. Muscle Nerve 29:834–842
Acknowledgements
We thank Mr. Gareth Wooley for the assistance during data collection. This work was funded and conducted in its entirety at Northumbria University, Newcastle, UK.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This project did not receive any funding and has no conflicts of interest to report.
Additional information
Communicated by Peter Krustrup.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Goodall, S., Thomas, K., Harper, L.D. et al. The assessment of neuromuscular fatigue during 120 min of simulated soccer exercise. Eur J Appl Physiol 117, 687–697 (2017). https://doi.org/10.1007/s00421-017-3561-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-017-3561-9