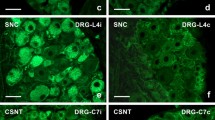
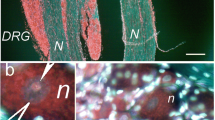
Abstract
Unilateral sciatic nerve compression (SNC) or complete sciatic nerve transection (CSNT), both varying degrees of nerve injury, induced activation of STAT3 bilaterally in the dorsal root ganglia (DRG) neurons of lumbar (L4-L5) as well as cervical (C6–C8) spinal cord segments. STAT3 activation was by phosphorylation at the tyrosine-705 (Y705) and serine-727 (S727) positions and was followed by their nuclear translocation. This is the first evidence of STAT3(S727) activation together with the well-known activation of STAT3(Y705) in primary sensory neurons upon peripheral nerve injury. Bilateral activation of STAT3 in DRG neurons of spinal segments anatomically both associated as well as non-associated with the injured nerve indicates diffusion of STAT3 activation inducers along the spinal cord. Increased levels of IL-6 protein in the CSF following nerve injury as well as activation and nuclear translocation of STAT3 in DRG after intrathecal injection of IL-6 shows that this cytokine, released into the subarachnoid space can penetrate the DRG to activate STAT3. Previous results on increased bilateral IL-6 synthesis and the present manifestation of STAT3 activation in remote DRG following unilateral sciatic nerve injury may reflect a systemic reaction of the DRG neurons to nerve injury.





Similar content being viewed by others
References
Aaronson DS, Horvath CM (2002) A road map for those who don’t know JAK-STAT. Science 296:1653–1655. https://doi.org/10.1126/science.1071545
Bareyre FM, Garzorz N, Lang C, Misgeld T, Buning H, Kerschensteiner M (2011) In vivo imaging reveals a phase-specific role of STAT3 during central and peripheral nervous system axon regeneration. Proc Nat Acad Sci USA 108:6282–6287. https://doi.org/10.1073/pnas.1015239108
Brazda V, Muller P, Brozkova K, Vojtesek B (2006) Restoring wild-type conformation and DNA-binding activity of mutant p53 is insufficient for restoration of transcriptional activity. Biochem Biophys Res Comm 351:499–506. https://doi.org/10.1016/j.bbrc.2006.10.065
Brazda V, Klusakova I, Svizenska IH, Dubovy P (2013) Dynamic response to peripheral nerve injury detected by in situ hybridization of IL-6 and its receptor mRNAs in the dorsal root ganglia is not strictly correlated with signs of neuropathic pain. Mol Pain https://doi.org/10.1186/1744-8069-9-42
Chang HL, Wang HC, Chunag YT, Chou CW, Lin IL, Lai CS, Chang LL, Cheng KI (2017) miRNA expression change in dorsal root ganglia after peripheral nerve injury. J Mol Neurosci 61:169–177. https://doi.org/10.1007/s12031-016-0876-7
Decker T, Kovarik P (2000) Serine phosphorylation of STATs. Oncogene 19:2628–2637. https://doi.org/10.1038/sj.onc.1203481
Dubovy P, Klusakova I, Svizenska I (2002) A quantitative immunohistochemical study of the endoneurium in the rat dorsal and ventral spinal roots. Histochem Cell Biol 117:473–480. https://doi.org/10.1007/200418-002-0411-5
Dubovy P, Klusakova I, Svizenska I, Brazda V (2010) Satellite glial cells express IL-6 and corresponding signal-transducing receptors in the dorsal root ganglia of rat neuropathic pain model. Neuron Glia Biol 6:73–83. https://doi.org/10.1017/s1740925x10000074
Dubovy P, Brazda V, Klusakova I, Hradilova-Svizenska I (2013) Bilateral elevation of interleukin-6 protein and mRNA in both lumbar and cervical dorsal root ganglia following unilateral chronic compression injury of the sciatic nerve. J Neuroinflamm https://doi.org/10.1186/1742-2094-10-55
Erta M, Quintana A, Hidalgo J (2012) Interleukin-6, a major cytokine in the central nervous system. Int J Biol Sci 8:1254–1266. https://doi.org/10.7150/ijbs.4679
Eulenfeld R, Dittrich A, Khouri C, Muller PJ, Mutze B, Wolf A, Schaper F (2012) Interleukin-6 signalling: More than Jaks and STATs. Eur J Cell Biol 91:486–495. https://doi.org/10.1016/j.ejcb.2011.09.010
