Abstract
Acute lung injury is a critical acute respiratory distress syndrome (ARDS) with high morbidity and mortality. MicroRNAs (miRNAs) have been demonstrated to play important roles regulating acute lung injury development. In this study, we found that the expression of miR-598 was significantly upregulated in the lung tissues of mice with lipopolysaccharide (LPS)-induced acute lung injury. Both loss-of-function and gain-of-function studies were performed to evaluate the function of miR-598 in acute lung injury. The results showed that inhibition of miR-598 attenuated inflammatory response, oxidative stress, and lung injury in mice treated with LPS, while overexpression of miR-598 exacerbated the LPS-induced acute lung injury. Mechanistically, transcription factor Early B-cell Factor-1 (Ebf1) was predicted and validated as a downstream target of miR-598. Overexpression of Ebf1 attenuated LPS-induced production of inflammatory cytokine TNF-α and IL-6, ameliorated LPS-induced oxidative stress, promoted proliferation, and inhibited apoptosis in murine lung epithelial-15 (MLE-15) cells. Moreover, we demonstrated that Ebf1 knockdown abolished the protective effect of miR-598 inhibition in LPS-treated MLE-15 cells. In summary, miR-598 inhibition ameliorates LPS-induced acute lung injury in mice through upregulating Ebf1 expression, which might provide potential therapeutic treatment for acute lung injury.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute lung injury is a critical acute respiratory distress syndrome (ARDS) with high morbidity and mortality (Wilson and Calfee 2020; Butt et al. 2016). The pathogenesis of acute lung injury involves the damage of both vascular endothelium and alveolar epithelium (Johnson and Matthay 2010). In the past decades, great efforts have been made to develop novel treatment for acute lung injury. However, no pharmacological treatment has been approved and current treatment is still supportive such as protective mechanical ventilation (Nieman et al. 2020; Xiong et al. 2016; Ortiz-Diaz et al. 2013). Thus, it is important to understand the pathogenesis of acute lung injury, and there is an urgent unmet medical need for acute lung injury treatment.
Animal models have been widely used to study the mechanisms and therapeutic treatment of acute lung injury (Matute-Bello et al. 2008; Windsor et al. 1993). Lipopolysaccharide (LPS) is a main component of Gram-negative bacterial cell walls, which could induce an inflammatory response in the lung and result in similar pathological characterization of acute lung injury in humans (Maeda et al. 2020; Lee and Downey 2001). Intratracheal administration of LPS has been widely used to establish the acute lung injury model in mice and explore the novel treatment for acute lung injury (Matute-Bello et al. 2008; Zhang et al. 2021; Jiang et al. 2017).
MicroRNAs (miRNAs) are a class of small non-coding RNAs involved in various biological processes (Hombach and Kretz 2016). Accumulating evidence suggest that miRNAs play important roles in the pathogenesis of acute lung injury (Rajasekaran et al. 2016; Cao et al. 2016). For instance, miRNA-454 has been demonstrated to play a protective role in LPS-induced acute lung injury (Tao et al. 2016). In contrast, miRNA-127 could regulate macrophage polarization and exacerbate acute lung injury via activating JNK signaling pathway (Ying et al. 2015). In addition, miRNAs could be utilized as biomarkers for diagnosis and therapeutic treatment (Ferruelo et al. 2018). MiR-598 has been studied as a tumor suppresser in gastric and lung cancers (Xu et al. 2019; Liu et al. 2018; Yang et al. 2018). MiR-598 was found to suppress cancer cell proliferation, migration, and invasion, and regulate the expression of epithelial–mesenchymal transition (EMT) hallmark genes, which are involved in the tumorigenesis of non-small cell lung cancer cancers (Xu et al. 2019; Liu et al. 2018; Yang et al. 2018). However, since acute lung injury is closely related to the disruption of the epithelial and endothelial barrier, the function of miR-598 during such injury rather than tumorigenesis of lung epithelial cells has not been defined.
In this study, we found that the expression of miR-598 was significantly upregulated in the lung tissues of mice with LPS-induced acute lung injury. By using both in vivo LPS-induced acute lung injury model and in vitro MLE-15 cell assays, we demonstrated that miR-598 inhibition ameliorates LPS-induced acute lung injury in mice through upregulating Ebf1 expression, which might provide potential therapeutic treatment for acute lung injury.
Materials and methods
Cell culture and treatment
Murine lung epithelial-15 (MLE-15) cells were purchased from American Type Culture Collection (ATCC, USA) and cultured in DMEM medium (Gibco, USA) supplemented with fetal bovine serum (FBS; Invitrogen, USA) at 37 °C in a 5% CO2 cell incubator. Cells were treated with different concentrations of LPS (Sigma-Aldrich, USA) for varying times and subjected for further analysis.
