Abstract
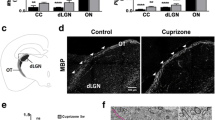
Demyelination induced by cuprizone in mice has served a useful model system for the study of demyelinating diseases, such as multiple sclerosis. Severity of demyelination by cuprizone, however, varies across different regions of the central nervous system; the corpus callosum is sensitive, while the optic nerves are resistant. Here, we investigated the effects of cuprizone on optic nerves, focusing on the axo-glial junctions. Immunostaining for sodium channels, contactin-associated protein, neurofascins, and potassium channels revealed that there were no massive changes in the density and morphology of the axo-glial junctions in cuprizone-treated optic nerves. However, when we counted the number of incomplete junctional complexes, we observed increased numbers of isolated paranodes. These isolated paranodes were immunopositive for both axonal and glial membrane proteins, indicating that they were the contact sites between axons and glia. These were not associated with sodium channels or potassium channels, suggesting the absence of physiological functions. When teased axons from cuprizone-treated optic nerves were immunostained, the isolated paranodes were found at the internode region of the myelin. From these observations, we conclude that cuprizone induces new contacts between axons and myelins at the internode region.








Similar content being viewed by others
References
Acs P, Selak MA, Komoly S, Kalman B (2013) Distribution of oligodendrocyte loss and mitochondrial toxicity in the cuprizone-induced experimental demyelination model. J Neuroimmunol 262:128–131
Bagchi B, Al-Sabi A, Kaza S, Scholz D, O’Leary VB, Dolly JO, Ovsepian SV (2014) Disruption of myelin leads to ectopic expression of K(V)1.1 channels with abnormal conductivity of optic nerve axons in a cuprizone-induced model of demyelination. PloS one 9:e87736
Bhat MA et al (2001) Axon-glia interactions and the domain organization of myelinated axons requires neurexin. IV/Caspr/Paranodin Neuron 30:369–383
Boyle ME, Berglund EO, Murai KK, Weber L, Peles E, Ranscht B (2001) Contactin orchestrates assembly of the septate-like junctions at the paranode in myelinated peripheral nerve. Neuron 30:385–397
Buttermore ED, Thaxton CL, Bhat MA (2013) Organization and maintenance of molecular domains in myelinated axons. J Neurosci Res 91:603–622
Craner MJ, Lo AC, Black JA, Waxman SG (2003) Abnormal sodium channel distribution in optic nerve axons in a model of inflammatory demyelination. Brain 126:1552–1561
Craner MJ, Newcombe J, Black JA, Hartle C, Cuzner ML, Waxman SG (2004) Molecular changes in neurons in multiple sclerosis: altered axonal expression of Nav1.2 and Nav1.6 sodium channels and Na+/Ca2 + exchanger. Proc Natl Acad Sci USA 101:8168–8173
Fancy SP, Chan JR, Baranzini SE, Franklin RJ, Rowitch DH (2011) Myelin regeneration: a recapitulation of development? Ann Rev Neurosci 34:21–43
Fruttiger M et al (1999) Defective oligodendrocyte development and severe hypomyelination in PDGF-A knockout mice. Development 126:457–467
Gudi V et al (2009) Regional differences between grey and white matter in cuprizone induced demyelination. Brain Res 1283:127–138
Hiremath MM, Saito Y, Knapp GW, Ting JP, Suzuki K, Matsushima GK (1998) Microglial/macrophage accumulation during cuprizone-induced demyelination in C57BL/6 mice. J Neuroimmunol 92:38–49
Honjin R, Sakato S, Yamashita T (1977) Electron microscopy of the mouse optic nerve: a quantitative study of the total optic nerve fibers. Archivum histologicum Japonicum = Nihon. soshikigaku kiroku 40:321–332
Howell OW et al (2006) Disruption of neurofascin localization reveals early changes preceding demyelination and remyelination in multiple sclerosis. Brain 129:3173–3185
Kipp M et al (2008) Brain-region-specific astroglial responses in vitro after LPS exposure. J Mol Neurosci 35:235–243
Kipp M, Clarner T, Dang J, Copray S, Beyer C (2009) The cuprizone animal model: new insights into an old story. Acta Neuropathol 118:723–736
Lee KK, de Repentigny Y, Saulnier R, Rippstein P, Macklin WB, Kothary R (2006) Dominant-negative beta1 integrin mice have region-specific myelin defects accompanied by alterations in MAPK activity. Glia 53:836–844
Mason JL, Jones JJ, Taniike M, Morell P, Suzuki K, Matsushima GK (2000) Mature oligodendrocyte apoptosis precedes IGF-1 production and oligodendrocyte progenitor accumulation and differentiation during demyelination/remyelination. J Neurosci Res 61:251–262
Peles E, Salzer JL (2000) Molecular domains of myelinated axons. Curr Opin Neurobiol 10:558–565
Pillai AM, Thaxton C, Pribisko AL, Cheng JG, Dupree JL, Bhat MA (2009) Spatiotemporal ablation of myelinating glia-specific neurofascin (Nfasc NF155) in mice reveals gradual loss of paranodal axoglial junctions and concomitant disorganization of axonal domains. J Neurosci Res 87:1773–1793
Skripuletz T, Gudi V, Hackstette D, Stangel M (2011) De- and remyelination in the CNS white and grey matter induced by cuprizone: the old, the new, and the unexpected. Histol Histopathol 26:1585–1597
Skripuletz T et al (2013) Astrocytes regulate myelin clearance through recruitment of microglia during cuprizone-induced demyelination. Brain 136:147–167
Snaidero N et al (2014) Myelin membrane wrapping of CNS axons by PI(3,4,5)P3-dependent polarized growth at the inner tongue. Cell 156:277–290
Taveggia C et al (2008) Type III neuregulin-1 promotes oligodendrocyte myelination. Glia 56:284–293
Torkildsen O, Brunborg LA, Myhr KM, Bo L (2008) The cuprizone model for demyelination. Acta Neurol Scand Suppl 188:72–76
Yang HJ et al (2009) Region-specific susceptibilities to cuprizone-induced lesions in the mouse forebrain: implications for the pathophysiology of schizophrenia. Brain Res 1270:121–130
Young KM et al (2013) Oligodendrocyte dynamics in the healthy adult CNS: evidence for myelin remodeling. Neuron 77:873–885
Zendedel A, Beyer C, Kipp M (2013) Cuprizone-induced demyelination as a tool to study remyelination and axonal protection. J Mol Neurosci 51:567–572
Zoupi L, Markoullis K, Kleopa KA, Karagogeos D (2013) Alterations of juxtaparanodal domains in two rodent models of CNS demyelination. Glia 61:1236–1249
Acknowledgements
We thank Ms. Hiroko Suzuki, Mr. Minoru Kato, and Mr. Hitoshi Nagai at Mitsubishi Tanabe Pharma Corporation for introducing us to this research field, supplying us some antibodies, and valuable contributions to discussions on the demyelination in animal models.
Funding
This study was funded by the Japanese Society for the Promotion of Science (JSPS KAKENHI Grant Number JP17K07083).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Kojima, W., Hayashi, K. Changes in the axo-glial junctions of the optic nerves of cuprizone-treated mice. Histochem Cell Biol 149, 529–536 (2018). https://doi.org/10.1007/s00418-018-1654-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-018-1654-0