German CL, Sauer BM, Howe CL (2011) The STAT3 beacon: IL-6 recurrently activates STAT 3 from endosomal structures. Exp Cell Res 317:1955–1969. https://doi.org/10.1016/j.yexcr.2011.05.009
Haghikia A, Ricke-Hoch M, Stapel B, Gorst I, Hilfiker-Kleiner D (2014) STAT3, a key regulator of cell-to-cell communication in the heart. Cardiovasc Res 102:281–289. https://doi.org/10.1093/cvr/cvu034
Heinrich PC, Behrmann I, Haan S, Hermanns HM, Muller-Newen G, Schaper F (2003) Principles of interleukin (IL)-6-type cytokine signalling and its regulation. Biochem J 374:1–20. https://doi.org/10.1042/bj20020407
Hombria JCG, Sotillos S (2008) Disclosing JAK/STAT links to cell adhesion and cell polarity. Semin Cell Dev Biol 19:370–378. https://doi.org/10.1016/j.semcdb.2008.06.002
Hori N, Narita M, Yamashita A, Horiuchi H, Hamada Y, Kondo T, Watanabe M, Igarashi K, Kawata M, Shibasaki M, Yamazaki M, Kuzumaki N, Inada E, Ochiya T, Iseki M, Mori T (2016) Changes in the expression of IL-6-mediated microRNAs in the dorsal root ganglion under neuropathic pain in mice. Synapse 70:317–324. https://doi.org/10.1002/syn.21902
Hylden JLK, Wilcox GL (1980) Intrathecal morphine in mice—a new technique. Eur J Pharmacol 67:313–316. https://doi.org/10.1016/0014-2999(80)90515-4
Joukal M, Klusakova I, Dubovy P (2016) Direct communication of the spinal subarachnoid space with the rat dorsal root ganglia. Ann Anat 205:9–15. https://doi.org/10.1016/j.aanat.2016.01.004
Loffler D, Brocke-Heidrich K, Pfeifer G, Stocsits C, Hackermuller J, Kretzschmar AK, Burger R, Gramatzki M, Blumert C, Bauer K, Cvijic H, Ullmann AK, Stadler PF, Horn F (2007) Interleukin-6-dependent survival of multiple myeloma cells involves the Stat3-mediated induction of microRNA-21 through a highly conserved enhancer. Blood 110:1330–1333. https://doi.org/10.1182/blood-2007-03-081133
Miao T, Wu DS, Zhang Y, Bo XN, Subang MC, Wang P, Richardson PM (2006) Suppressor of cytokine signaling-3 suppresses the ability of activated signal transducer and activator of transcription-3 to stimulate neurite growth in rat primary sensory neurons. J Neurosci 26:9512–9519. https://doi.org/10.1523/jneurosci.2160-06.2006
Murakami M, Hirano T (2012) The pathological and physiological roles of IL-6 amplifier activation. Int J Biol Sci 8:1267–1280. https://doi.org/10.7150/ijbs.4828
Murphy PG, Borthwick LS, Johnston RS, Kuchel G, Richardson PM (1999a) Nature of the retrograde signal from injured nerves that induces interleukin-6 mRNA in neurons. J Neurosci 19:3791–3800
Murphy PG, Ramer MS, Borthwick L, Gauldie J, Richardson PM, Bisby MA (1999b) Endogenous interleukin-6 contributes to hypersensitivity to cutaneous stimuli and changes in neuropeptides associated with chronic nerve constriction in mice. Eur J Neurosci 11:2243–2253. https://doi.org/10.1046/j.1460-9568.1999.00641.x
Ng DCH, Lin BH, Lim CP, Huang GC, Zhang T, Poli V, Cao XM (2006a) Stat3 regulates microtubules by antagonizing the depolymerization activity of stathmin. J Cell Biol 172:245–257. https://doi.org/10.1083/jcb.200503021
Ng YP, Cheung ZH, Ip NY (2006b) STAT3 as a downstream mediator of Trk signaling and functions. J Biol Chem 281:15636–15644. https://doi.org/10.1074/jbc.M601863200
Nicolas CS, Peineau S, Amici M, Csaba Z, Fafouri A, Javalet C, Collett VJ, Hildebrandt L, Seaton G, Choi SL, Sim SE, Bradley C, Lee K, Zhuo M, Kaang BK, Gressens P, Dournaud P, Fitzjohn SM, Bortolotto ZA, Cho K, Collingridge GL (2012) The JAK/STAT pathway is involved in synaptic plasticity. Neuron 73:374–390. https://doi.org/10.1016/j.neuron.2011.11.024
Patodia S, Raivich G (2012) Role of transcription factors in peripheral nerve regeneration. Front Mol Neurosci. https://doi.org/10.3389/fnmol.2012.00008
Pellegrino MJ, Habecker BA (2013) STAT3 integrates cytokine and neurotrophin signals to promote sympathetic axon regeneration. Mol Cell Neurosci 56:272–282. https://doi.org/10.1016/j.mcn.2013.06.005