Transfection
Mmu-miR-598 mimics (5ʹ-GCGGUGAUGCCGAUGGUGCGAGC-3ʹ), miR-598 inhibitor (5′-GCUCCGACCAUCGGCAUCACCGC-3ʹ), and corresponding negative controls si-Ebf1, and relative control siRNA were obtained from GenePharma (Shanghai, China). MLE-15 cells at ~75% confluence were transfected using Lipofectamine 3000 (Invitrogen, USA), following the manufacturer’s protocol. Ebf1 overexpression vector was constructed by cloning the ORF of Ebf1 cDNA into pcDNA3.1 vector (Invitrogen, USA). Typically, 1 × 106 MLE-15 cells in one well of a 6-well plate were transfected with 2.5 µg plasmid or 100 pmol miRNA mimic or inhibitor. At 48 h after transfection, cells were subjected to the additional assays.
Luciferase reporter assay
Full length of Ebf1 3′-UTR was amplified by PCR and cloned into luciferase reporter vector (Promega, USA) and site mutations were generated using QuickChange Site-directed mutagenesis kit (Stratagen, USA). MLE-15 cells were transfected with luciferase reporter vectors, together with miR-598 mimics or negative control. At 48 h after transfection, cells were lysed and the relative luciferase activity was measured using the Dual-Luciferase Reporter Assay kit (Promega, USA).
CCK-8 assay
Cell growth was analyzed by Cell Counting Kit-8 (CCK-8) assay (Dojindo, Japan), following the manufacturer’s protocol. After adding CCK-8 reagent, the absorbance at 450 nm was measured to determine the cell viability.
Apoptosis assay
Terminal deoxynucleotidyltransferase-mediated dUTP-biotin nick end labeling (TUNEL) assay was performed to assess cell apoptosis. Briefly, cells were fixed, permeabilized and incubated with FITC-labeled dUTP and terminal deoxynucleotidyltransferase for 1 h and nuclei was counterstained with DAPI. The TUNEL-positive cells were counted under a fluorescence microscope (SOPTOP, ICX41, China), and data were analyzed with Image-Pro Plus software (Media Cybernetics, USA).
Enzyme-linked immunosorbent assay (ELISA)
Inflammatory cytokine IL-1β, IL-6, and TNF-α in cell culture supernatant or BALF were analyzed by using corresponding ELISA kits purchased from eBioscience (USA), following the manufacturer’s protocol.
Cellular ROS analysis
Cellular ROS was determined by flow cytometry analysis of DCFDA staining. Briefly, transfected cells were incubated with PBS or LPS, together with 10 μM H2DCFDA (molecular probes, Eugene) at 37 °C for 15 min. Cells were collected after treatment with 0.25% Trypsin–EDTA (Thermo Fisher, USA) and washed with PBS, followed by flow cytometry analysis of mean fluorescence intensity for DCFDA staining. Cell samples in PBS were excited with a 488-nm argon ion laser in a flow cytometer (Coulter-XL, Beckman, USA), and the emission was measured at 525 nm. At least 10,000 singlet cells based on the FSC-A/FSC-H and SSC-A/SSC-H parameters were gated for analysis using the EXP032 ADC Analysis Software (Beckman, USA).
MDA/SOD/GSH/LDH activity analysis
The levels of antioxidant and apoptotic markers, including malondialdehyde (MDA), superoxide dismutase (SOD), glutathione peroxidase (GSH), and lactate dehydrogenase (LDH), in culture supernatants of lung tissues were determined with commercial ELISA kits and other enzymatic activity measurement kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China), according to the manufacturer’s instructions.
Acute lung injury animal model
BALB/c mice at 5–6 weeks old were purchased from Shanghai Laboratory Animal Center (Shanghai, China). Randomly grouped mice received 1.4% isoflurane in 100% oxygen, and were intratracheally administrated with 5 mg/kg of LPS (Sigma–Aldrich, USA) to establish the acute lung injury model. For the treatment group, mice (n = 5) were injected with antagomir-598 (30 mg/kg/day) or antagomir control via the tail vein for a consecutive 3 days. The efficiency was analyzed 1 week later in murine lungs. All animal experiment were approved by the Animal Care and Use Committee of Shaanxi University of Chinese Medicine.
Haematoxylin and eosin (H&E) staining
The left lungs of the mice were harvested after LPS treatment and fixed with 4% PFA, followed by embedding in paraffin, sectioning into 5-μm sections, and staining with haematoxylin and eosin. The histopathological analysis was performed under a microscope (SOPTOP, CX40, China) by two pathologists who were blinded to the study. Image data were analyzed with Image-Pro Plus software (Media Cybernetics, USA). Lung injury scores were assessed by the following criteria: cellular infiltration, alveolar wall thickness, and hemorrhage, which were each scored from 0 to 4 (0 no injury, 1 injury in 25% of the field, 2 injury in 50%, 3 injury in 75%, 4 injury throughout the field). Areas were determined in a blinded manner in five equally spaced fields in each lung section. Counts of the average score for each lung section were summed and used as the ALI score.
Evaluation of lung permeability
Lung permeability was analyzed by using the Evans blue dye extravasation method, as previously described (Duan et al. 2012). Briefly, Evans blue dye was injected into the external jugular vein, and then each lung was perfused with PBS. Evans blue dye was extracted using formamide for 18 h at 60 °C and then the absorbance at 620 nm was analyzed.
Lung wet-to-dry ratio analysis
Right lung was harvested from each mouse treated with LPS and the weight of the wet lung was recorded. The lung was incubated at 60 °C for 4 days to remove moisture, and the dry lung weight was recorded to calculate the wet-to-dry ratio.
Myeloperoxidase activity assay
The myeloperoxidase activity in lung tissue was measured by using a myeloperoxidase activity assay kit (Nanjing Jiancheng, China) following the manufacturer’s protocol.
Real-time quantitative polymerase chain reaction (RT-qPCR)
Total RNA was purified using miRNeasy mini kit (Qiagen, Germany) and reverse-transcribed using miRNA specific stem-loop primers (Applied Biosystems, USA). RT-qPCR was performed using SYBR Master mixture (Applied Biosystems, USA) on a QuantStudio 6 real-time PCR machine (ThermoFisher, USA). U6 or β-actin was used as an internal control. The specific primer sequences are listed below: miR-598, forward 5ʹ-GCGGUGAUGCCGAUGGUGCGAGC-3ʹ and reverse, universal PCR Reverse Primer (cat. no. B532451; Sangon Biotech Co., Ltd.), U6, forward 5ʹ-CTCGCTTCGGCAGCACA-3ʹ and reverse 5ʹ-AACGCTTCACGAATTTGCGT-3ʹ, Ebf1, forward 5ʹ-AGATTGAGAGGACGGCCTTTGT-3ʹ and reverse 5ʹ-TCTGTCCGTATCCCATTGCTG-3ʹ, and β-actin, forward 5ʹ-CGTGGGCCGCCCTAGGCACCA-3ʹ and reverse 5ʹ-TTGGCCTTAGGGTTCAGGGGG-3ʹ.
Western blot
Cultured cells or lung tissues were lysed in radioimmunoprecipitation assay (RIPA) buffer with proteinase inhibitor (Beyotime, China). Equal amounts of protein (30 µg per lane) were separated by sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene difluoride (PVDF) membrane (Millipore, USA). The membranes were blocked with 5% non-fat milk in 1× phosphate-buffered saline supplemented with 0.1% Tween 20 detergent (PBST), followed by incubation with indicated primary antibodies overnight at 4 °C. Subsequently, the membranes were further incubated with a secondary antibody conjugated with horseradish peroxidase (HRP). The protein levels were visualized by using a western blot enhanced chemiluminescence (ECL) kit (PIERCE, USA). The following antibodies were used in the study: anti-mouse Ebf1 (1:2000 dilution; #ab126135; Abcam), anti-mouse β-actin (1:2000 dilution; #ab8226; Abcam), and anti-rabbit secondary antibody (1:5000 dilution; #ab6721; Abcam).
Statistical analysis
All data were presented as mean ± standard error of the mean (SEM). The statistical analyses were performed using two-tailed Student t test or analysis of variance (ANOVA) with GraphPad Prism V8 software (Prism, USA). A p value < 0.05 was considered as statistically significant.
Results
MiR-598 expression is upregulated in lung tissues of mice with LPS-induced acute lung injury
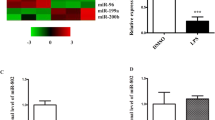
Intratracheal administration of LPS has been widely used to establish the acute lung injury model in mice (Matute-Bello et al. 2008). We established the LPS-induced acute lung injury model as previously described (Fang et al. 2017). As shown in Fig. 1a, LPS treatment led to significant neutrophil infiltration, edema, and necrosis in mouse lung tissue, with a much higher lung injury score. The lung wet-to-dry ratio was markedly high in the lungs from LPS treatment mice (Fig. 1b). LPS treatment also enhanced lung permeability, as demonstrated by Evans blue dye extravasation assay (Fig. 1c). The myeloperoxidase (MPO) activity, which could be used to quantify the neutrophil infiltration to lung, was significantly higher in LPS-treated lung tissue (Fig. 1d). Consistently, we found that LPS administration markedly enhanced the proinflammatory cytokine expression in the bronchoalveolar lavage fluid (BALF), such as IL-1β, IL-6, and TNF-α (Fig. 1e). The above findings suggested that LPS administration in mice successfully established an acute lung injury model. Interestingly, a significant upregulated expression of miR-598 in lung tissue post LPS treatment was identified by qPCR analysis (Fig. 1f). In addition, we found that LPS treatment upregulated the expression of miR-598 in mouse lung tissue dose-dependently (Fig. 1g). We also treated mice with LPS for different durations and found that LPS elevated the expression of miR-598 in a time-dependent manner (Fig. 1h).