Pencik J, Pham HTT, Schmoellerl J, Javaheri T, Schlederer M, Culig Z, Merkel O, Moriggl R, Grebien F, Kenner L (2016) JAK-STAT signaling in cancer: from cytokines to non-coding genome. Cytokine 87:26–36. https://doi.org/10.1016/j.cyto.2016.06.017
Qiu J, Cafferty WBJ, McMahon SB, Thompson SWN (2005) Conditioning injury-induced spinal axon regeneration requires signal transducer and activator of transcription 3 activation. J Neurosci 25:1645–1653. https://doi.org/10.1523/jneurosci.3269-04.2005
Schmid AB, Coppieters MW, Ruitenberg MJ, McLachlan EM (2013) Local and remote immune-mediated inflammation after mild peripheral nerve compression in rats. J Neuropath Exp Neurol 72:662–680. https://doi.org/10.1097/NEN.0b013e318298de5b
Schuringa JJ, Schepers H, Vellenga E, Kruijer W (2001) Ser727-dependent transcriptional activation by association of p300 with STAT3 upon IL-6 stimulation. FEBS Lett 495:71–76. https://doi.org/10.1016/s0014-5793(01)02354-7
Schwaiger FW, Hager G, Schmitt AB, Horvat A, Streif R, Spitzer C, Gamal S, Breuer S, Brook GA, Nacimiento W, Kreutzberg GW (2000) Peripheral but not central axotomy induces changes in Janus kinases (JAK) and signal transducers and activators of transcription (STAT). Eur J Neurosci 12:1165–1176. https://doi.org/10.1046/j.1460-9568.2000.00005.x
Shah M, Patel K, Mukhopadhyay S, Xu F, Guo G, Sehgal PB (2006) Membrane-associated STAT3 and PY-STAT3 in the cytoplasm. J Biol Chem 281:7302–7308. https://doi.org/10.1074/jbc.M508527200
Sheu JY, Kulhanek DJ, Eckenstein FP (2000) Differential patterns of ERK and STAT3 phosphorylation after sciatic nerve transection in the rat. Exp Neurol 166:392–402. https://doi.org/10.1006/exnr.2000.7508
Silver DL, Naora H, Liu JS, Cheng WJ, Montell DJ (2004) Activated signal transducer and activator of transcription (STAT) 3: Localization in focal adhesions and function in ovarian cancer cell motility. Cancer Res 64:3550–3558. https://doi.org/10.1158/0008-5472.can-03-3959
Taga T (1996) gp130, a shared signal transducing receptor component for hematopoietic and neuropoietic cytokines. J Neurochem 67:1–10
Tsujino H, Kondo E, Fukuoka T, Dai Y, Tokunaga A, Miki K, Yonenobu K, Ochi T, Noguchi K (2000) Activating transcription factor 3 (ATF3) induction by axotomy in sensory and motoneurons: A novel neuronal marker of nerve injury. Mol Cell Neurosci 15:170–182. https://doi.org/10.1006/mcne.1999.0814
Wakahara R, Kunimoto H, Tanino K, Kojima H, Inoue A, Shintaku H, Nakajima K (2012) Phospho-Ser727 of STAT3 regulates STAT3 activity by enhancing dephosphorylation of phospho-Tyr705 largely through TC45. Genes Cells 17:132–145. https://doi.org/10.1111/j.1365-2443.2011.01575.x
Wu DS, Zhang Y, Bo XN, Huang WL, Xiao F, Zhang XY, Miao TZ, Magoulas C, Subang MC, Richardson PM (2007) Actions of neuropoietic cytokines and cyclic AMP in regenerative conditioning of rat primary sensory neurons. Exp Neurol 204:66–76. https://doi.org/10.1016/j.expneurol.2006.09.017
Wu D, Raafat M, Pak E, Hammond S, Murashov AK (2011) MicroRNA machinery responds to peripheral nerve lesion in an injury-regulated pattern. Neuroscience 190:386–397. https://doi.org/10.1016/j.neuroscience.2011.06.017
Zamboni L, Demartin C (1967) Buffered picric acid-formaldehyde—a new rapid fixative for electron microscopy. J Cell Biol 35:A148
Acknowledgements
We thank Ms. Dana Kutějová, Ms. Jitka Mikulášková, Ms. Marta Lněníčková, Mgr. Jana Vachová and Mr. Lumír Trenčanský for their skillful technical assistance. Supported by grant 16-08508S of The Czech Science Foundation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Dubový, P., Hradilová-Svíženská, I., Klusáková, I. et al. Bilateral activation of STAT3 by phosphorylation at the tyrosine-705 (Y705) and serine-727 (S727) positions and its nuclear translocation in primary sensory neurons following unilateral sciatic nerve injury. Histochem Cell Biol 150, 37–47 (2018). https://doi.org/10.1007/s00418-018-1656-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-018-1656-y