MiR-598 expression is upregulated in LPS-induced acute lung injury in mice. a–g Mice were treated with 1 mg/kg LPS or saline control for 24 h (n = 5 per group). Lung tissue samples and BALF were collected for subsequent analysis. a Lung tissues were processed for a histopathological analysis, b wet-to-dry weight ratio, c Evans blue dye extravasation assay, and d myeloperoxidase activity analysis to evaluate the lung injury. e IL-1β, IL-6, and TNF-α levels in BALF were analyzed by ELISA. f The relative expression of miR-598 in lung tissues from saline or LPS treatment group was analyzed by qPCR. g Mice were treated with different doses of LPS and the expression of miR-598 in lung tissue was analyzed by qPCR. h Mice were treated with LPS at 1 mg/kg and over time and the expression of miR-598 was analyzed by qPCR. *p < 0.05, ** p < 0.01, *** p < 0.001
Inhibition of miR-598 attenuates inflammation, oxidative stress, and lung injury in mice treated with LPS
To further characterize the function of miR-598 in LPS-induced acute lung injury, we treated mice with LPS or saline, together with miR-598 antogomir or negative control for 3 consecutive days. Lung injury in mice was analyzed 1 week later. LPS administration led to enhanced expression of miR-598, while antigomir-598 treatment inhibited miR-598 expression (Fig. 2a). Functionally, inhibition of miR-598 markedly decreased the total cells in BALF, with significantly reduced neutrophils and macrophages (Fig. 2b). Consistently, antigomir-598 treatment suppressed the inflammatory response in lung tissues, with decreased expression of IL-1β, IL-6, and TNF-α levels in BALF (Fig. 2c). Moreover, we found that antagonizing miR-598 also suppressed the oxidative stress in the lung tissues of mice treated with LPS, showing markedly reduced MPO activity, malondialdehyde (MDA, lipid peroxidation marker), and glutathione (GSH) levels (Fig. 2d). Inhibition of miR-598 also enhanced superoxide dismutase (SOD) activity, but suppressed lactate dehydrogenase (LDH) activity in the lung tissues of mice treated with LPS (Fig. 2e). Moreover, miR-598 inhibition significantly attenuated lung injury, as evidenced by histopathological analysis (Fig. 2f, g). Suppression of miR-598 consistently decreased the lung wet/dry weight ratio in mice treated with LPS (Fig. 2h).
Inhibition of miR-598 prevents inflammation, oxidative stress, and lung injury upon LPS treatment in mice. Mice were intravenously treated with LPS (5 mg/kg), mmu-miR-598 antagomir (30 mg/kg/day), or control antagomir for a consecutive 3 days, and efficiency was detected 1 week later in murine lungs. a The expression level of miR-598 in lung tissue was analyzed using RT-qPCR. b–f mmu-miR-598 antagomir/ctrl was administrated 12 h prior to LPS (5 mg/kg) treatment. b The numbers of total cells, neutrophils, and macrophages in BALF were analyzed. c The levels of IL-1β, IL-6, and TNF-α in BALF were detected by ELISA (n = 5). d MPO activity, MDA content, and GSH level in lung tissues were analyzed. e Total SOD activity and LDH activity in lung tissues were analyzed. f, g Lung tissues were processed for histopathological analysis. h Lung wet/dry weight ratio in different treatment groups was analyzed. * p < 0.05, ** p < 0.01
Overexpression of miR-598 exacerbates LPS-induced acute lung injury in mice
Next, we overexpressed miR-598 in mice treated with LPS to further characterize its function (Fig. 3a). As shown in Fig. 3b, overexpression of miR-598 significantly increased the total cells, neutrophils, and macrophages in BALF. MiR-598 overexpression also upregulated the inflammatory response in lung tissues, with enhanced expression of IL-1β, IL-6, and TNF-α levels in BALF (Fig. 3c). In addition, we found that miR-598 overexpression also enhanced the oxidative stress in the lung tissues of mice treated with LPS, showing markedly upregulated MDA levels, GSH levels, and enhanced LDH activity, while the SOD activity was decreased (Fig. 3d–f). Moreover, miR-598 overexpression significantly exacerbated lung injury, as evidenced by histopathological analysis (Fig. 3g and h). Consistently, overexpression of miR-598 increased the lung wet/dry weight ratio in mice treated with LPS (Fig. 3i). It is worth noting that intravenous (IV) administration of miR-598 mimics did increase miR-598 expression in other tissues, including spleen, liver, and thyroid (data not shown). Thus, it is possible that overexpressed miR-598 in other organ systems might also contribute to these changes.
Overexpression of miR-598 exacerbates LPS-induced acute lung injury in mice. Mice were intravenously treated with LPS (5 mg/kg), mmu-miR-598 agomir (30 mg/kg/day), or control agomir for a consecutive 3 days, and the efficiency was detected 1 week later in murine lungs. a The expression level of miR-598 in lung tissue was analyzed using RT-qPCR. b–g mmu-miR-598 agomir/control was administrated 12 h prior to LPS (5 mg/kg) treatment. b The numbers of total cells, neutrophils, and macrophages in BALF were analyzed. c The levels of IL-1β, IL-6, and TNF-α in BALF were detected by ELISA (n = 5). d MDA content. e GSH level in lung tissues was analyzed. f Total SOD activity and LDH activity in lung tissues was analyzed. g, h Lung tissues were processed for histopathological analysis. i Lung wet/dry weight ratio in different treatment groups was analyzed. * p < 0.05, ** p < 0.01
Ebf1 is a downstream target of miR-598
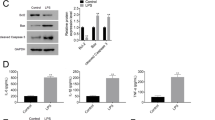
We performed bioinformatics analysis to predict the potential downstream targets of miR-598. As shown in Fig. 4a, the 3′-UTR of Ebf1 had the complementary sequences pairing with miR-598. The WT or mutated 3′-UTR of Ebf1 was constructed into luciferase reporter, and the dual luciferase assay demonstrated that miR-598 mimics specifically suppressed luciferase activity in mouse lung epithelial cell MLE-15 transfected with luciferase reporter vector containing WT 3′-UTR of Ebf1 (Fig. 4b). Overexpression of miR-598 suppressed Ebf1 expression, while inhibition of miR-598 markedly enhanced Ebf1 expression in MLE-15 cells (Fig. 4c and d). In addition, we found that LPS treatment led to the downregulation of Ebf1 in MLE-15 cells in a dose-dependent and time course-dependent manner (Fig. 4e and f). Moreover, the expression of Ebf1 in lung tissues of LPS-treated mice was significantly lower than that in control mice (Fig. 4g). These findings suggest that Ebf1 might be a downstream target of miR-598.
Ebf1 is a downstream target of mmu-miR-598, and Ebf1 expression in MLE-15 cells is downregulated by LPS stimulation. a Sequence alignment of Ebf1 3'UTR and miR-598. WT, wild type; MUT, mutated. b Immortalized mouse lung epithelial MLE-15 cells were transfected with luciferase reporter vector containing WT or mutated 3′-UTR of Ebf1, together with miR-598 mimics or negative control. The dual luciferase assay was performed to validate the interaction between miR-598 and Ebf1. c, d MLE-15 cells were transfected with miR-598 mimics, miR-598 specific inhibitor, or relative negative control miRNA. At 48 h after transfection, cells were harvested to analyze the mRNA and protein levels of Ebf1 by qPCR (c) and western blot (WB) (d), respectively. e, f MLE-15 cells were treated with different concentration of LPS (e) or treated with LPS for different durations, and the relative expression of Ebf1 mRNA was quantitated by RT-qPCR. g The relative expression level of Ebf1 in lung tissues of sham/LPS treated mice at 24 h after challenge was determined by RT-qPCR. * p < 0.05, ** p < 0.01, *** p < 0.001
Ebf1 overexpression attenuates LPS-induced inflammation, oxidative stress, impaired proliferation, and apoptosis in MLE-15 cells
We further investigated the function of Ebf1 in LPS-induced acute lung injury by over expressing Ebf1 in MLE-15 cells (Fig. 5a and b). LPS stimulation promoted TNF-α and IL-6 expression in MLE-15 cells. However, overexpression of Ebf1 attenuated the enhancement of TNF-α and IL-6 in MLE-15 cells (Fig. 5c). In addition, LPS stimulation led to oxidative stress with enhanced ROS production in MLE-15 cells, which could be ameliorated by Ebf1 overexpression (Fig. 5d). Similarly, LPS treatment suppressed cell proliferation and enhanced cell apoptosis, while the Ebf1 overexpression could partially reverse the effect of LPS stimulation (Fig. 5e–g).
Ebf1 overexpression attenuates LPS-induced inflammation, oxidative stress, impaired proliferation, and apoptosis in MLE-15 cells. MLE-15 cells were transduced with control lentivirus or Ebf1-overexpressing lentivirus. a, b The expression levels of Ebf1 mRNA (a) and protein (b) were quantitated by RT-qPCR and western blot assays, respectively. c–g MLE-15 cells after transfection with control lentivirus or Ebf1-expressing virus were further cultured in medium supplemented with PBS or 100 ng/mL LPS. c The TNF-α and IL-6 levels in MLE-15 cell culture supernatant was quantitated by ELISA. d Cellular ROS was determined by flow cytometric analysis of DCFDA staining. e Cell proliferation was determined by CCK-8 assay. f, g Apoptosis was determined by TUNEL staining. Scale bar, 100 µm. * p < 0.05, ** p < 0.01, *** p < 0.001
Ebf1 knockdown abolishes the protective effects of miR-598 inhibition in MLE-15 cells
To further study the functional relationship between miR-598 and Ebf1, we used siRNA to knockdown Ebf1 expression in MLE-15 cells (Fig. 6a). While inhibition of miR-598 could decrease the production of TNF-α and IL-6 induced by LPS stimulation, knockdown of Ebf1 significantly enhanced TNF-α and IL-6 production, and Ebf1 knockdown antagonized the effect of miR-598 (Fig. 6b). Consistently, LPS stimulation led to enhanced ROS production, decreased cell proliferation, and enhanced cell apoptosis, which could suppressed by miR-598 inhibition or further enhanced by Ebf1 knockdown (Fig. 6c–g). However, the protective effect of miR-598 inhibition on oxidative stress, cell proliferation, and cell apoptosis, could be abolished by Ebf1 knockdown (Fig. 6c–g).
The protective effects of miR-598 inhibition in MLE-15 cells were abolished by Ebf1 knockdown. a MLE-15 cells were transfected with Ebf1-specific siRNA or negative control siRNA. The mRNA and protein expression of Ebf1 in MLE-15 cells were analyzed by RT-qPCR and western blot, respectively. b–g MLE15 cells were transfected with negative control siRNA or Ebf1 siRNA oligos, together with control or miR-598 inhibitor. After stimulation with LPS for 24 h, cells were subjected to analyses: b inflammation as quantitated by TNF-α/IL-6 cytokine levels in cell culture supernatant, c, d oxidative stress as quantitated by DCFDA flow cytometry, e proliferation as quantitated by CCK-8 assay, and f, g apoptosis as quantitated by TUNEL staining. Scale bar, 100 µm. * p < 0.05, ** p < 0.01, *** p < 0.001
Discussion
Acute lung injury is a critical acute respiratory distress syndrome with high morbidity and mortality, while current therapeutic treatments are only supportive (Ortiz-Diaz et al. 2013). The discovery of non-coding RNA such as miRNAs and long non-coding RNAs have provided new insights for acute lung injury diagnosis, prevention and treatment. By using LPS-induced acute injury mouse model, we found that miR-598 was significantly upregulated in the lung tissue. In addition, we found miR-598 promoted inflammatory response, oxidative stress, and lung injury in mice treated with LPS. Moreover, we demonstrated that inhibition of miR-598 could ameliorate LPS-induced lung injury via enhancing the expression of Ebf1. These findings indicated that miR-598 could be a therapeutic target for acute lung injury.
MiR-598 has been studied as a tumor suppressor in multiple cancer types, such as colorectal cancer, osteosarcoma, gastric cancer, and also non-small lung cancer (Xu et al. 2019; Liu et al. 2017; Chen et al. 2017). Yang et.al reported that miR-598 negatively regulated Derlin-1 and epithelial-mesenchymal transition in non-small lung cancer (Yang et al. 2018). However, there is no report of miR-598 function on acute lung injury. Previous study has demonstrated that miR-598 was involved in the anti-inflammatory effect of alpha-1 antitrypsin augmentation therapy by inhibiting NF-κB signaling (Hassan et al. 2017). The NF-κB-IL-6 signaling pathway is widely known in mediating vascular inflammation (Brasier 2010). Consistently, we found that LPS-induced acute lung injury led to upregulation of miR-598 and enhanced production of inflammatory cytokine TNF-α and IL-6. In addition, we also revealed that miR-598 modulated oxidative stress and cell proliferation/apoptosis in LPS-induced acute lung injury.
Bioinformatics analysis predicted that Ebf1 was a direct target of miR-598 and luciferase reporter assay, further validated the interaction between miR-598 and Ebf1. Ebf1 is a transcription factor, which is important for early B-cell development (Gao et al. 2009). Previous study has demonstrated that Ebf1 expression was significantly upregulated in the early stages of sepsis (Bhatty et al. 2012). Intriguingly, Ebf1 mRNA was reported to be regulated by multiple altered miRNAs in a rat model of ARDS (Huang et al. 2014). Here we confirmed that overexpression of Ebf1 could attenuate LPS-induced inflammation, oxidative stress, impaired proliferation, and apoptosis in MLE-15 via antagonizing the effect of miR-598. It should be noted that due to the multiple-to-multiple relationship between miRNAs and target mRNAs, we could not exclude the possibility that miR-598 has multiple targets, including Ebf1 contributing to the LPS-induced acute lung injury, while Ebf1 could also be regulated by multiple miRNAs.
In conclusion, the current study proved that miR-598 was significantly enhanced in the lung of mice upon LPS treatment, while inhibition of miR-598 attenuated the acute lung injury induced by LPS administration. MiR-598 exacerbated LPS-induced acute lung injury via negatively regulating Ebf1 expression. These findings unveil a novel role of miR-598/Ebf1 in acute lung injury, which could be utilized to develop novel therapeutic treatment for acute lung injury.
Availability of data and materials
The datasets used and/or analyzed during the current study available from the corresponding author on reasonable request.
References
Bhatty M, Fan R, Muir WM, Pruett SB, Nanduri B (2012) Transcriptomic analysis of peritoneal cells in a mouse model of sepsis: confirmatory and novel results in early and late sepsis. BMC Genomics 13:509. https://doi.org/10.1186/1471-2164-13-509
Brasier AR (2010) The nuclear factor-kappaB-interleukin-6 signalling pathway mediating vascular inflammation. Cardiovasc Res 86(2):211–218. https://doi.org/10.1093/cvr/cvq076
Butt Y, Kurdowska A, Allen TC (2016) Acute lung injury: a clinical and molecular review. Arch Pathol Lab Med 140(4):345–350. https://doi.org/10.5858/arpa.2015-0519-RA
Cao Y, Lyu YI, Tang J, Li Y (2016) MicroRNAs: novel regulatory molecules in acute lung injury/acute respiratory distress syndrome. Biomed Rep 4(5):523–527. https://doi.org/10.3892/br.2016.620
Chen J, Zhang H, Chen Y, Qiao G, Jiang W, Ni P, Liu X, Ma L (2017) miR-598 inhibits metastasis in colorectal cancer by suppressing JAG1/Notch2 pathway stimulating EMT. Exp Cell Res 352(1):104–112. https://doi.org/10.1016/j.yexcr.2017.01.022
Duan Y, Learoyd J, Meliton AY, Leff AR, Zhu X (2012) Inhibition of Pyk2 blocks lung inflammation and injury in a mouse model of acute lung injury. Respir Res 13:4. https://doi.org/10.1186/1465-9921-13-4
Fang Y, Gao F, Hao J, Liu Z (2017) microRNA-1246 mediates lipopolysaccharide-induced pulmonary endothelial cell apoptosis and acute lung injury by targeting angiotensin-converting enzyme 2. Am J Transl Res 9(3):1287–1296
Ferruelo A, Penuelas O, Lorente JA (2018) MicroRNAs as biomarkers of acute lung injury. Ann Transl Med 6(2):34. https://doi.org/10.21037/atm.2018.01.10
Gao H, Lukin K, Ramirez J, Fields S, Lopez D, Hagman J (2009) Opposing effects of SWI/SNF and Mi-2/NuRD chromatin remodeling complexes on epigenetic reprogramming by EBF and Pax5. Proc Natl Acad Sci U S A 106(27):11258–11263. https://doi.org/10.1073/pnas.0809485106
Hassan T, de Santi C, Mooney C, McElvaney NG, Greene CM (2017) Alpha-1 antitrypsin augmentation therapy decreases miR-199a-5p, miR-598 and miR-320a expression in monocytes via inhibition of NFkappaB. Sci Rep 7(1):13803. https://doi.org/10.1038/s41598-017-14310-2
Hombach S, Kretz M (2016) Non-coding RNAs: classification, biology and functioning. Adv Exp Med Biol 937:3–17. https://doi.org/10.1007/978-3-319-42059-2_1
Huang C, Xiao X, Chintagari NR, Breshears M, Wang Y, Liu L (2014) MicroRNA and mRNA expression profiling in rat acute respiratory distress syndrome. BMC Med Genomics 7:46. https://doi.org/10.1186/1755-8794-7-46
Jiang K, Zhang T, Yin N, Ma X, Zhao G, Wu H, Qiu C, Deng G (2017) Geraniol alleviates LPS-induced acute lung injury in mice via inhibiting inflammation and apoptosis. Oncotarget 8(41):71038–71053. https://doi.org/10.18632/oncotarget.20298
Johnson ER, Matthay MA (2010) Acute lung injury: epidemiology, pathogenesis, and treatment. J Aerosol Med Pulm Drug Deliv 23(4):243–252. https://doi.org/10.1089/jamp.2009.0775
Lee WL, Downey GP (2001) Neutrophil activation and acute lung injury. Curr Opin Crit Care 7(1):1–7. https://doi.org/10.1097/00075198-200102000-00001
Liu K, Sun X, Zhang Y, Liu L, Yuan Q (2017) MiR-598: a tumor suppressor with biomarker significance in osteosarcoma. Life Sci 188:141–148. https://doi.org/10.1016/j.lfs.2017.09.003
Liu N, Yang H, Wang H (2018) miR-598 acts as a tumor suppressor in human gastric cancer by targeting IGF-1R. Onco Targets Ther 11:2911–2923. https://doi.org/10.2147/OTT.S166597
Maeda A, Hayase N, Doi K (2020) Acute kidney injury induces innate immune response and neutrophil activation in the lung. Front Med (Lausanne) 7:565010. https://doi.org/10.3389/fmed.2020.565010
Matute-Bello G, Frevert CW, Martin TR (2008) Animal models of acute lung injury. Am J Physiol Lung Cell Mol Physiol 295(3):L379-399. https://doi.org/10.1152/ajplung.00010.2008
Nieman GF, Gatto LA, Andrews P, Satalin J, Camporota L, Daxon B, Blair SJ, Al-Khalisy H, Madden M, Kollisch-Singule M, Aiash H, Habashi NM (2020) Prevention and treatment of acute lung injury with time-controlled adaptive ventilation: physiologically informed modification of airway pressure release ventilation. Ann Intensive Care 10(1):3. https://doi.org/10.1186/s13613-019-0619-3
Ortiz-Diaz E, Festic E, Gajic O, Levitt JE (2013) Emerging pharmacological therapies for prevention and early treatment of acute lung injury. Semin Respir Crit Care Med 34(4):448–458. https://doi.org/10.1055/s-0033-1351118
Rajasekaran S, Pattarayan D, Rajaguru P, Sudhakar Gandhi PS, Thimmulappa RK (2016) MicroRNA regulation of acute lung injury and acute respiratory distress syndrome. J Cell Physiol 231(10):2097–2106. https://doi.org/10.1002/jcp.25316
Tao Z, Yuan Y, Liao Q (2016) Alleviation of lipopolysaccharides-induced acute lung injury by MiR-454. Cell Physiol Biochem 38(1):65–74. https://doi.org/10.1159/000438609
Wilson JG, Calfee CS (2020) ARDS subphenotypes: understanding a heterogeneous syndrome. Crit Care 24(1):102. https://doi.org/10.1186/s13054-020-2778-x
Windsor AC, Mullen PG, Fowler AA (1993) Acute lung injury: what have we learned from animal models? Am J Med Sci 306(2):111–116. https://doi.org/10.1097/00000441-199308000-00010
Xiong B, Wang C, Tan J, Cao Y, Zou Y, Yao Y, Qian J, Rong S, Huang Y, Huang J (2016) Statins for the prevention and treatment of acute lung injury and acute respiratory distress syndrome: a systematic review and meta-analysis. Respirology 21(6):1026–1033. https://doi.org/10.1111/resp.12820
Xu L, Wei B, Hui H, Sun Y, Liu Y, Yu X, Dai J (2019) Positive feedback loop of lncRNA LINC01296/miR-598/Twist1 promotes non-small cell lung cancer tumorigenesis. J Cell Physiol 234(4):4563–4571. https://doi.org/10.1002/jcp.27235
Yang F, Wei K, Qin Z, Liu W, Shao C, Wang C, Ma L, Xie M, Shu Y, Shen H (2018) MiR-598 suppresses invasion and migration by negative regulation of Derlin-1 and epithelial–mesenchymal transition in non-small cell lung cancer. Cell Physiol Biochem 47(1):245–256. https://doi.org/10.1159/000489803
Ying H, Kang Y, Zhang H, Zhao D, Xia J, Lu Z, Wang H, Xu F, Shi L (2015) MiR-127 modulates macrophage polarization and promotes lung inflammation and injury by activating the JNK pathway. J Immunol 194(3):1239–1251. https://doi.org/10.4049/jimmunol.1402088
Zhang Q, Feng A, Zeng M, Zhang B, Shi J, Lv Y, Cao B, Zhao C, Wang M, Ding Y, Zheng X (2021) Chrysosplenol D protects mice against LPS-induced acute lung injury by inhibiting oxidative stress, inflammation, and apoptosis via TLR4-MAPKs/NF-kappaB signaling pathways. Innate Immun 27(7–8):514–524. https://doi.org/10.1177/17534259211051069
Acknowledgements
Not applicable.
Funding
This work was financially supported by the key research and development project of Xianyang City in 2019 [no. 2019k02-38], key research and development plan “COVID-19 Emergency Prevention and Control” science and technology special project of Xianyang City in 2020 [no. 2020k02-85], and Subject Innovation Team of Shaanxi University of Chinese Medicine [no. 2019-YS03].
Author information
Authors and Affiliations
Contributions
Q.Z., J.Z., and H.L. designed the experiments, Q.Z. wrote the manuscript, L.H., Y.L., and Z.Z. performed the experiments, L.H. and W.L. analyzed the data, and J.Z. and H.L. revised the manuscript. All the authors have read and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Shaanxi University of Chinese medicine. Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhao, Q., He, L., Zhang, J. et al. MicroRNA-598 inhibition ameliorates LPS-induced acute lung injury in mice through upregulating Ebf1 expression. Histochem Cell Biol 160, 51–61 (2023). https://doi.org/10.1007/s00418-023-02192-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-023-02192-7